What is silver staining?
Silver staining is a special yet powerful staining technique that is used for the detection and identification of proteins in gels.
- This is because silver binds to the chemical terminal or side chains of amino groups i.e carboxyl and sulfhydryl groups.
- It has been used for decades now to separate proteins from polyacrylamide gel electrophoresis.
- The nucleation sites where there are tiny crevices where the free gas-liquid surface is maintained in proteins, promote formaldehyde reduction of silver ions into microscopic silver crystals which facilitate their detection.
- The protein detection by silver staining is a highly sensitive method yet specific and selective for proteins. It produces an image with reduced background and less mass spectrometry interference.
- The general procedure for silver staining includes the fixation of silver, sensitization, impregnation of silver, and development of an image. Several variants of the technique have emerged, some performed within an hour and others taking over 24 hours to complete.
- However, the end stain can remain stable for several weeks before it loses effectiveness for observation.
Principle of silver staining
Silver staining has two major protocols defined by the phase of silver impregnation.
- The alkaline protocol
- This method uses a diamine complex of silver nitrate in an alkaline space made up of ammonium and sodium hydroxide.
- the protein patterns are developed in dilute acidic solutions of formaldehyde.
- The acidic protocol
- This method uses silver nitrate solution in water for gel impregnation and protein patterns are developed in formaldehyde solution under an alkaline environment of ammonia and sodium hydroxide.
- Principally, the silver staining technique is a simple method that works on the selective reduction of silver at the initiation site that is close to the protein molecule, to insoluble metallic silver.
- The stages of silver staining are:
- Fixation where proteins are immobilized and any interfering compounds removed.
- Gel treatment with elements that accelerate protein reactivity to silver and/or silver reduction (sensitization and washing).
- Silver impregnation using plain silver nitrate or ammoniacal silver.
- Gel rinsing and obtaining of silver metal image.
- The image shades produced depends on the number of protein bands attached to the silver.
- The silver-stained protein bands appear dark brown or black depending on the color intensity of the stained silver.
- The variations in color are attributed to the scattered diffractions by silver grains of different sizes.
Properties of silver stain
- It has a boiling point of 2162 °C (3924 °F)
- It has a melting point of 961.78 °C (1763.2 °F)
- The heat of vaporization: 254 kJ/mol
- It has a density of 10.49 g/cm3
- Molar heat capacity: 25.350 J/(mol·K)
Reagents and solutions of Silver staining
- Sample buffer
- 3% acrylamide solution: prepared by mixing 2.0 ml 0.8 M Tris-HCl, pH 8.6, 0.75 ml 38.9% (w/v) acrylamide and 1.1% (w/v) bisacrylamide in 7.25 ml of water, and 8 mg ammonium persulfate
- 20% acrylamide solution: prepared by mixing 2.0 ml 0.8 M Tris-HCl, pH 8.6, 5.0 ml 38.9% acrylamide and 1.1% bisacrylamide in 3 ml of water, and 8 mg ammonium persulfate)
- Fixation solution prepared by mixing 40% ethanol, 10% acetic acid, 50% water)
- The protein treatment solution is prepared by mixing 20% ethanol, 5% acetic acid, 75% water, 4 mg dithiothreitol
- 0.5% dichromate
- 0.1% silver nitrate
- Complex formation solution is prepared by 0.02% paraformaldehyde, 3% sodium carbonate
- 1% acetic acid
Procedure for silver staining
- There are variants of protocols of silver staining technique but we will discuss two of the most important.
- Initially, a silver staining technique is performed after a polyacrylamide gel electrophoresis (SDS-PAGE) has been performed.
Procedure 1: SDS-PAGE
- Add 20 μg of protein in 10 μL of sample buffer and leave it for 60 minutes at room temperature before separation.
- Fill 8 ml each of 3% acrylamide solution and 20% acrylamide solution using a gradient mixer. Pump the solution at a flowing rate of 5ml/min into a glass cuvette.
- Load the protein samples in the gel using a tracking gel, most preferably phenol red.
- Run the SDS-PAGE gel at 4 °C and an electrophoresis current of 15 mA.
- Calculate the protein concentration using bovine serum albumin.
Procedure 2A: Silver staining
- Add the fixation solution for 30 minutes to fix the gel.
- Treat the gel with a protein treatment solution for 30 minutes.
- Rinse the gel with a 0.5% dichromate for 5 minutes.
- Wash the gel with water for 5 minutes.
- Equilibrate the gel with 0.1% silver nitrate for 30 minutes.
- Wash the gel with water for 1 minute.
- Using the complex formation solution, incubate the gel at a pH 12.
- To stop the complex formation, add 1% acetic acid.
- Fix the gels onto glass or polyester sheets for observation and/or storage.
