Urine contains a large number of different chemicals. Under some circumstances, these chemicals may solidify into salt crystals. This is called crystalluria. Crystals have a characteristic refractile appearance.
Testing for crystals in urine test is often part of a urinalysis, a test that measures different substances in the urine. Normal urine contains many chemicals from which crystals can form, and therefore the finding of most crystals has little importance.
This is to say that, crystals can be seen in the urine of clinically healthy animals or in animals with no evidence of urinary disease (such as obstruction and/or urolithiasis). However, some crystals can be pathologically relevant in certain circumstances. Crystals should be looked for in fresh urine when calculi (stones) in the urinary tract are suspected.
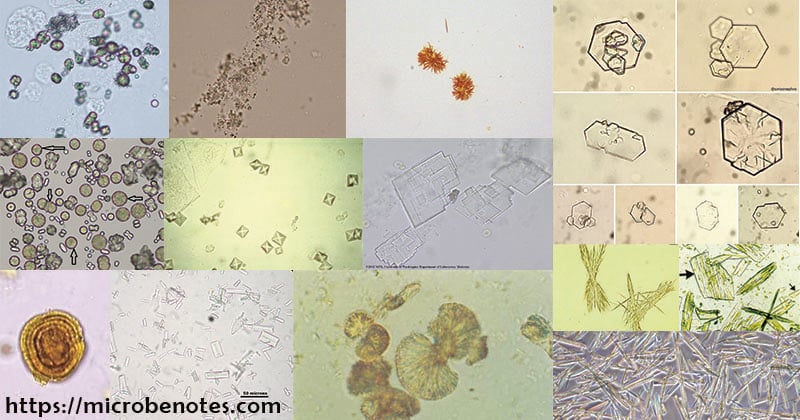
There are many types of crystals present in the urine. Some are regarded as normal crystals while others as abnormal crystals.
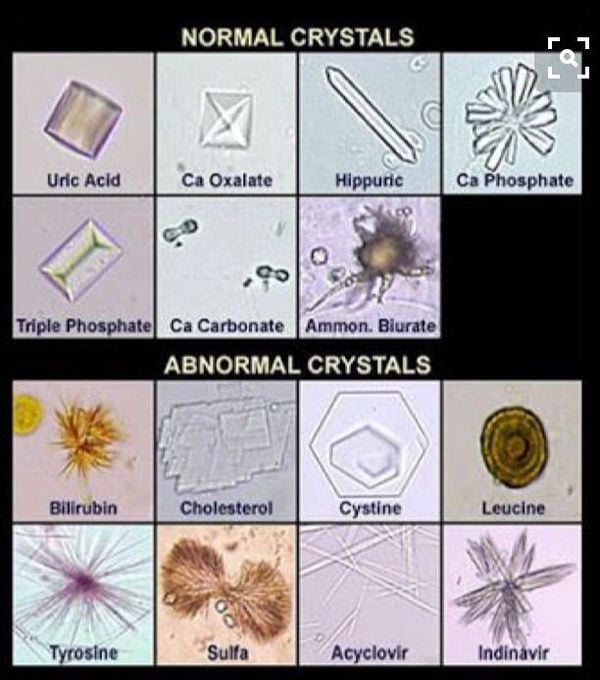
Normal crystals: Uric Acid crystals, calcium oxalate crystals, hippuric acid crystals, calcium phosphate crystals, triple phosphate crystals (struvite), calcium carbonate crystals, ammonium biurate crystals, etc.
Abnormal crystals: Bilirubin crystals, Cholesterol crystals, cystine crystals, leucine crystals, tyrosine crystals, acyclovir crystals, indinavir crystals, etc.
Struvite crystals

- Struvite crystals (magnesium ammonium phosphate, triple phosphate) usually appear as colorless, three-dimensional, prism-like crystals (“coffin lids”).
- They are usually large and can be readily observed at low magnification.
- Struvite crystals are the most common type in urine from dogs and cats. They are often seen in urine from clinically normal individuals.
- Though they can be found in urine of any pH, their formation is favored in neutral to alkaline urine.
- Urinary tract infection with urease-positive bacteria can promote struvite crystalluria (and urolithiasis) by raising urine pH and increasing free ammonia.
Cystine crystals

- They are recognized by their six-sides. The crystals, when found in urine, are often shaped like hexagons and may be colorless.
- They may have either equal or unequal sides and often aggregate in layers.
- Their formation is favored in acidic urine.
- These crystals vary in size from small to medium and should be visible to some extent under low magnification.
- They are soluble in 30% v/v hydrochloric acid (unlike uric acid crystals which they may resemble).
- They can be found in cystinuria, a rare congenital metabolic disorder in which cystine is excreted in the urine.
- Symptoms may include blood in the urine, nausea and vomiting, and pain in the groin or back.
Cholesterol crystals

