The movement of nitrogen between the atmosphere, biosphere, and geosphere in different forms is called the nitrogen cycle and is one of the major biogeochemical cycles.
- It may also be considered as the movement of nitrogen through the food chain from simple inorganic compounds, mainly ammonia, to complex organic compounds.
- This complex cycle involves bacteria, plants, and animals.
- All organisms can convert ammonia (NH3) to organic nitrogen compounds that are compounds containing C–N bonds. However, only a few microorganisms can synthesize ammonia from nitrogen gas (N2).
- Although N2 gas makes up about 80% of the earth’s atmosphere, it is a chemically unreactive compound and thus needs to be changed in order to be utilized by living beings.
- Within the biosphere, there is a balance between total inorganic and total organic forms of nitrogen.
- The conversion of organic to inorganic nitrogen comes about through catabolism, de-nitrification, and decay.
Importance of Nitrogen in Ecosystems
Nitrogen is vital for maintaining life on Earth as it is a key component of essential biomolecules such as amino acids, proteins, nucleic acids, and chlorophyll. Nitrogen manages primary productivity in ecosystems by sustaining plant growth, which is the foundation of the food web. A balanced amount of nitrogen promotes soil fertility, biomass production, and crop yields, thus promoting food security. In aquatic ecosystems, nitrogen is also vital to the development of phytoplankton, which power oceanic primary production and carbon cycling. Yet biologically available nitrogen is frequently in shortage, forming a choke on ecosystem productivity. Therefore, nitrogen is both a resource and a controller in ecological systems, having an impact on biodiversity, species interaction, and biogeochemical fluxes.
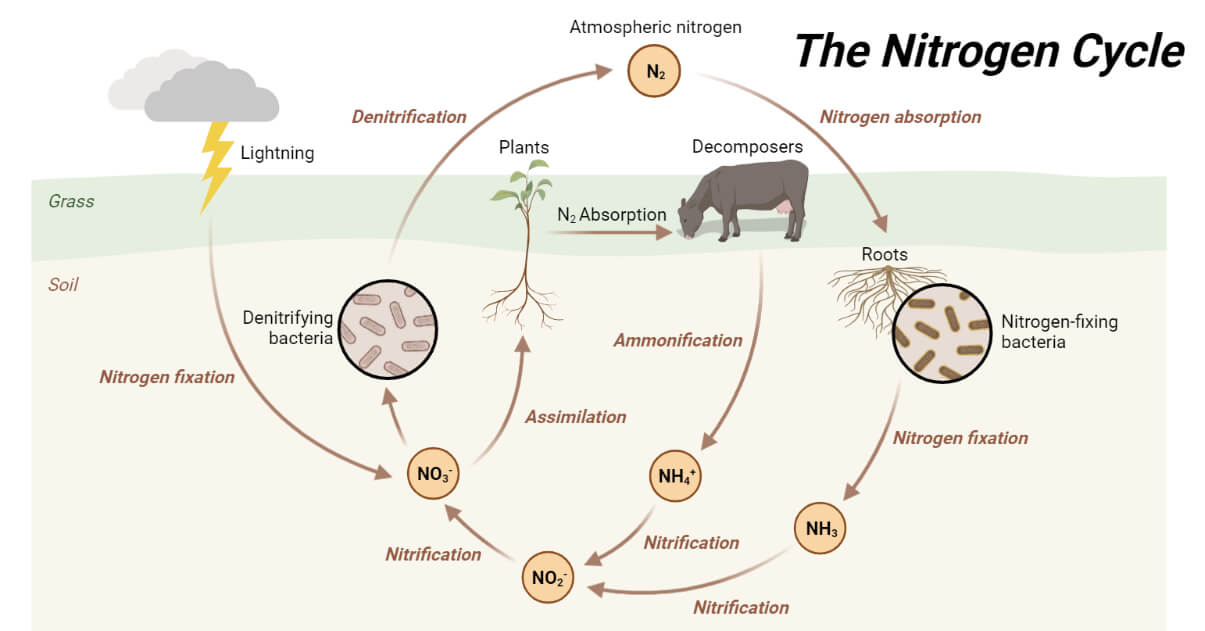
Steps of Nitrogen Cycle
1. Nitrogen Fixation
The first stage in the nitrogen cycle is the reduction of N2 gas to ammonia, a process called nitrogen fixation.
