Monoclonal Antibodies are the antibodies that are made by identical immune cells that are all clones of a unique parent cell. Monoclonal antibodies can have monovalent affinity, in that they bind to the same epitope.
A technique to produce monoclonal antibodies was devised by Georges Kohler and Cesar Milstein in 1975. The method relies on fusing B cells from an immunized animal (typically a mouse) with an immortal myeloma cell line and growing the cells under conditions in which the unfused normal and tumor cells cannot survive. The resultant fused cells that grow out are called hybridomas; each hybridoma makes only one Immunoglobulin, derived from one B cell from the immunized animal. The antibodies secreted by many hybridoma clones are screened for binding to the antigen of interest, and this single clone with the desired specificity is selected and expanded. The products of these individual clones are monoclonal antibodies, each specific for a single epitope on the antigen used to immunize the animal and to identify the immortalized antibody-secreting clones.
What are Monoclonal Antibodies?
- The body naturally produces antibodies, which are elements of the immune system produced by B-lymphocytes, that bind to foreign proteins in the body known as antigens, which the aim of eliminating them.
- They naturally circulate in the body searching for foreign bodies (antigens) and once they attach to the antigen, they destroy the antigen using various immune mechanisms.
- On the other hand, monoclonal antibodies are proteins prepared in the laboratory to target specific antigens on body cells such as receptors and other foreign proteins on the surface of normal and cancer cells in the body.
- So what are monoclonal antibodies? Monoclonal antibodies are artificial antibodies that are produced from a single clone of cells by fusing B-lymphocytes to myeloma cells. The fusion of B-lymphocytes with myeloma cells by somatic cell hybridization secretes desired antibody-producing elements which are immortalized cell lines known as a hybridoma. These hybridomas produce homogenous monoclonal antibodies.
- Monoclonal antibodies (mAbs) have the ability to recognize unique binding sites (epitopes) found on the specific antigens. This differentiates monoclonal antibodies from polyclonal antibodies i.e monoclonal antibodies are derived from a single B-cell clone to target single epitopes, unlike polyclonal antibodies that target multiple epitopes.
- Monoclonal antibodies (mAbs) have been produced to target receptors or other foreign proteins that are present on the surface of normal cells and cancer cells.
- Monoclonal antibodies are used in the treatment of many diseases including some cancers.
- The specificity of monoclonal antibodies (mAbs) allows them to bind to cancerous cells coupled with a cytotoxic agent such as a strong radioactive agent. The radioactive agent seeks to destroy the cancer cells without harming the healthy ones.
Types of monoclonal antibodies
Being synthetically manufactured to act like human antibodies in the immune system, monoclonal antibodies are prepared in 4 different ways and they are named after what they are made of.
A. Murine monoclonal antibodies
- They were the first monoclonal antibodies to be produced on a lab scale by the hybridoma technology in 1975.
- They were named murine because of their origin from rodent hosts (mice and rats) belonging to the Muridae family.
- Preclinical trials for its use in the treatment of Cryptococcus neoformans.
- The murine mAbs have played a crucial role in the development of modern antibody production techniques and the potential applications of these artificial immunoglobulins in therapeutics and analytical applications.
- In therapeutic applications, they are used as a framework for the development of antibody and engineering techniques that include chimerization, humanization, and development of bispecific antibodies from antibody fragments known as single-chain antibody fragments (scFvs).
- Preclinical trials to use murine mAbs on the capsular polysaccharide and enhance the therapeutic activity of amphotericin B administration.
- The name of their treatments ends with -omab.
B. Chimeric monoclonal antibodies
- These are structural chimeras that are made of a combination of mouse parts and human parts, by fusion. This involves fusing variable regions of one species such as mice and the constant regions of the other species such as the human species.
- Chimeric monoclonal antibodies are produced to reduce immunogenicity and to increase the serum half-life when preparing them for therapeutic reasons.
- Chimeric antibodies retain the original antibody’s antigen specificity and affinity.
- The name of their treatments ends with -ximab.
C. Humanized monoclonal antibodies
- They are an extension of the chimeric monoclonal antibodies whereby, all regions of the mouse antibody in chimeric mAbs are replaced with human ones except for the complementarity-determining regions (CDRs) which are the amino acids that make direct contact with the antigen. This means that humanized mAbs have small parts of the mouse protein that are attached to the human protein.
- The names of treatments end in -zumab.
D. Human monoclonal antibodies
- These are fully human proteins that have been manipulated by molecular biological techniques so as to modify the amino acid sequences.
