Lac operon definition
- The lactose or lac operon of Escherichia coli is a cluster of three structural genes encoding proteins involved in lactose metabolism and the sites on the DNA involved in the regulation of the operon.
- Many protein-coding genes in bacteria are clustered together in operons which serve as transcriptional units that are coordinately regulated.
- It was Jacob and Monod in 1961 who proposed the operon model for the regulation of transcription.
The operon model proposes three elements:
- A set of structural genes (i.e. genes encoding the proteins to be regulated);
- An operator site, which is a DNA sequence that regulates transcription of the structural genes;
- A regulator gene which encodes a protein that recognizes the operator sequence.
Lac operon
- One of the most studied operons is the lac operon in E. coli.
- This codes for key enzymes involved in lactose metabolism:
- Galactoside permease (also known as lactose permease).
Transports lactose into the cell across the cell membrane.
- Galactosidase
Hydrolyzes lactose to glucose and galactose.
- Thiogalactoside transacetylase
Structure of Lac operon
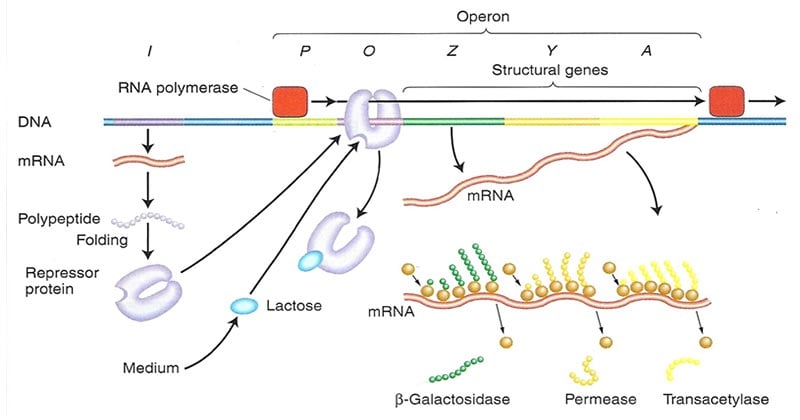
- In the lac operon, the structural genes are the lacZ, lacY and lacA genes encoding _-galactosidase, the permease, and the transacetylase, respectively.
- Transcription occurs from a single promoter (Plac) that lies upstream of these structural genes and binds RNA polymerase.
- However, also present are an operator site (Olac) between the promoter and the structural genes, and a lacI gene that codes for the lac repressor protein.
- The lacI gene has its own promoter (PlacI) that binds RNA polymerase and leads to transcription of lac repressor mRNA and hence the production of lac repressor protein monomers.
- Four identical repressor monomers come together to form the active tetramer which can bind tightly to the lac operator site, Olac.
Inducers and the Induction of Lac operon
- Normally, E. coli cells make very little of any of these three proteins but when lactose is available it, causes a large and coordinated increase in the amount of each enzyme.
- Thus each enzyme is an inducible enzyme and the process is called induction.
