- Actinomycetes are classified as a group of gram-positive bacteria that are unique for their spore forming abilities and formation of mycelia structures.
- They show marked chemical and morphological diversity but form a distinct evolutionary line of organisms that range from coccoid and pleomorphic forms to branched filaments.
- These bacteria have been noted to serve as rich reservoirs of medicinal antibiotics and are therefore extremely relevant to scientists, pharmaceutical industries and agricultural industries.
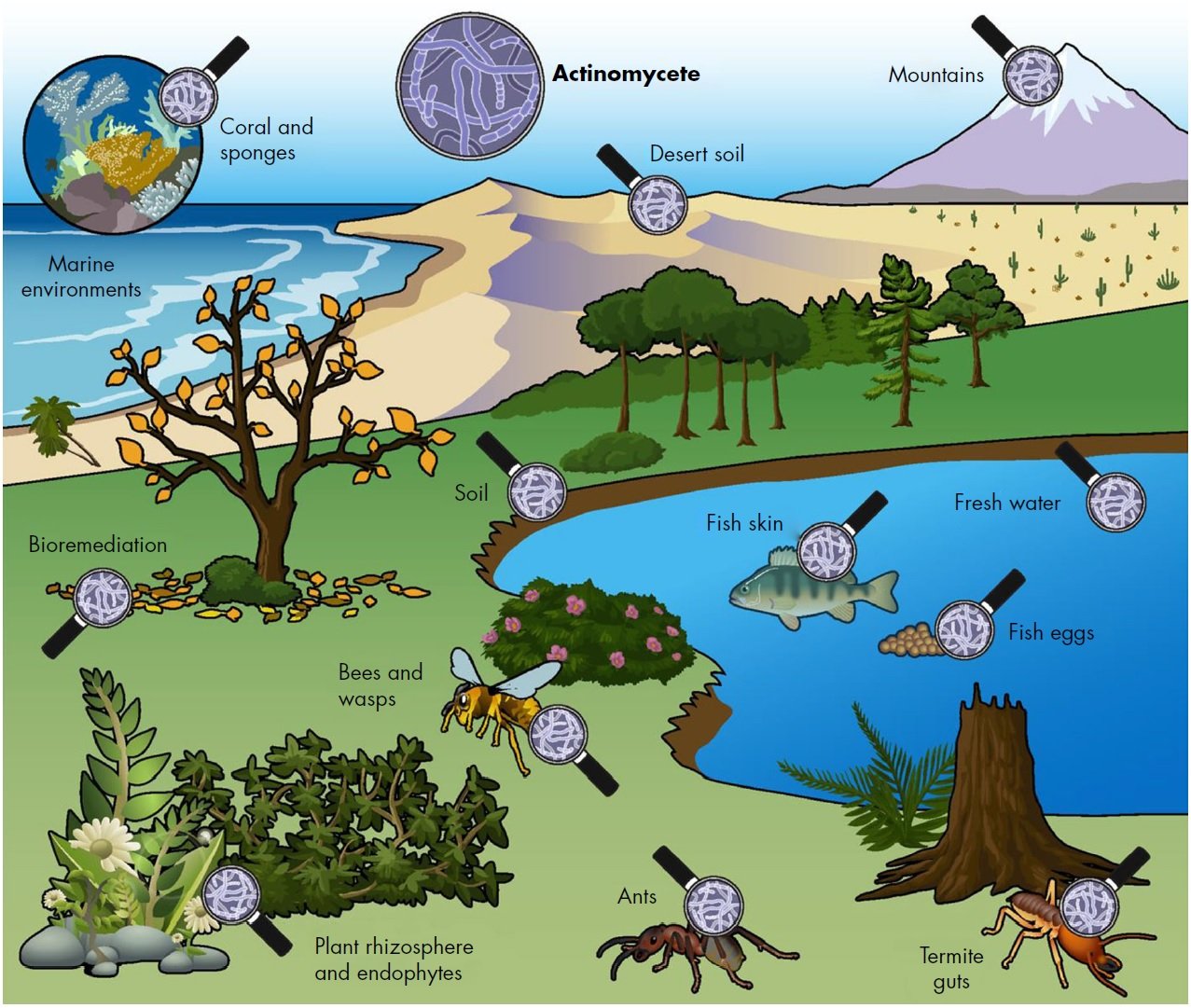
Figure: Free living actinomycetes are ubiquitous in soil environments as well as in marine and fresh water ecosystems. In addition, they have an important ecological role in the turnover of organic material. Many actinomycetes have evolved to live in symbiosis with plants, fungi, insects and animals. Most such actinomycete–host interactions are beneficial, whereby actinomycetes produce NPs that allow their host to protect itself against pathogens or pests, or enzymes to degrade resilient natural polymers like lignocellulose.
Image Source: Gilles P. van Wezel, Molecular Biotechnology, Institute of Biology, Leiden University, Sylviusweg 72, 2333 BE Leiden, The Netherlands.