- This alters the specificity, affinity, or biological functions, acquiring sequences that are not part of the human repertoire.
- The name of the treatments of human mAbs end with -umab.
Types of monoclonal antibodies based on functions
Monoclonal antibodies can also be classified based on the functions they play, such as monoclonal antibodies used in cancer treatment include:
1. Naked monoclonal antibodies
These are mAbs that do not have a drug or radioactive agent attached to them. they are the most common mAbs used in the treatment of cancer. Most naked mAbs attach to the antigens on the cancer cells and others bind to antigens or other non-cancerous cells or free-floating proteins.
These naked mAbs function differently for example:
- Boost immune response against the cancer cells by attaching to the cancer cells and acting as markers for the body’s immune systems to destroy them e.g Alemtuzumab (Campath®) used for treating Chronic lymphocytic leukemia (CLL). This drug Alemtuzumab binds to the CD52 antigen found on the lymphocytes including leukemic cells, thereby attracting immune cells to them and thus destroying them.
- Boosting the immune response by targeting the immune system checkpoints
- Attaching to and blocking the antigens on cancer cells that facilitate the growth and spread of cancer cells and other neighboring cells. eg trastuzumab (Herceptin) is a mAb designed against the HER2 protein of breast and stomach cancer and blocks their activation. This HER2 protein facilitates the growth of cancer cells.
2. Conjugated (labeled or tagged or loaded) monoclonal antibodies
- Conjugated mAbs are conjugated or combined with chemotherapy drugs or a radioactive agent.
- They are mainly used as homing devices in driving the chemotherapy drug directly to the cancer cells.
- Conjugated mAbs circulate freely throughout the body until it finds and attaches (hooks) to the target antigen and delivers the antigen to immune elimination processes.
- The advantage of conjugated monoclonal antibodies is that they reduce the risks of damaging normal cells in other body parts.
a. Radiolabeled monoclonal antibodies
- They possess a small radioactive particle attached to them where the drug along with radioactive agents are delivered directly to the target cells, causing the traditions to affect the target and the neighboring cells to some extent.
- For example, ibritumomab tiuxetan (Zevalin) acts against the CD20 antigen found on the B-lymphocytes.
- They deliver the radioactivity directly to the cancer cells.
- Ibritumomab tiuxetan drugs are made up of mAb drugs known as rituximab and a radioactive agent called Yttrium-90.
- Radiolabelled monoclonal antibody treatment is known as radioimmunotherapy (RIT).
b. Chemolabeled antibodies
These mAbs have powerful chemotherapy (or other) drugs attached to them. Examples include:
- Brentuximab vedotin (Adcetris), an antibody that targets the CD30 antigen (found on lymphocytes), is attached to a chemo drug called MMAE.
- Ado-trastuzumab emtansine (Kadcyla, also called TDM-1), an antibody that targets the HER2 protein, is attached to a chemo drug called DM1.
3. Bispecific monoclonal antibodies
- These are monoclonal antibody drugs that are made up of two different monoclonal antibodies attached to each other.
- For example, a leukemic drug referred to as blinatumomab (Blincyto) has one part attached to the CD19 protein found on leukemia and lymphoma cells and another part attaches to the CD3 protein found on immune T-cells.
- This drug brings together the target cancer cell and the immune cells enabling the immune system to attack the cancer cell promptly.