- The process of converting atmospheric N2 gas into ammonia is carried out by only a few microorganisms, termed diazotrophs which have an enzyme called “nitrogenase” that combines nitrogen atoms with hydrogen atoms.
- These are some free-living soil bacteria such as Klebsiella and Azotobacter, cyanobacteria (blue-green algae), and the symbiotic bacteria (mainly Rhizobium).
- The amount of N2 fixed by these diazatrophic microorganisms has been estimated to be in the order of 1011 kg per year, about 60% of the earth’s newly fixed nitrogen.
- Lightning and ultraviolet radiation fix another 15%, with the remainder coming from industrial processes.
- Ammonia can also be obtained by the reduction of the nitrate ion (NO3–) that is present in the soil.
- Nitrate reduction can be carried out by most plants and microorganisms.
- The ammonia resulting from these two processes can then be assimilated by all organisms.
Reaction: N2 + 3 H2 –> <– 2 NH3
- This process is carried out by the nitrogenase complex, which consists of a reductase and an iron–molybdenum-containing nitrogenase.
- At least 16 ATP molecules are hydrolyzed to form two molecules of ammonia.
- Leghemoglobin is used to protect the nitrogenase in the Rhizobium from inactivation by O2.
Classification of Nitrogen Fixation
- Atmospheric fixation: A natural process where the energy released from lightning smashes nitrogen into nitrogen oxides and is utilized by plants.
- Industrial nitrogen fixation: This is a human-made substitute that facilitates nitrogen fixation through the use of ammonia. Ammonia is formed through the direct reaction of nitrogen and hydrogen and subsequently converted into different fertilizers like urea.
- Biological nitrogen fixation: We already know that nitrogen cannot be directly utilized from the air by plants and animals. Some bacteria such as Rhizobium and blue-green algae convert the unusable state of nitrogen into other forms that can be more easily utilized. These nitrogenous substances are fixed into the soil by these microbes.
2. Nitrification
- Nitrification is a two-step process in which ammonia (NH3/ NH4+) is converted to nitrate (NO3– ).
- First, the soil bacteria Nitrosomonas and Nitrococcus convert NH3 to NO2–, and then another soil bacterium, Nitrobacter, oxidizes NO2– to NO3–.
- These bacteria are “chemotrophs” who obtain their energy from volatile chemicals. They gain energy through these conversions, both of which require oxygen to occur.
Nitrosomonas Nitrobacter
[NH4+] ————————— > NO2- ————————-> NO3-
Ammonium pH 4 – 10 nitrite pH 6 – 9 nitrate
Optimum conditions for nitrification:
- Adequate aeration
- Optimum temperature 25 – 35oC
- Adequate soil moisture
- Adequate exchangeable bases – particularly Calcium (Ca)
- NPK availability
- Requires a low C/N ratio
The Nitrification reaction is given below:
2NH4+ + 3O2 → 2NO2– + 4H+ + 2H2O 2NO2– + O2 → 2NO3–
3. Nitrogen Assimilation
- The next step in the nitrogen cycle is the assimilation of inorganic nitrogen, into organic nitrogen-containing compounds.
- It is the process by which plants and animals incorporate the NO3– and ammonia formed through nitrogen fixation and nitrification.
- All organisms assimilate ammonia via two main reactions catalyzed by glutamate dehydrogenase and glutamine synthetase giving rise to the amino acids glutamate (Glu) and glutamine (Gln), respectively.
- The amino nitrogen in Glu and the amide nitrogen in Gln are then used in further biosynthetic reactions to give rise to other compounds.
Glutamate dehydrogenase
Glutamate dehydrogenase catalyzes the reductive amination of the citric acid cycle intermediate α-ketoglutarate. Although the reaction is reversible, the reductant used in the biosynthetic reaction is NADPH. This enzyme is also involved in the catabolism of amino acids.
Glutamine synthetase
Glutamine synthetase catalyzes the incorporation of ammonia into glutamine, deriving energy from the hydrolysis of ATP. This enzyme is named a synthetase, rather than a synthase because of the reaction couples bond formation with the hydrolysis of ATP. In contrast, a synthase does not require ATP.
