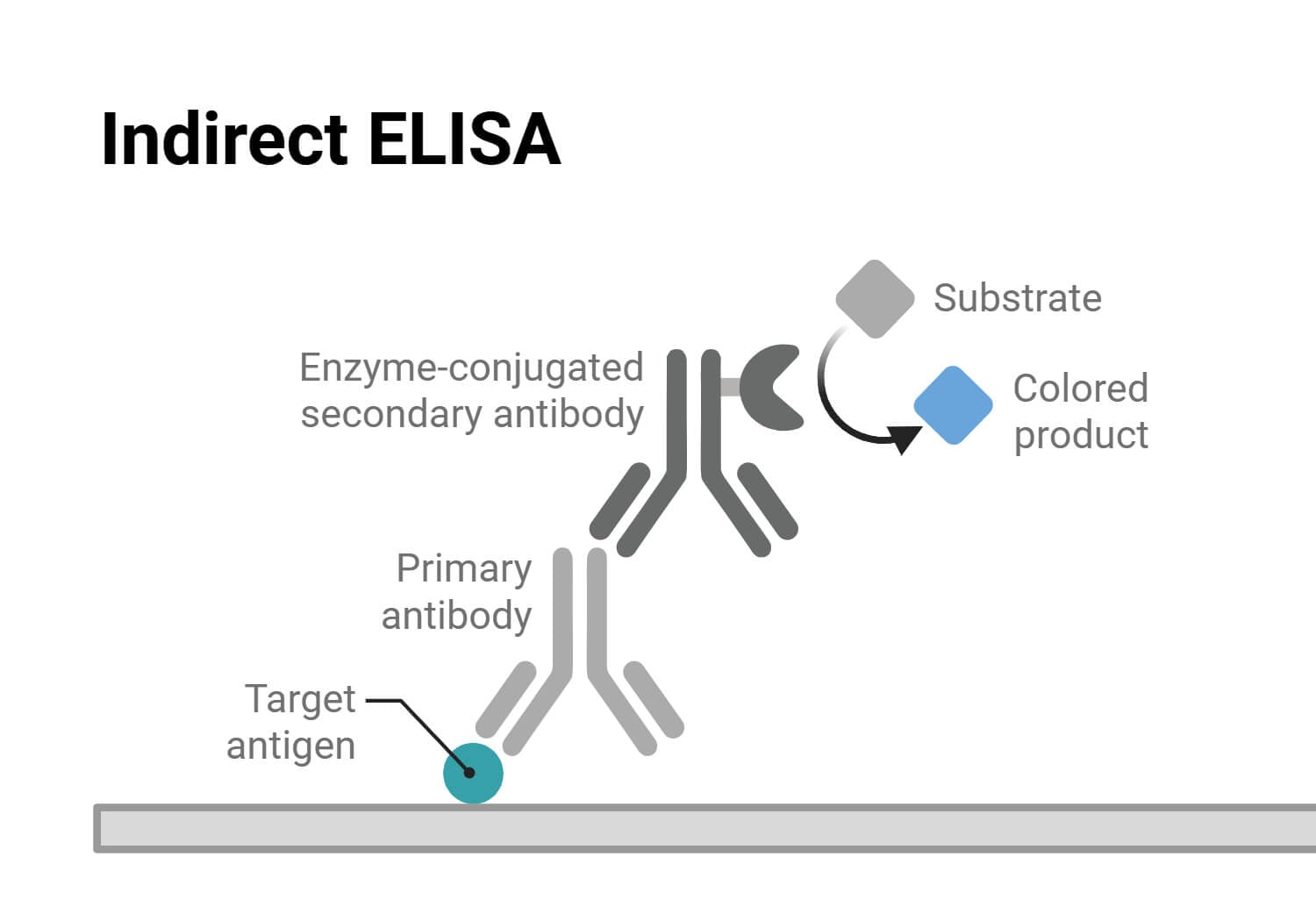
Indirect ELISA (Enzyme-Linked Immunosorbent Assay) is a type of ELISA that is widely used for detecting and quantifying antibodies in samples such as serum and other biological fluids.
It is a two-step process, where a secondary antibody linked with an enzyme is used to detect the primary antibody produced against a specific antigen in the human body.
Principle of Indirect ELISA
Indirect ELISA relies on two antibodies: primary and secondary antibodies. In this assay, a specific antigen is coated on a 96-well plate, and the primary antibody produced in the human body against that antigen binds to it. This binding is detected using a secondary antibody linked to an enzyme, followed by the addition of a colorimetric substrate. The enzyme-substrate reaction produces a colored product, which is then measured with a spectrophotometric plate reader. The intensity or signal of the colored compound produced is directly proportional to the amount of antibody present in the samples.