Production of monoclonal antibodies
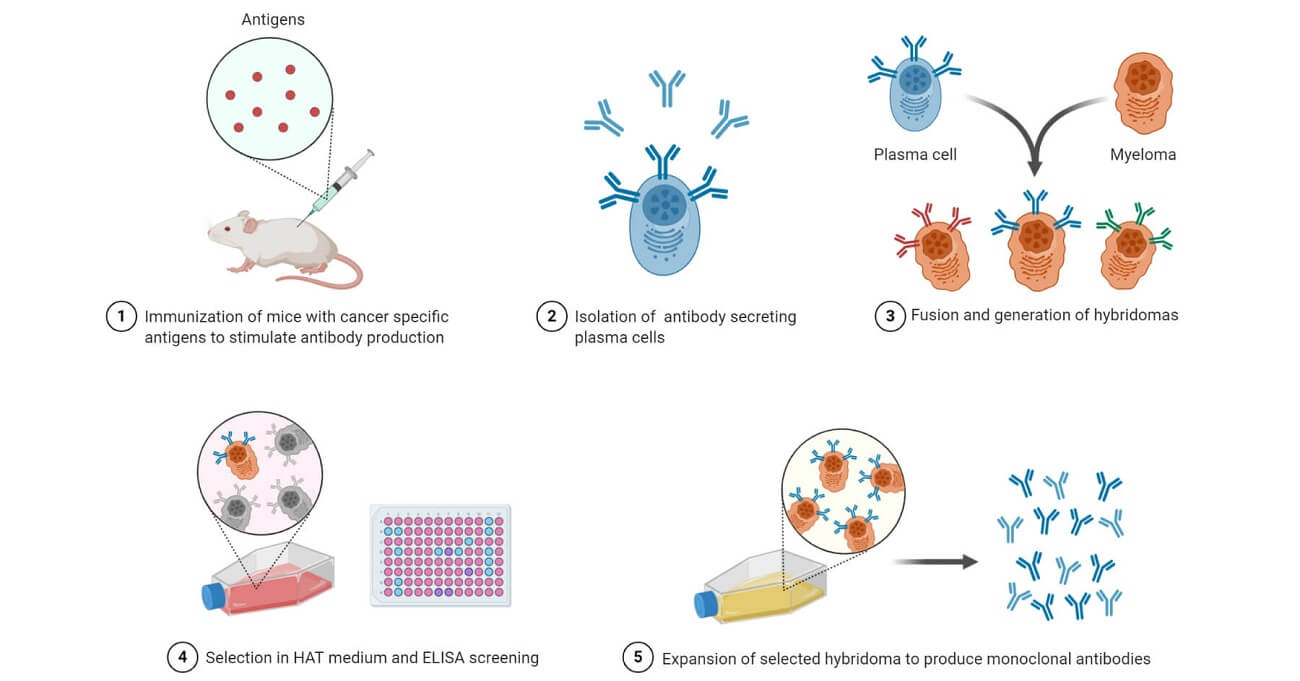
- The production of monoclonal antibodies is an in vitro process by the use of tissue-culture techniques.
- Producing monoclonal antibodies (mAbs) is initially done by identifying a specific antigen, and immunizing an animal with the antigen multiple times. The most commonly used animal models are laboratory mice.
- The B-cells of the immunized animals are removed from the spleen and then fused with cancer B-cells known as the myeloma cells.
- The fusion of adjacent plasma membranes of the myeloma cells is done using polyethylene glycol, however, it has a low success rate, and therefore the selective medium must have the fusion activity as well to enhance cell growth.
- Myeloma cancer cells have an immortal characteristic of continuously proliferating, unlike the normal B cells which proliferate for a period of 6-8 hours, and they normally have lost the ability to synthesize hypoxanthine-guanine-phosphoribosyl transferase (HGPRT), an enzyme necessary for the degradative synthesis of nucleic acids.
- The myeloma cells are placed in a selective medium known as the Hypoxanthine Aminopetrin Thymidine (HAT)-which is made up of hypoxanthine, aminopterin, and thymidine, where it allows the growth and yield of fused hybridoma cells, and the infused myeloma cells do not grow and infused B-cells die off.
- Hybridoma cells have the ability to grow continuously in culture as they produce antibodies. They are then screened for the desired or specific monoclonal antibodies, and those producing the desired mAbs are then transferred and grown in tissue culture.
- Harvesting is done periodically and the monoclonal antibodies are then purified from the medium.
- Growing and harvesting of these monoclonal antibodies are done for several weeks in large media quantities in order to produce enough mAbs that can be used for experimentation or to treat at least a single patient.
- The monoclonal antibodies produced are in millions of numbers and they are specific for the antigen that was initially injected into the animal model.
Functions and Applications of Monoclonal Antibodies
- Monoclonal antibodies are used in the treatment of several diseases and disorders and their application is known as immunotherapy.
- Some diseases and disorders treated using mAbs include: Cancers, Rheumatoid arthritis, Multiple sclerosis, Systemic Lupus erythematous, Cardiovascular diseases, Crohn’s disease, Ulcerative colitis, Psoriasis, and Rejections associated with transplantation
- Monoclonal antibodies are widely used in therapies, laboratory technique studies, and research for potential treatments for certain infections, disorders, and cancers
- Commonly, monoclonal antibodies were initially studied in cancer treatment where they are currently used in the treatment of some types of cancer.