Procedure 2B: Long Silver Nitrate staining
- After SDS-PAGE, fix the gels in 30% ethanol and 10% acetic acid for 60 minutes.
- Renew the fixation bath and leave overnight.
- Sensitize the gel using a tetrathionate sensitizing solution for 45 minutes.
- Rinse the gel with 20% ethanol in two-part (twice), at least 10 minutes for each wash.
- Rinse the gel four times with water, 10 minutes for each wash.
- Impregnate the gel with 12 mM silver nitrate.
- Arrange the gels soaking in silver nitrate in a box half-filled with water, basic developer, and a box containing the stop solution (40 g of Tris and 20 ml of acetic acid per liter).
- Rinse with deionized water, and pull the gel out of the silver solution.
- Dip it in a water bath for 10 seconds and transfer to the basic developer solution.
- Redissolve the precipitates by shaking off the developer-gel containing box.
- After achieving the degree of staining, transfer the gel to the Tris-stop solution for 30 minutes.
- Wash the gel with water and visualize it or store it.
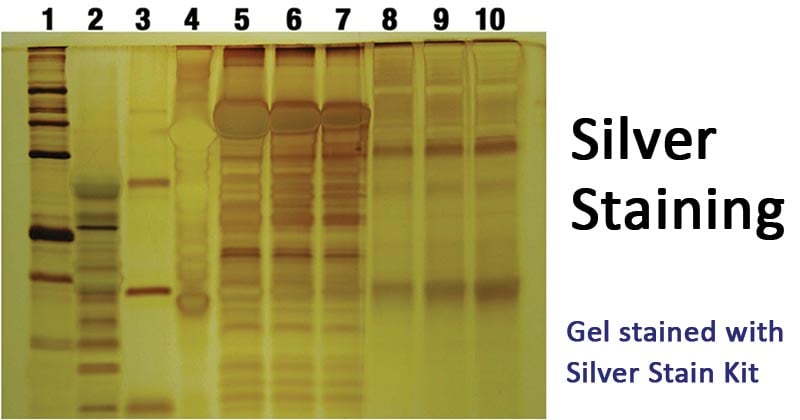
Figure: Gel stained with Silver Stain Kit. Image Source: Polysciences, Inc.
Applications of Silver staining
- Generally, silver staining is and can be used as a diagnostic tool for bacterial and fungal infections such as infections caused by Pseudomonas app, Treponema palladium, Helicobacter pylori, Legionella, Leptospira, Bartonella, Pneumocystis, Candida, Histoplasma, Cryptococcus. All these organisms stain in the silver stain.
- It is used to detect and visualize for proteins
- It can be used to identify the structural differences of proteins
- It can be used quantitatively to define the number of proteins in a sample
- It can also be used for genomic analysis by detecting for DNA and RNA molecules from samples
- It is also used to detect bacterial lipopolysaccharide in SDS-PAGE
- It can be used to detect for fungal lipopolysaccharide such as Histoplasms in liver biopsies
Advantages of silver staining
- It is simple to perform.
- It is cheaper
- It is reliable
- It is very sensitive
- Its permanence and simplicity makes is better than the fluorescent probe
- It can also be used to stain DNA and RNA
Disadvantages of Silver staining
- It has a high and erratic background
- It requires a strong protein-to-protein linkage for variability.
References and Sources
- Hempelmann, Ernst & Krafts, K. (2017). The mechanism of silver staining of proteins separated by SDS polyacrylamide gel electrophoresis. Biotechnic & Histochemistry. 92. 1-7. 10.1080/10520295.2016.1265149.
- Aboulaich, N. (2011). Silver Staining. Bio-101: e26. DOI: 10.21769/BioProtoc.26.
- https://www.sciencedirect.com/topics/biochemistry-genetics-and-molecular-biology/silver-staining
- Chevallet M, Luche S, Rabilloud T. Silver staining of proteins in polyacrylamide gels. Nat Protoc. 2006;1(4):1852-1858. doi:10.1038/nprot.2006.288
- Kumar G. Principle and Method of Silver Staining of Proteins Separated by Sodium Dodecyl Sulfate-Polyacrylamide Gel Electrophoresis. Methods Mol Biol. 2018;1853:231-236. doi:10.1007/978-1-4939-8745-0_26
- Hempelmann E, Krafts K. The mechanism of silver staining of proteins separated by SDS polyacrylamide gel electrophoresis. Biotech Histochem. 2017;92(2):79-85. doi:10.1080/10520295.2016.1265149
- https://arxiv.org/ftp/arxiv/papers/0911/0911.4458.pdf
- https://en.wikipedia.org/wiki/Silver_staining#:~:text=Silver%20staining%20is%20the%20use,electrophoresis%3B%20and%20in%20polyacrylamide%20gels.
- Silver staining protocol by Conduct Science