Principle of Isolation of Actinomycetes
Actinomycetes is a non-taxonomic term for a group of common soil microorganisms sometimes called “thread or ray bacteria.” They are known for decomposing more resistant organic materials such as chitin, a complex sugar found in the outer skeleton of insects and elsewhere. The richness and diversity of actinomycetes present in any specific soil, however, is greatly influenced by the soil type, geographical location, cultivation and organic matter amongst other factors.
Different agar media can be used for the isolation of Actinomycetes from soil sample after dilution of the soil slurry. Actinomycetes Isolation Agar is a selective medium commonly employed. It contains sodium caseinate as nitrogen source. Asparagine in addition to being an amino acid is also a source of nitrogen. Sodium propionate is used as a substrate in anaerobic fermentation. Dipotassium phosphate provides the buffering system. The sulphates serve as source of sulphur and metallic ions. Glycerol serves as an additional source of carbon. Cycloheximide and nystatin used in the agar, further prevents fungi interference by inhibiting mRNA translation, hence causing cell growth arrest and cell death of opportunistic fungi.
The chitin agar, on the other hand, contains chitin, a complex sugar which is generally hydrolysed by Actinomycetes. Thus it is able to support the growth of only the actinomycetes and also in addition prevent the growth of other soil bacteria and fungi.
Procedure of Isolation of Actinomycetes
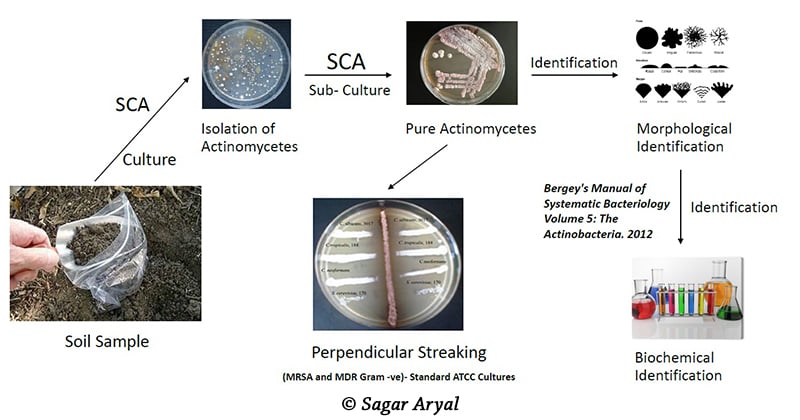
- First, soil slurry is made by suspending 1 gm of the collected dry soil in 10 ml distilled water. The slurry is mixed by vortexing for 2 minutes.
- Four 1 in 10 fold serial dilutions is made from the slurry in duplicates.
- 1 ml portions of each dilution, are then plated by spreading on the set agar plates (Actinomycete Isolation Agar supplemented with cyclohexamide (50 μg/ml) and nystatin (50 μg/ml) and Chitin agar/ Starch Casein Agar).
- All spread plates are labelled and incubated at 25 ̊C for a period of 7 to 14 days.
- On day 14, the number of colonies formed in each dilution of both groups of, are counted and recorded.
- All plates are carefully observed under the microscope to detect diversity in colonies formed. The richness, evenness and diversity index, are also calculated and recorded.
- Different colony types are then picked out with sterile forceps and streaked out on glucose yeast extract agar plates, to obtain pure single colonies.
- The streaked plates are then incubated and subsequently tested for antibiotic production.
Expected Results of Isolation of Actinomycetes
- On Actinomycete Isolation Agar supplemented with cyclohexamide (50 μg/ml) and nystatin (50 μg/ml), after 7 days incubation, whitish pin-point colonies, characteristic of actinomycetes, with a clear zone of inhibition around them can be observed.
- Colonies appearing as large bright white filaments with net-like mycelia, pale white branching filaments with powdery appearance, dark brown uniformity and crumb-like appearance, light brown appearance with cilia-like mycelia on its boundaries, a dark brown appearance with embedded with concentric circular patterns, yellow colonies with beautiful mycelia and transparent boundaries can be expected.
- Some of the actinomycete colonies on chitin agar are surrounded by a clear zone of hydrolysis which facilitates macroscopic recognition.
References
- Collee J. G., Fraser A. G., Marmion B. P., Simmons A., (Eds.), Mackie and McCartney, Practical Medical Microbiology, 1996, 14th Edition, Churchill Livingstone.
- http://article.sapub.org/pdf/10.5923.j.microbiology.20140403.02.pdf
- https://aem.asm.org/content/aem/29/3/422.full.pdf
- https://pdfs.semanticscholar.org/5611/41ee7326d711cfd8851971e1dd5a5fda6af5.pdf
- https://www.researchgate.net/post/what_is_the_best_procedure_to_isolate_the_actinomcetes_from_soil
- Eaton A. D., Clesceri L. S. and Greenberg A. W., (Eds.), 2005, Standard Methods for the Examination of Water and Wastewater, 21st Ed., APHA, Washington, D.C.

sir can we use antibacterial antibiotics for the isolation with AIA and also elaborate the extraction of the secondary metabolites
Sir how can study in this project working easily such as production of antibiotics and others production……