Some of the specific applications include:
- Some monoclonal antibodies are designed to target specific tumor antigens. They have been used to stimulate the production of anti-idiotypic antibodies stimulating a strong antitumor immune response when they are inoculated in patients with B-cell lymphoma. However, anti-idiotypic antibodies are developed in animal models, which hiders production of monoclonal antibodies in humans. Though, humanized antiHer2 monoclonal antibodies Herceptin has proved effective in patients with chemotherapy-resistant breast cancer.
- Monoclonal antibodies are used to effectively bind the Tumor Necrotic Factor-alpha (TNF-alpha), which is a cytokine that helps in the progression of Rheumatoid arthritis (RA). Hence monoclonal antibodies are used as a therapeutic means for Rheumatoid Arthritis.
- Monoclonal antibodies have been generated against Tumor-specific Transplantation Antigens (TSTAs). These are antigens that result from gene mutations that cause altered proteins that are expressed by tumor cells. Practically, the patient tumor cells are tagged with monoclonal antibodies that have toxins or radioactive materials. This delivers a direct ‘magic-bullet’ therapeutic effect to the tumor and spares the healthy cells.
- They are used as an identification tool for several cancers and to also deliver drug therapies to target cancer cells and initiate immune responses against the cancer cells.
- Monoclonal antibodies are used in the diagnosis of several diseases by detecting specific antigens circulating in the body tissues and detecting them by the use of immunoassay techniques.
- Currently, monoclonal antibodies are being studied by the COVID-19 Prevention Network. for the treatment of COVID-19. Some trials have been rallied out in the US to understand the role of monoclonal antibodies in providing short-term protection against SARS-CoV-2 the causative agent of COVID-19.
Uses of Monoclonal Antibodies
Monoclonal antibodies have many practical applications in research, medical diagnosis, and therapy. Some of their common applications include the following:
- Identification of phenotypic markers unique to particular cell types. The basis for the modern classification of lymphocytes and other leukocytes is the recognition of individual cell populations by specific monoclonals These antibodies have been used to define clusters of differentiation (CD) markers for various cell types.
- The diagnosis of many infectious and systemic diseases relies on the detection of particular antigens or antibodies in the blood, urine, or tissues by the use of monoclonal antibodies in immunoassays.
- Tumor identification. Labeled monoclonal antibodies specific for various cell proteins are used to determine the tissue source of tumors by staining histological tumor sections.
- Advances in medical research have led to the identification of cells and molecules that are involved in the pathogenesis of many diseases. Monoclonal antibodies, because of their exquisite specificity, provide a means of targeting these cells and molecules. A number of monoclonal antibodies are used therapeutically. Some examples include antibodies against the cytokine tumor necrosis factor (TNF) used to treat rheumatoid arthritis and other inflammatory diseases, antibodies against CD20 for the treatment of B cell leukemia and for depleting B cells in certain autoimmune disorders, antibodies against epidermal growth factor receptors to target cancer cells, antibodies against vascular endothelial growth factor (a cytokine that promotes angiogenesis) in patients with colon cancer, etc.
- Functional analysis of cell surface and secreted molecules. In biologic research, monoclonal antibodies that bind to cell surface molecules and either stimulate or inhibit particular cellular functions are invaluable tools for defining the functions of surface molecules, including receptors for antigens. Monoclonal antibodies are also widely used to purify selected cell populations from complex mixtures to facilitate the study of the properties and functions of these cells.
Limitations of Monoclonal Antibodies
Monoclonal antibodies are most easily produced by immunizing mice, but patients treated with mouse antibodies may make antibodies against the mouse Ig, called human anti-mouse antibody (HAMA). These anti- Ig antibodies block the function or enhance clearance of the injected monoclonal antibody and can also cause a disorder called serum sickness.
Genetic engineering techniques have been used to expand the usefulness of monoclonal antibodies. The complementary DNAs (cDNAs) that encode the polypeptide chains of a monoclonal antibody can be isolated from a hybridoma and these genes can be manipulated in vitro.
Fully human monoclonal antibodies are also in clinical use. These are derived using phage display methods or in mice with B cells expressing human Ig transgenes. Humanized antibodies are far less likely than mouse monoclonals to appear foreign in humans and to induce anti-antibody responses.
Side effects of Monoclonal Antibodies
The use of its man-made monoclonal antibodies can cause side effects and these effects vary from individual to individual. these effects depend on what the patient is being treated for, the progression of the disease/disorder and the type of monoclonal being administered, and its dosage.
