- Immuno-diffusion is a technique for the detection or measurement of antibodies and antigens by their precipitation which involves diffusion through a substance such as agar or gel agarose. Simply, it denotes precipitation in gel.
- It refers to one of the several techniques for obtaining a precipitate between an antibody and its specific antigen.
- Immunodiffusion reactions are classified based on the:
- Number of reactants diffusing (Single diffusion/Double diffusion)
- Direction of diffusion (One dimension/Two dimension)
- They thus may be of the following types:
- Single diffusion in one dimension
- Single diffusion in two dimensions
- Double diffusion in one dimension
- Double diffusion in two dimensions
Double Immuno-diffusion
- Double immunodiffusion is an agar gel immunodiffusion.
- It is a special precipitation reaction on gels where antibodies react with specific antigens forming large antigen-antibody complexes which can be observed as a line of the precipitate.
- In double immunodiffusion, both the antibody and antigen are allowed to diffuse into the gel.
- After application of the reactants in their respective compartments, the antigen and the antibody diffuse toward each other in the common gel and a precipitate is formed at the place of equivalence.
Double diffusion in one dimension
The method also called Oakley–Fulthrope procedure involves the incorporation of the antibody in agar gel in a test tube, above which a layer of plain agar is placed. The antigen is then layered on top of this plain agar. During incubation, the antigen and antibody move toward each other through the intervening layer of plain agar. In this zone of plain agar, both antigen and antibody react with each other to form a band of precipitation at their optimum concentration.
Double diffusion in two dimensions
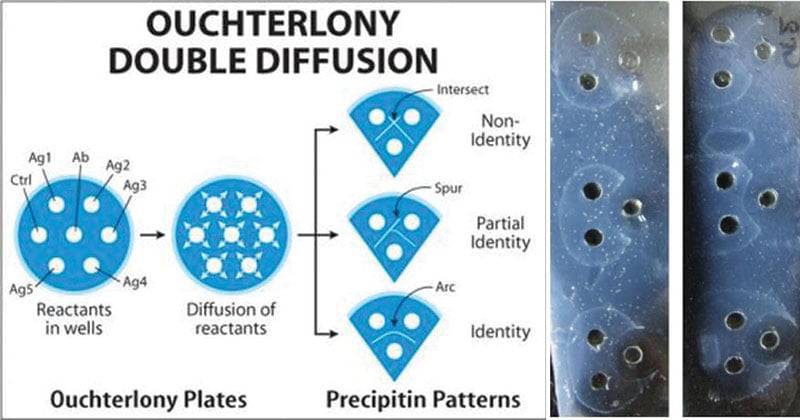
It is more commonly known as Ouchterlony double diffusion or passive double immunodiffusion. In this method, both the antigen and antibody diffuse independently through agar gel in two dimensions, horizontally and vertically.
Objectives
The Ouchterlony double immunodiffusion test may be carried out with one or more of the following objectives:
- To detect antigen-antibody complexes.
- Describe the circumstances under which antigen-antibody complexes precipitate out.
- Detect the presence of an antigen-specific antibody.
- To test the similarity between antigens.
Principle
In the test, an antigen solution or a sample extract of interest is placed in wells bore on gel plates while sera or purified antibodies are placed in other remaining wells (Mostly, an antibody well is placed centrally). On incubation, both the antigens in the solution and the antibodies each diffuse out of their respective wells. In case of the antibodies recognizing the antigens, they interact together to form visible immune complexes which precipitate in the gel to give a thin white line (precipitin line) indicating a reaction.
In case multiple wells are filled with different antigen mixtures and antibodies, the precipitate developed between two specific wells indicate the corresponding pair of antigen-antibodies.
Materials Required
Glass plate or Petri plate, Agarose, Gel borer, Buffer, Antiserum, Antigen solutions
Procedure
- Dissolve 100 mg of agarose in 10 ml of the buffer by boiling to completely dissolve the agarose.
- Cool solution to 55 °C and pour agarose solution to a depth of 1 – 2 mm on a clean glass plate (petri dish or rectangular plate) placed on a horizontal surface.
- Allow the gel to set for 30 minutes.
- Wells are punched into the gel using a gel borer corresponding to the marks on the template if used.
- Fill wells with solutions of antigen and antiserum (of same or different dilutions) until the meniscus just disappears. Antiserum is usually placed in the central well and different antigens are added to the wells surrounding the center well.
- Incubate the glass plate in a moist chamber overnight at 37 °C.
Results
- The presence of an opaque precipitant line between the antiserum and antigen wells indicates antigen-antibody interaction.
- Absence of precipitant line suggests the absence of reaction.
- When more than one well is used there are many possible outcomes based on the reactivity of the antigen and antibody selected.
- The results may be either of the following:
- A full identity (i.e. a continuous line): Line of precipitation at their junction forming an arc represents serologic identity or the presence of a common epitope in antigens.
- Non-identity (i.e. the two lines cross completely): A pattern of crossed lines demonstrates two separate reactions and indicates that the compared antigens are unrelated and share no common epitopes.
- Partial identity (i.e. a continuous line with a spur at one end): The two antigens share a common epitope, but some antibody molecules are not captured by the antigen and traverse through the initial precipitin line to combine with additional epitopes found in the more complex antigen.
- The pattern of the lines that form can determine whether the antigens are the same.
Applications
- It is useful for the analysis of antigens and antibodies.
- It is used in the detection, identification, and quantification of antibodies and antigens, such as immunoglobulins and extractable nuclear antigens.
- Agar gel immunodiffusions are used as serologic tests that historically have been reported to identify antibodies to various pathogenic organisms such as Blastomyces.
- Demonstration of antibodies in serodiagnosis of smallpox.
- Identification of fungal antigens.
- Elek’s precipitation test in the gel is a special test used for demonstration of toxigenicity of Corynebacterium diphtheriae.
References
- https://www.abbexa.com/double-Immunodiffusion
- Bailey, Graham S. (1996). “135: Ouchterlony Double Immunodiffusion”. In Walker, John M. The Protein Protocols Handbook (pdf). VII: Immunochemical Techniques. Totowa, New Jersey: Humana Press. pp. 749–752. Retrieved 2018-07-04.
- Lydyard, P.M., Whelan,A.,& Fanger,M.W. (2005).Immunology (2 ed.).London: BIOS Scientific Publishers.
- Parija S.C. (2012). Textbook of Microbiology & Immunology.(2 ed.). India: Elsevier India.
- amrita.edu,. (2011). Ouchterlony Double Diffusion – Patterns. Retrieved 4 July 2018, from vlab.amrita.edu/?sub=3&brch=70&sim=689&cnt=1

What are the precautions that one needs to take while carrying out immunodiffusion techniques?
Thank you so much for the contents.