Laboratory diagnosis of Leprosy caused by Mycobacterium leprae
Specimen
Skin biopsies, nasal discharges, scrapings from the nasal mucosa and slit-skin smears which are prepared by making superficial incisions in the skin, scraping out some tissue fluid and cells.
Skin smears
- The skin smears are collected from the leprous lesions, such as nodules, thick papules, and areas of infiltration.
- In cases of patches, the samples are obtained from the edge of the lesion rather than from the center.
- The skin smears from these sites are collected by slit and scrap method.
Nasal smears
- Smears from the nasal patches are collected by scraping material from the mucous membrane of the internal nasal septum, particularly from inferior turbinate folds of the nasal septum.
Skin and nerve biopsy
- Skin biopsy is collected from active edge of the patches and nerve biopsy from thickened nerve for histological confirmation of tuberculoid leprosy.
Microscopy
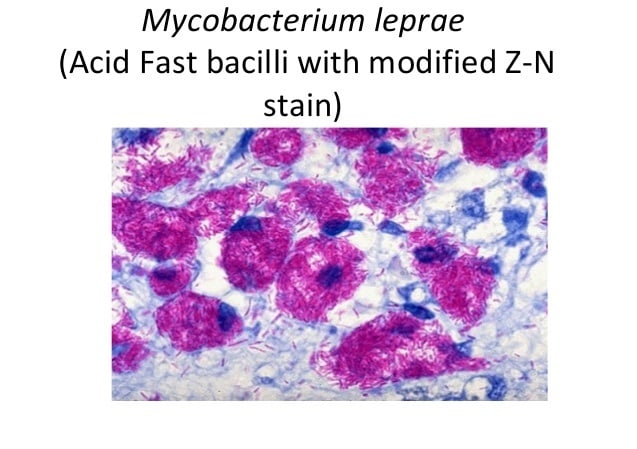
- The slit-skin smears, nasal smears, or smears from other specimen on the slide are stained by Ziehl- Neelsen technique by using 5% sulfuric acid for decolorizarion.
- Under oil immersion objective, red acid fast bacilli are seen, arranged singly or in groups (cigar like bundles), bound together by lipid-like substance, the glia to form
- The globi are present inside the foamy macrophages called Virchow’s lepra cells or foamy cells.
Bacillary index:
- The bacillary index or BI is an expression of the extent of bacterial load.
- It is calculated by counting six to eight stained smears under the oil immersion lens.
- This index is obtained by totaling the number of pluses.
- The bacillary index is usually affected by the thickness of the film, depth of the skin incision, and thoroughness of the scrap.
Morphological index:
- The uniformity of the bacilli is used as a criterion to differentiate live bacilli from dead bacilli in stained smears.
- The lepra bacilli that stain uniformly with carbol fuchsin as solid acid-fast rods are believed to be viable, and the bacilli that stain irregularly are considered dead and degenerated.
- The percentage of uniformly stained bacilli in the tissues is known as morphological index (MI).
Lepromin test
- The lepromin test is used to study host immunity to M. leprae.
- The test is an intradermal skin test performed by using lepromin antigen, which is a suspension of killed M. leprae obtained from infected human or armadillo tissue.
- The lepromin test is not used to confirm the diagnosis of leprosy.
- It is also not useful to indicate prior contact of the person with leprae bacilli.
- Leprosins, which are ultrasonicates of tissue-free bacilli extracted from lesions (the suffixes -H and -A are used to denote human and armadillo origins, respectively).
- These reagents elicit two types of reaction:
- The Fernandez reaction is analogous to tuberculin reactivity and appears in sensitized subjects 48 hours after skin testing. Positive reaction is characterized by the appearance of a localized area of inflammation with congestion and edema measuring 10 mm and more in diameter during 24–48 hours of injection. These lesions disappear within 3–4 days. Positive reaction suggests that the patient has been infected by leprae bacilli during sometime in the past.
- The Mitsuda reaction is characterized by development of a nodule at the site of inoculation after 3–4 weeks after testing with lepromin. The nodule subsequently may undergo necrosis followed by ulceration. This reaction is indicative of the host’s ability to give a granulomatous response to antigens of M. leprae, and is positive at or near the TT pole.
Mouse Foot Pad Cultivation
- M. leprae is not cultivable either in artificial culture media or in tissue culture.
- The only certain way to cultivate M. leprae is by inoculating the specimens into fool pad of mice and keeping at 20°C for 6-9 months.