- They look like clear long rectangles with cut-out corners.
- They’re most likely to appear after the urine sample has been refrigerated.
- Cholesterol crystals can be found in both neutral and acid urine.
- They are insoluble in acids and alkalis but soluble in ether, ethanol, and chloroform.
- They are rarely found except in severe kidney disease or when a lymphatic vessel has ruptured into the renal pelvis.
- They may be caused by renal tubular disease, which can lead to renal failure if left untreated.
- Treatment may involve alkali therapy to help treat chronic metabolic conditions, like renal tubular disease.
Tyrosine crystals
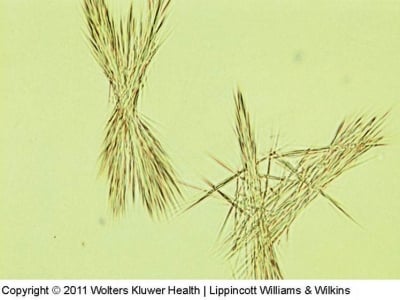
- They are yellow or dark coloured and look like needles massed together.
- They are insoluble in ethanol, ether, and acetone.
- They’re often found in acidic urine, and they may be caused by metabolic disorders like liver disease or tyrosinemia.
- Symptoms of tyrosinemia include difficulty gaining weight, fever, diarrhea, bloody stools, and vomiting.
- Treatment includes exercising, eating a healthy diet, and taking medications that may be able to treat high blood pressure, high cholesterol, and diabetes.
Bilirubin crystals
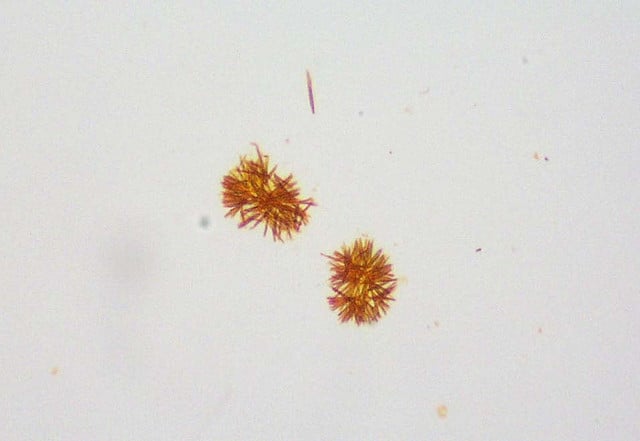
- Bilirubin crystals have a needle-like, granular appearance and are often very small and yellow in color.
- They tend to precipitate onto other formed elements in the urine.
- They are usually small crystals that are usually only observed at high magnification (unless there are large aggregates of crystals).
- High levels of bilirubin or bilirubin crystals in urine could indicate liver disease or poor liver function.
- Other symptoms may include nausea, pain, vomiting, jaundice, and fever.
- Treatment depends on the underlying cause. Medications may be used to change the amount of protein that’s absorbed in the diet, especially in cases of cirrhosis.
Calcium carbonate crystals
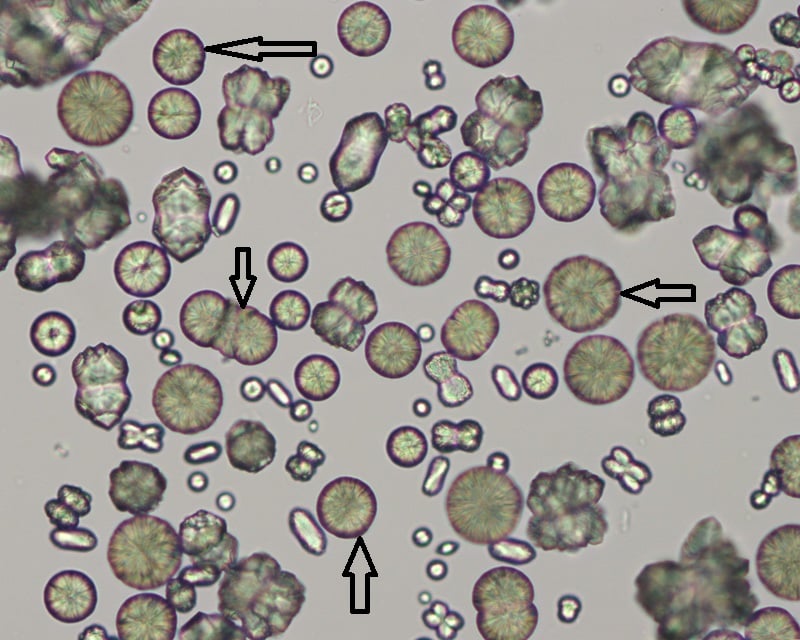
- Calcium carbonate crystals are variably sized crystals that frequently appear as large spheroids with radial striations.
- They can also be seen as smaller crystals with round to ovoid shapes.
- They are colorless to yellow-brown and can impart a brownish tinge to the urine, when they occur in high numbers.
Calcium oxalate crystals
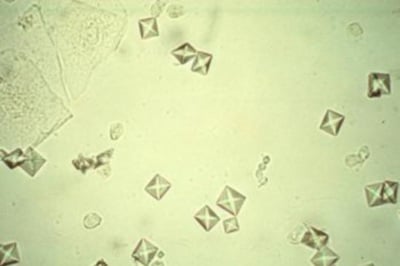
- Calcium oxalate dihydrate crystals typically are colorless squares whose corners are connected by intersecting lines (resembling an envelope).
- They can occur in urine of any pH and thus frequently seen.
- These crystals vary in size from quite large (can be observed under low magnification) to very small (require big magnification to visualize).
- In some cases, large numbers of tiny oxalates may appear as amorphous unless examined at high magnification.
- When found in freshly passed urine they may indicate calculi in the urinary tract.
Ammonium Biurate crystals
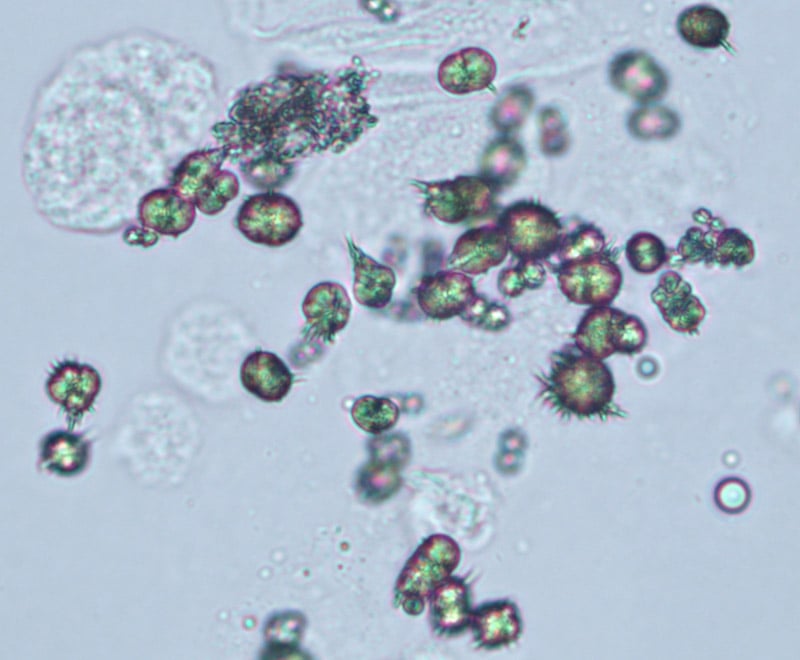
- These crystals are brown spheres with spiky thorns.
- They almost resemble small bugs.
- They’re often found in alkaline urine, but they can also be seen in normal urine.
- Sometimes, ammonium biurate crystals only appear because the urine sample is old or has been poorly preserved.
- Because of this, recollecting the urine sample may be advised if these crystals appear.
Leucine crystals