- Plants take up these forms of nitrogen through their roots and incorporate them into plant proteins and nucleic acids. Animals are then able to utilize nitrogen from the plant tissues.
4. Ammonification
- Assimilation produces large quantities of organic nitrogen, including proteins, amino acids, and nucleic acids.
- Ammonification is the conversion of organic nitrogen into ammonia.
- Here, organic nitrogen from dead plants and soil organisms is converted to ammonium (NH4+) by mineralization. Mineralization is the decomposition of organic matter to NH4+ which is a useable nutrient ion.
- Many different soil organisms are involved in the ammonification process including bacteria and fungi. These soil organisms break down organic matter using the carbon and energy produced, but nitrogen is released.
- Ammonia is also released by the process of excretion into the environment and is then available for either nitrification or assimilation.
5. Denitrification
- Denitrification refers to the change (via denitrifying bacteria) of nitrate to gaseous forms of nitrogen such as nitrogen gas (N2) or nitrous oxide (N2O).
- The process is rapid and large amounts of N2 are lost from the soil system and end up in the atmosphere.
- This process only occurs where there is little to no oxygen, such as deep in the soil near the water table.
- Hence, areas such as wetlands provide a valuable place for reducing excess nitrogen levels via denitrification processes.
Conditions favoring denitrification:
- Lack of adequate O2
- Requires energy source of oxidizable organic matter for bacteria
- Warm, slightly acidic soils.
Significance of Nitrogen Cycle
The nitrogen cycle illustrates the relationship between different forms of nitrogen in the soil, water, air, and living organisms. It is considered to be a cycle since nitrogen moves around from place to place in different forms but is always present. It is important for the following reasons:
- Neither plants nor animals can obtain nitrogen directly from the atmosphere and thus depend on the nitrogen fixation process.
- Nitrogen (N) is an essential component of DNA, RNA, and proteins, the building blocks of life. All organisms require nitrogen to live and grow.
- Nitrogen is a key part of amino and nucleic acids and also an important part of ATP, which is the basic energy molecule for living things.
Human Influence on the Nitrogen Cycle
Human activities during the last century have profoundly changed the global nitrogen cycle. Industrial fixation of nitrogen by the Haber-Bosch process, which is employed to produce synthetic fertilizers, has added significant amounts of reactive nitrogen to land systems. Apart from this, burning fossil fuels emits nitrogen oxides (NO), which are responsible for smog, acid rain, and the production of ground-level ozone.
The increase in agriculture and the extensive application of nitrogenous fertilizers have caused nutrient runoff into freshwater and marine systems, and eutrophication. This results in algal blooms, hypoxia, and loss of biodiversity. Livestock production also causes nitrogen pollution due to ammonia emissions from manure.
Consequences of nitrogen cycle disruption
Disturbing the nitrogen cycle has a long-term ecological and social impact. Excess reactive nitrogen in water bodies and soils results in nutrient imbalances that impact plant community composition, diminish biodiversity, and destabilize food webs. Aquatic eutrophication causes areas of low oxygen (dead zones) that result in the mass death of aquatic life. Nitrogen compounds in the atmosphere like nitrous oxide (N2O) are powerful greenhouse gases that contribute to climate change and ozone depletion. Excess nitrogen inputs also cause soil acidification, which destroys soil health and reduces the ability of plants to absorb nutrients, making sustainable agriculture challenging.
Mitigation Strategies Against Nitrogen Pollution
- Solutions to nitrogen pollution depend on a mix of technological, ecological, and policy-based measures.
- Optimizing the application of fertilizer according to crop needs and soil analysis is a key approach in precision agriculture that can help minimize excess nitrogen applications. Implementation of organic farming and biological nitrogen fixation using legumes are sustainable alternatives.
- Buffer zones around bodies of water and constructed wetlands can trap nitrogen runoff and curtail eutrophication.
- Environmental regulation and policymaking, e.g., reducing NO emissions and promoting sustainable agriculture, are vital in the control of anthropogenic nitrogen flux.