- Other animals such as nine banded armadillo can also be used which is a natural host and reservoir of the pathogen.
Serodiagnosis
- Serodiagnosis of leprosy is based on demonstration of antibodies to M. leprae, specific PGL-1 antigens.
- Enzyme linked immunosorbent assay (ELISA) and latex agglutination test are used to detect serum antibodies.
- The serology is useful primarily in patients with untreated lepromatous leprosy, as most of patients have higher levels of serum antibodies.
- The serology, however, is less useful for diagnosis of paucibacillary disease, because serum antibodies are present in only 40–60% of such patients.
- FLA-ABS (Fluorescent leprosy antibody absorption test) is widely used to identify subclinical cases.
- It detects M. leprae specific antibodies irrespective of duration and stage of the disease.
Molecular Diagnosis
- Polymerase chain reaction (PCR) for identifying DNA that encodes 65 kDa and 18 kDa M. leprae proteins and repetitive sequences of M. leprae is being used to detect and identify M. leprae in clinical specimens.
- PCR is used to monitor treatment, diagnose relapses, or determine the need for chemotherapy.
- The technique is most useful in cases of leprosy showing atypical clinical or histopathological features but positive for acid-fast bacilli.
- It is not useful for diagnosis of cases when acid-fast bacilli are not detected by light microscopy.
Treatment of Leprosy
- The management of leprosy depends on treatment by chemotherapeutic agents, immunotherapy, and physical, social, and psychological rehabilitation.
- Tuberculoid form is treated with rifampicin and dapsone for 6 months; clofazimine is added to this regimen for treatment of the lepromatous form, and therapy is extended to a minimum of 12 month
- Chemotherapy: The goals of chemotherapy against leprosy are to
- Stop infection
- Reduce morbidity
- Prevent complications
- Eradicate the disease
- Dapsone was the first effective chemotherapeutic agent used against leprosy.
- Earlier it was used as a monotherapy, but it resulted in development of resistance against the drug by lepra bacilli.
- Hence since 1981, the World Health Organization (WHO) has advocated multiple drug therapy (MDT) against leprosy.
Multiple drug therapy (MDT) against leprosy
- The MDT quickly decreases contagiousness of the disease, reduces relapse and reactions, and reduces disabilities.
- Moreover, MDT prevents dapsone resistance.
- The duration of treatment varies from 6 months to 2 years.
- The recommendations of WHO for treatment of leprosy in adults are as follows:
- Single skin lesion: A single dose of the drug which includes rifampicin 600 mg, ofloxacin 400 mg, and minocycline 100 mg.
- Paucibacillary disease: Dapsone 100 mg daily and rifampicin 600 mg once a month given for 6 months.
- Multibacillary disease: Rifampin (rifampicin) 600 mg once a month, dapsone 100 mg daily, clofazimine 300 mg once a month, and 50 mg daily are given for 1 year.
- A minimum 2 years follow-up for paucibacillary and 8 years for multibacillary cases is required to detect any relapse.
- Treatment schedule for children consists of dapsone (2 mg/kg) daily, clofazimine (6 mg/kg) once a month under supervision and 1 mg/kg daily self-administered, and rifampin (10 mg/kg) once a month.
- Reactions during treatment with MDT are a major problem.
- These reactions need urgent treatment, otherwise they can give rise to irreversible deformities.
- Early diagnosis and timely initiation of anti-inflammatory measures are important.
- MDT is not stopped during reactions and continued at full doses without interruption.
Immunotherapy
- Many immunotherapy agents have been evaluated for treatment of leprosy.
- These include immunomodulatory drugs, transfer factor, acetoacetylated leprae, and delipidified cell components of M. leprae.
- These agents have shown to enhance CMI, which results in increased killing and rapid clearing of dead lepra bacilli.
- Immunotherapy in combination with chemotherapy has shown to be more beneficial, better tolerated by patients, and is not associated with increased lepra reactions.
Prevention and Control of Leprosy
- The preventive and control measures include
- Early diagnosis and treatment of leprosy and surveillance of contacts
- Health education
- Vaccines
- Chemoprophylaxis
- The fact that vaccination with BCG has shown some protective effect in leprosy has encouraged further interest in vaccine development.
- Thalidomide, an inhibitor of tumor necrosis factor is being distributed under tight restrictions for use as a treatment for erythema nodosum leprosum, a serious and severe skin complication of leprosy.