- These crystals are yellow-brown disks with concentric rings like a tree trunk.
- Leucine crystals are typically not found in healthy urine. They are found in acidic urine.
- They’re typically a symptom of severe liver disease. Other symptoms may include abdominal swelling, vomiting, nausea, disorientation, and malaise.
- Treatment involves improving liver function and health immediately. This will include medications to reduce the risk of bleeding and reduce swelling caused by excess fluid.
Amorphous Crystals
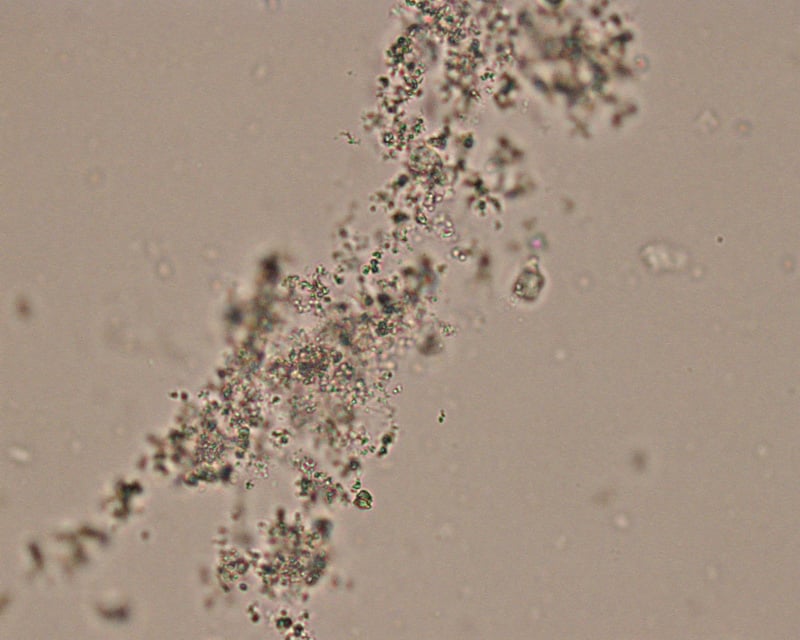
- Amorphous” crystals appear as aggregates of finely granular material without any defining shape at the light microscopic level.
- They can be comprised of urates, phosphates or xanthine.
- They are usually small crystals that are usually only observed at high magnification (unless there are large amounts of them), i.e. they mimic bacteria.
- No specific clinical interpretation can be made based on the finding of amorphous crystals. Small amorphous crystals can be confused with bacterial cocci in some cases, but can be distinguished by gram-staining.
- Degenerating crystals or cells can also resemble “amorphous” crystals.
Other crystals found in urine
- Occasionally sulfonamide crystals are found in the urine of patients being treated with sulphonamides. When deposited in the urinary tract they can cause haematuria and other complications.