- Public education and public awareness campaigns can also have a major role in ensuring the proper use of nitrogen.
- There is a need for an integrated, systems-based solution to prevent the harmful effects of disruption of the nitrogen cycle while keeping food production and ecosystem services intact.
Conclusion
The nitrogen cycle is an essential process that supports life on our planet. It facilitates the conversion of inert atmospheric nitrogen into biologically usable forms, fuels primary productivity, and maintains the delicate web of life in terrestrial and aquatic ecosystems. With increasing demand for agricultural productivity, the need to apply sustainable policies and practices by ecological principles is pressing. Improving our knowledge of nitrogen dynamics and incorporating scientific innovations into ecosystem-based management tactics will be essential in bringing balance to the nitrogen cycle. Finally, protecting the integrity of the nitrogen cycle is essential not just for resilience in the environment but also for the well-being and health of present and future generations.
Nitrogen Cycle Key Terms
- NITROGEN FIXATION: The process of nitrogen gas is converted into ammonia by lightning or bacteria.
- NITRIFICATION: The process of ammonia being converted into nitrates and nitrites.
- AMMONIFICATION: The process of dead organisms breaking down into ammonia.
- DENITRIFICATION: The process of nitrates and nitrites being converted into nitrogen gas or gaseous nitrogen.
References
- Admin. (2023, May 10). Nitrogen cycle. BYJUS. https://byjus.com/biology/nitrogen-cycle/
- What is the nitrogen cycle and why is it key to life? (n.d.). Frontiers for Young Minds. https://kids.frontiersin.org/articles/10.3389/frym.2019.00041
- Nitrogen – Understanding global change. (2020, September 10). Understanding Global Change. https://ugc.berkeley.edu/background-content/nitrogen/
- The Editors of Encyclopaedia Britannica. (2025, April 25). Nitrogen fixation | Definition, Process, Examples, Types, & Facts. Encyclopedia Britannica. https://www.britannica.com/science/nitrogen-fixation
- The nitrogen cycle. (n.d.). Science Learning Hub. https://www.sciencelearn.org.nz/resources/960-the-nitrogen-cycle
- Dolla, T. H., Matthews, T., Maxakato, N. W., Ndungu, P., & Montini, T. (2022). Recent advances in transition metal sulfide-based electrocatalysts and photocatalysts for nitrogen fixation. Journal of Electroanalytical Chemistry, 928, 117049. https://doi.org/10.1016/j.jelechem.2022.117049
- The nitrogen Cycle: processes, players, and human impact | Learn Science at Scitable. (n.d.). https://www.nature.com/scitable/knowledge/library/the-nitrogen-cycle-processes-players-and-human-15644632/
- Lin, J., Chen, N., Yuan, X., Tian, Q., Hu, A., & Zheng, Y. (2020). Impacts of human disturbance on the biogeochemical nitrogen cycle in a subtropical river system revealed by nitrifier and denitrifier genes. The Science of the Total Environment, 746, 141139. https://doi.org/10.1016/j.scitotenv.2020.141139
- Sources and solutions: Agriculture | US EPA. (2025, March 20). US EPA. https://www.epa.gov/nutrientpollution/sources-and-solutions-agriculture
- Hoang, H. G., Thuy, B. T. P., Lin, C., Vo, D. N., Tran, H. T., Bahari, M. B., Le, V. G., & Vu, C. T. (2022). The nitrogen cycle and mitigation strategies for nitrogen loss during organic waste composting: A review. Chemosphere, 300, 134514. https://doi.org/10.1016/j.chemosphere.2022.134514
- David Hames and Nigel Hooper (2005). Biochemistry. Third ed. Taylor & Francis Group: New York.
- https://www.fondriest.com/news/nitrogencycle.htm
- https://www.acs.edu.au/info/sciences/chemical-sciences/what-is-the-nitrogen-cycle.aspx
- https://www.fondriest.com/news/nitrogencycle.htm
- https://biologydictionary.net/nitrogen-cycle/
- https://microbiologynotes.org/nitrogen-cycle/

wow
a very good and energetic article
lavoro sintetico e esauriente . complimenti Sagar Aryal
Avery organise and agood notes,
thanks for the good information Sagar Aryal