Reactions depend on several factors including:
- Needle site reaction associated with Pain, swelling, soreness, redness, itchiness, rash
- Flu-like symptoms associated with chills, fatigue, nausea, vomit, fever, diarrhea, muscle ache, pain
- Effects of monoclonal antibodies associated:
- Mouth and skin sores that can lead to serious infections,
- High blood pressure,
- Congestive heart failure,
- Heart attacks, Inflammatory lung disease,
- Mild to a severe allergic reaction when receiving the drug, and can even cause death.
- Capillary leak syndrome causes fluid and protein to leak out of tiny blood vessels and flowing into the surrounding tissues leading to extremely low pressure and can lead to multiple organ failures and shock.
- Cytokine release syndrome due to elevated levels in their production which is associated with monoclonal antibodies activating various mechanisms in the body. The syndrome is associated with symptoms such as fever, nausea, headache, rash, rapid heartbeat, low blood pressure, and breathing difficulties.
References
- Ali Ahmad Bayat, Omid Yeganeh, Roya Ghods, Amir Hassan Zarnani, Reza Bahjati Ardekani, Ahmad Reza Mahmoudi, Jafar Mahmoudian, Farzaneh Haghighat-Noutash, and Mahmood Jeddi-Tehrani; Production and Characterization of a Murine Monoclonal Antibody Against Human Ferritin; Avicenna Journal of Medical Biotechnology, 2013.
- Arturo Casadevall, Wendy Cleare, Marta Feldmesser, Aharona Glatman-Freedman, David L. Goldman, Thomas R. Kozel, Nikoletta Lendvai, Jean Mukherjee, Liise-Anne Pirofski, Johanna Rivera, Angel L. Rosas, Matthew D. Scharff, Philippe Valadon, Katherine Westin, Zhaojing Zhong; Characterization of a Murine Monoclonal Antibody to cryptococcus neoformans Polysaccharide That Is a Candidate for Human Therapeutic Studies; antimicrobial agents and Chemotherapy Journal, 1998.
- Pamela Holzlöhner, Katja Hanack, Generation of Murine Monoclonal Antibodies by Hybridoma Technology; National Library of Medicine, 2017.
- Wayne M Yokoyama, Michelle Christensen, Gary Dos Santos, Diane Miller, Jason Ho, Tao Wu, Michael Dziegelewski, Francisca A Neethling; Production of monoclonal antibodies; National Library of Medicine, 2013.
- http://crdd.osdd.net/raghava/absource/humanized.html
- http://www.receptabio.com.br/en/pesquisa-desenvolvimento/anticorpos-monoclonais-recepta-detem-potencial-para-tratar-diversos-tipos-de-cancer/
- https://absoluteantibody.com/our-technology/formats-we-have-made/chimeric-antibodies/
- https://en.wikipedia.org/wiki/Monoclonal_antibody
- https://link.springer.com/referenceworkentry/10.1007%2F3-540-27806-0_699
- https://link.springer.com/referenceworkentry/10.1007%2F3-540-29662 X_1874.
- https://www.cancer.gov/about-cancer/treatment/types/immunotherapy/monoclonal-antibodies
- https://www.cancer.org/treatment/treatments-and-side-effects/treatment-types/immunotherapy/monoclonal-antibodies.html
- https://www.genscript.com/how-to-make-monoclonal-antibodies.html
- https://www.intechopen.com/books/fermentation-processes/production-processes-for-monoclonal-antibodies
- https://www.mayoclinic.org/diseases-conditions/cancer/in-depth/monoclonal-antibody/art-20047808
- https://www.medicinenet.com/monoclonal_antibodies/article.htm
- https://www.medicinenet.com/script/main/art.asp?articlekey=3823
- https://www.moleculardevices.com/applications/monoclonal-antibody-production#gref
- https://www.nature.com/articles/d41586-019-02840-w
- https://www.ncbi.nlm.nih.gov/books/NBK100203/: Monoclonal Antibody Production
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5022998/
- https://www.nih.gov/news-events/news-releases/clinical-trials-monoclonal-antibodies-prevent-covid-19-now-enrolling
- https://www.nuventra.com/resources/blog/monoclonal-antibodies-past-present-and-future/
- https://www.prospecbio.com/monoclonal_antibodies
- https://www.proteogenix.science/scientific-corner/antibody-production/murine-monoclonal-antibodies-and-their-use-in-modern-medicine
- https://www.sinobiological.com/resource/antibody-technical/chimeric-monoclonal-antibody

superb
very clear and very useful for brushing up the details learnt long ago Thank you Faith Mokobi.