Virulence factors of Legionella pneumophila
- Heat shock protein 60: enhances invasion and cytokine expression in macrophages.
- Outer membrane protein: binding with and delivery of packaged materials into the eukaryotic cells. inhibit the fusion of phagosomes with lysosomes.
- Macrophage infectivity potentiator (Mip) protein promotes adherence and phagocytosis.
- Genes encoding for the type II secretion systems required for intracellular growth is required for full virulence and environmental persistence of Legionella, including intracellular growth , promotes the intracellular infection of lung epithelial cells, dampens the cytokine secretion from infected macrophages and epithelia, and limits the levels of cytokine transcripts in infected macrophages.
- Type IV pili: entry of L. pneumophila into a macrophage, influenced trafficking of the L. pneumophila vacuole.
- Flagella
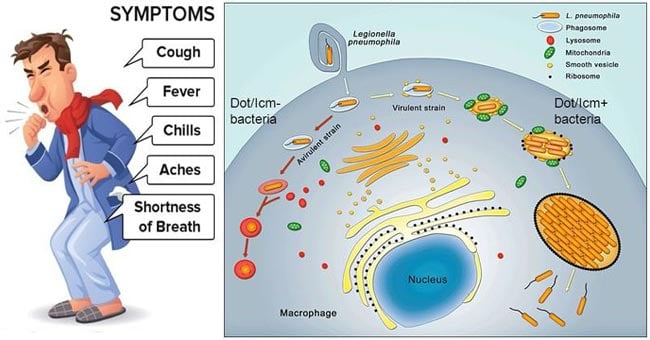
- Dot/icm type IV secretion system (defective organelle trafficking) or icm (intracellular multiplication): secreting and translocating multiple bacterial effector proteins into the vacuolar membrane and cytosol of the host cell. The Dot/Icm T4SS is not only required for intracellular replication and the establishment of the LCV, but also involved in bacterial entry, the inhibition of host cell apoptosis, and the egress of L. pneumophila from host cells.
- MOMPs-adherence and intracellular replication, biofilm development, and formation, horizontal gene transfer.
Pathogenesis of Legionella pneumophila
- L. pneumophila is considered a facultative intracellular pathogen.
- Infection in humans follows the inhalation of fine aerosol (<5 µm particle size) containing organisms that are both viable and virulent.
- Once within the alveoli the legionellae are taken up by resident alveolar macrophages where they survive and replicate within a specialized, membrane-bound vacuole by resisting acidification and evading fusion with lysosomes.
- These phagocytic cells, which are normally regarded as a front line of defense against contaminants, fail to kill or even inhibit the growth of L. pneumophila.
- This ability to infect and replicate in macrophages is critical for pathogenesis.
- The replicative cycle is initiated by binding complement component C3b to an outer membrane porin protein on the bacterial surface.
- The bacteria then binds to the CR3 complement receptors on mononuclear phagocytes, after which the organisms penetrate the cell through endocytosis.
- This mode of entry appears to limit the oxidative burst of the phagocyte and hence enhances the intracellular survival of the legionellae.
- Chemokines and cytokines released by the infected macrophages stimulate a robust inflammatory response that is characteristic of infections with Legionella.
- The organisms proliferate in their intracellular vacuole, phagosomal vacuoles (Legionella-containing vacuole, LCV), and produce proteolytic enzymes (phosphatase, lipase, and nuclease) that eventually kill the host cell when the vacuole is lysed.
- During the late replicative phase the legionella-containing phagosome merges with lysosomes, but replication continues until the host cell is packed with organisms and finally disrupts releasing bacteria to infect further host cells.
- In addition to this process, phagosome survival and organism replication are facilitated by the elaboration of a type IV secretion system call Dot/Icm which is essential for L. pneumophila
- Following replication, the organisms will kill the phagocyte releasing them into the lungs and will again be phagocytized by a mononuclear cell, and multiplication of the organism will increase.
- The presence of iron (transferrin iron) is also essential for the process of intracellular growth of the bacteria.
- The first means by which L. pneumophila acquires iron is through the Fe3+-chelating activity of a siderophore, the most common factor for iron acquisition and promoting lung infection.
- Immunity to a disease is primarily cell-mediated, with humoral immunity playing a minor role.
- The bacteria are not killed until sensitized helper T cells (TH1 cells) activate the parasitized macrophages. Production of interferon-γ is critical for elimination of Legionella
Clinical manifestations of Legionella pneumophila
Legionnaire’s disease
- Legionnaire’s disease is characteristically more severe and, if untreated, promptly causes considerable morbidity, often leading to the death of previously healthy individuals and in immunocompromised patients.
- After an incubation period of 2 to 10 days, systemic signs of an acute illness appear abruptly (e.g., fever and chills, a dry, nonproductive cough, headache).
- Multiorgan disease involving the gastrointestinal tract, central nervous system, liver, and kidneys is common.
- The primary manifestation is pneumonia, with multilobar consolidation and inflammation and microabscesses in lung tissue observed on histopathologic studies.
- Pulmonary function steadily deteriorates in susceptible patients with untreated disease.
- The clinical presentation of pneumonia caused by Legionella is not unique, so laboratory tests are required to confirm the diagnosis.
- The clinical picture is characterized by a multifocal, sometimes necrotizing pneumonia.
- Occurrence is more likely in patients with cardiopulmonary primary diseases or other immunocompromising conditions.
- The onset of LD is generally more insidious than in typical pneumococcal pneumonia, with fever, headaches, and malaise being the first manifestations.
- In about half of the cases, gastrointestinal abnormalities are present.
- Respiratory signs often only appear later in the course of the disease.
- A cough is rare and if present it is generally non-productive early in the illness although later small amounts of purulent, and sometimes bloody sputum, may be expectorated.
- Lung examination usually reveals abnormalities at this stage of the disease, and very often lung consolidation being evident.
- A small pleural effusion, sometimes suspected from the presence of thoracic pains, is present in about one-third of patients but is difficult to recognize on clinical examination.
- Progression of a lung infection may be rapid and be responsible for increasing dyspnoea and acute respiratory failure.
Pontiac fever
- Pontiac fever is an acute, non-pneumonic, flu-like illness that is usually self-limiting.
- The syndrome is characterized by fever and chills, myalgia, malaise, and headache that develop over 6–12 hours and persist for 2–5 days.
- Dizziness, photophobia, neck stiffness, and confusion also occur.
- Respiratory symptoms are much less prominent in patients with Pontiac fever than in those with Legionnaires’ disease and include mild cough and sore throat.
- This illness is self-limited and does not require treatment with antibiotics.
- The attack rate is usually very high (>90%) and the disease affects previously healthy, and often young, individuals.

this is very helpful