- The mechanism is that the few molecules of ß-galactosidase in the cell before induction convert the lactose to allolactose which then turns on the transcription of these three genes in the lac operon.
- Thus allolactose is an inducer.
- Another inducer of the lac operon is isopropylthiogalactoside (IPTG). Unlike allolactose, this inducer is not metabolized by E. coli and so, is useful for experimental studies of induction only.
Lac Operon in absence of Inducers
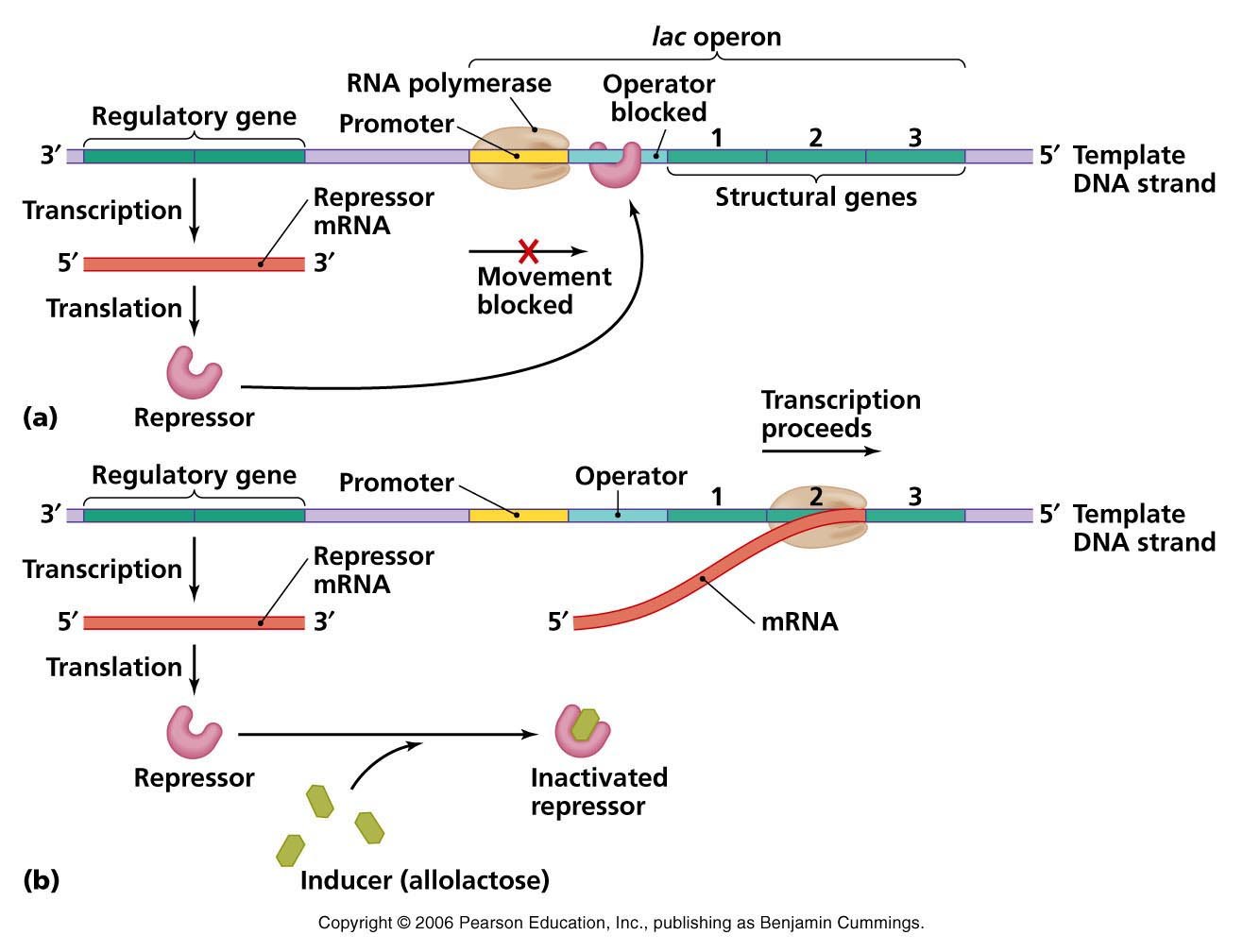
- In the absence of an inducer such as allolactose or IPTG, the lacI gene is transcribed and the resulting repressor protein binds to the operator site of the lac operon, Olac, and prevents transcription of the lacZ, lacY and lacA genes.
Lac Operon in presence of Inducers
- During induction, the inducer binds to the repressor.
- This causes a change in the conformation of the repressor that greatly reduces its affinity for the lac operator site.
- The lac repressor now dissociates from the operator site and allows the RNA polymerase (already in place on the adjacent promoter site) to begin transcribing the lacZ, lacY and lacA genes.
- They are transcribed to yield a single polycistronic mRNA that is then translated to produce all three enzymes in large amounts.
- The existence of a polycistronic mRNA ensures that the amounts of all three gene products are regulated coordinately.
- If the inducer is removed, the lac repressor rapidly binds to the lac operator site and transcription is inhibited almost immediately.
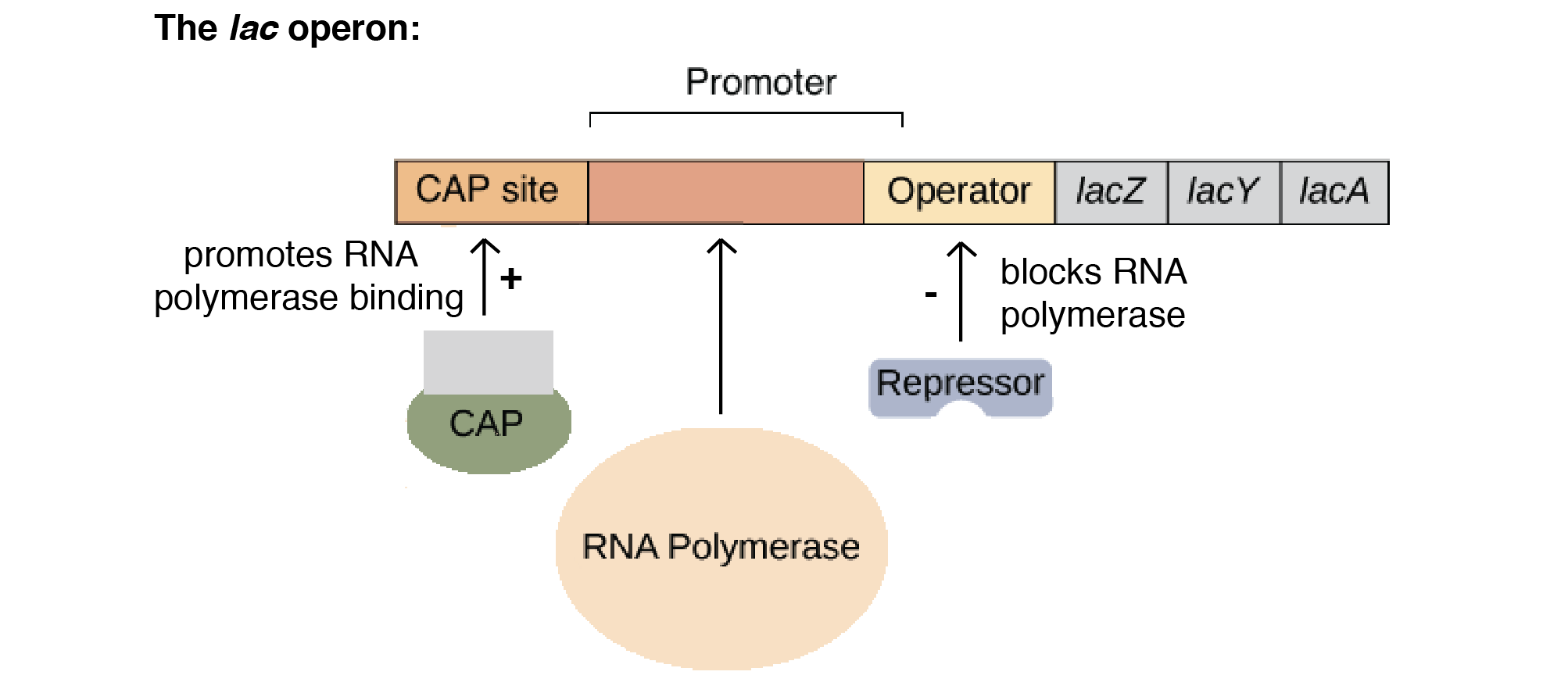
CRP/CAP
- High-level transcription of the lac operon requires the presence of a specific activator protein called catabolite activator protein (CAP), also called cAMP receptor protein (CRP).
- This protein, which is a dimer, cannot bind to DNA unless it is complexed with 3’5′ cyclic AMP (cAMP).
- The CRP–cAMP complex binds to the lac promoter just upstream from the binding site for RNA polymerase.
- It increases the binding of RNA polymerase and so stimulates transcription of the lac operon.
- Whether or not the CRP protein is able to bind to the lac promoter depends on the carbon source available to the bacterium.