- Indinavir is a medication used to treat HIV. It can cause the formation of crystals in the urine. Indinavir crystals may resemble starbursts, rectangular plates, or fans.
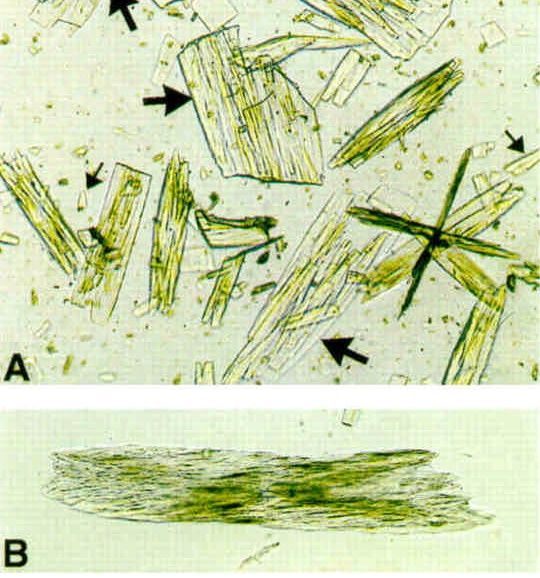
- Uric acid crystals are yellow or pink-brown.
- They can sometimes be found with calculi.

Detecting and Reporting Crystals in Urine
- For crystals that are large, e.g. calcium carbonate and struvite, this assessment is made from low power (10x objective).
- For smaller crystals (e.g. amorphous, calcium oxalate dihydrate), the high power (40x objective) is used.
- Crystals are subjectively quantified in urine as few, moderate and many.
Factors Influencing the Types and Numbers of Urinary Crystals
In vivo factors
- Concentration and solubility of crystallogenic substances contained in the specimen
- Urine pH
- Diet
- Excretion of diagnostic imaging and therapeutic agents
In vitro factors
- Temperature (solubility decreases with temperature)
- Evaporation (increases solute concentration)
- Urine pH (changes with standing and bacterial overgrowth)
References
- Cheesbrough M. (2006). District Laboratory Practice Tropical Countries, 2nd Edition. Cambridge University Press.
- http://www.eclinpath.com/urinalysis/crystals/
- https://medlineplus.gov/lab-tests/crystals-in-urine/
- https://www.healthline.com/health/urine-crystals#types
- http://www.eclinpath.com/atlas/urinalysis/
- https://www.labce.com/spg961161_table_normal_crystals.aspx

This is very archievable to me. may the almighty God gives you more wisdom
It’s so interesting.l would like to request for More notes if available.
The uric acid crystals pictured are not how they appear in urine, the picture is more of the morphological shape of uric acid crystals found in synovial fluid in the case of gout.
Thank you so much for the comments. Yes, they may not be actually from the urine, but its an overall picture of uric acid crystals.
Dear Sir , this is very informative and upto date information about crystals commonly seen and rarely seen in urine samples during microscopy.Your lectures or other postings are always very informative. I am basically Sr.Medical Lab Technologist.
I also teach the students of Medical Laboratory Technology at undergraduate level.
GOD bless you for your dedicated professionalism and I believe when you distribute your knowledge ALLAH gives you more and more knowlege.