Lac operon in the presence of Glucose
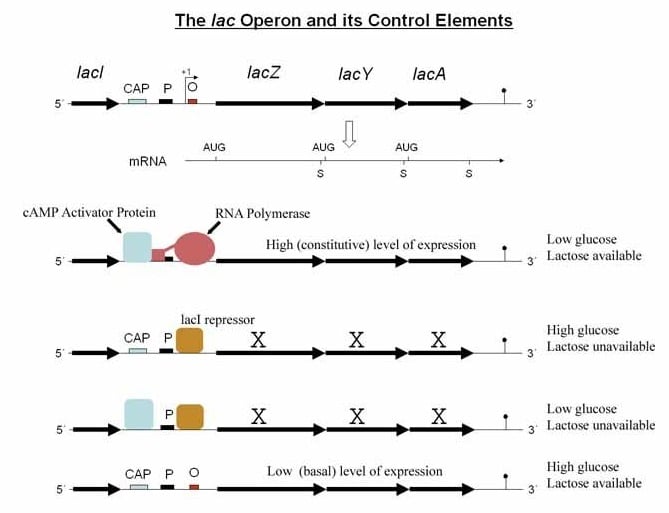
- When glucose is present, E. coli does not need to use lactose as a carbon source and so the lac operon does not need to be active.
- Thus the system has evolved to be responsive to glucose.
- Glucose inhibits adenylate cyclase, the enzyme that synthesizes cAMP from ATP.
- Thus, in the presence of glucose the intracellular level of cAMP falls, so CRP cannot bind to the lac promoter, and the lac operon is only weakly active (even in the presence of lactose).
Lac operon in the absence of Glucose
- When glucose is absent, adenylate cyclase is not inhibited, the level of intracellular cAMP rises and binds to CRP.
- Therefore, when glucose is absent but lactose is present, the CRP–cAMP complex stimulates transcription of the lac operon and allows the lactose to be used as an alternative carbon source.
- In the absence of lactose, the lac repressor, of course, ensures that the lac operon remains inactive.
- These combined controls ensure that the lacZ, lacY and lacA genes are transcribed strongly only if glucose is absent and lactose is present.
Positive and Negative Regulation of Lac Operon
- The lac operon is a good example of the negative control (negative regulation) of gene expression in that bound repressor prevents transcription of the structural genes.
- Positive control or regulation of gene expression is when the regulatory protein binds to DNA and increases the rate of transcription.
- In this case, the regulatory protein is called an activator. The CAP/CRP involved in regulating the lac operon is a good example of an activator.
- Thus the lac operon is subject to both negative and positive control.
References
- David Hames and Nigel Hooper (2005). Biochemistry. Third ed. Taylor & Francis Group: New York.
- Sastry A.S. & Bhat S.K. (2016). Essentials of Medical Microbiology. New Delhi: Jaypee Brothers Medical Publishers.
- Parija S.C. (2012). Textbook of Microbiology & Immunology.(2 ed.). India: Elsevier India.

Thumbs upp its a good site for students
Totally agree it’s a very helpful site for students.
Good explanatory principle.