Materials and Reagents Required
Materials:
- 96-well plate
- Micropipettes and tips
- Incubator
- Refrigerator
- Microplate reader
- Centrifuge tubes
Reagents:
- Target antigen
- Coating Buffer
- Washing Buffer
- Blocking Buffer
- Samples
- Secondary Antibody
- Substrate solution
- Reaction Terminator solution
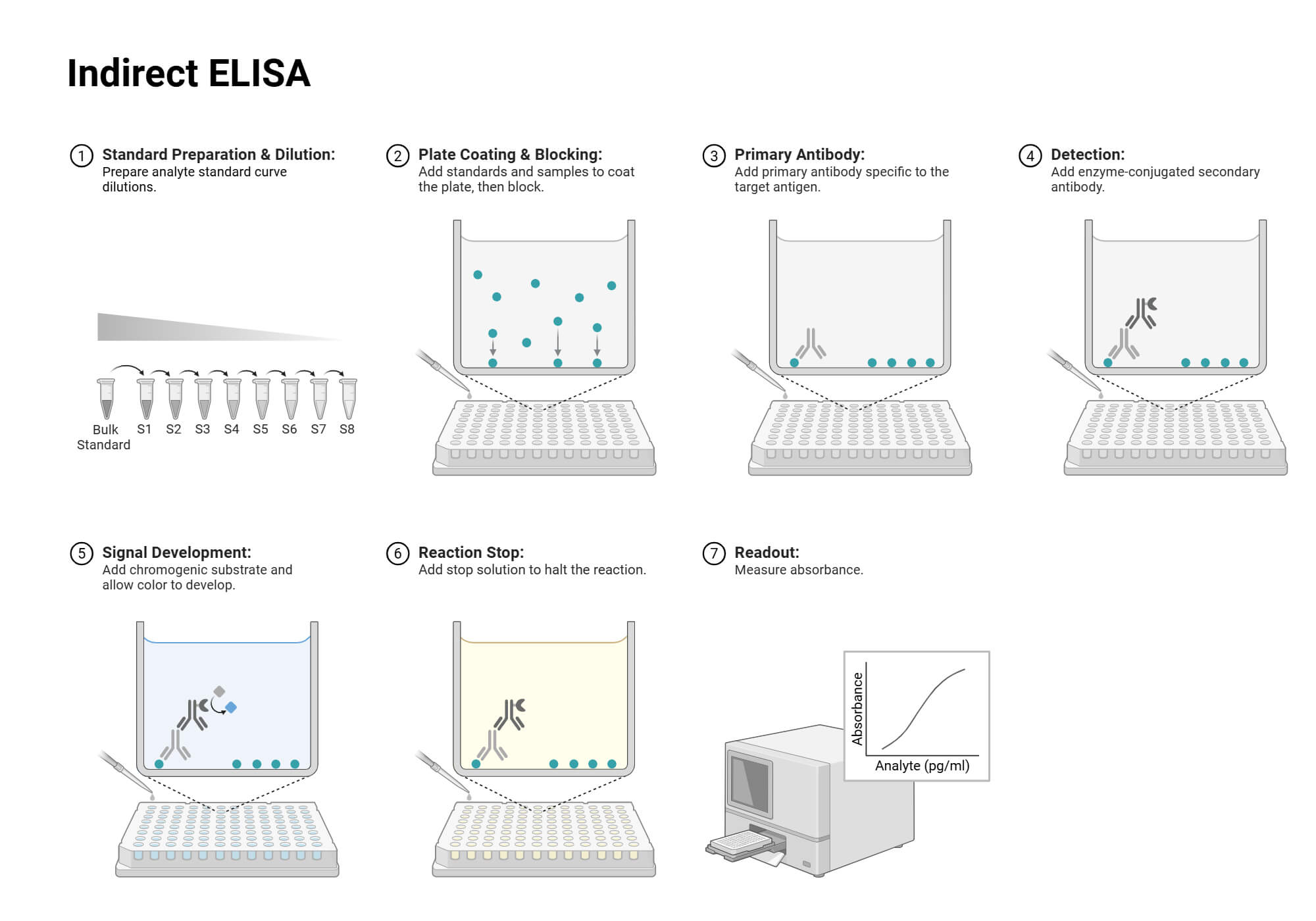
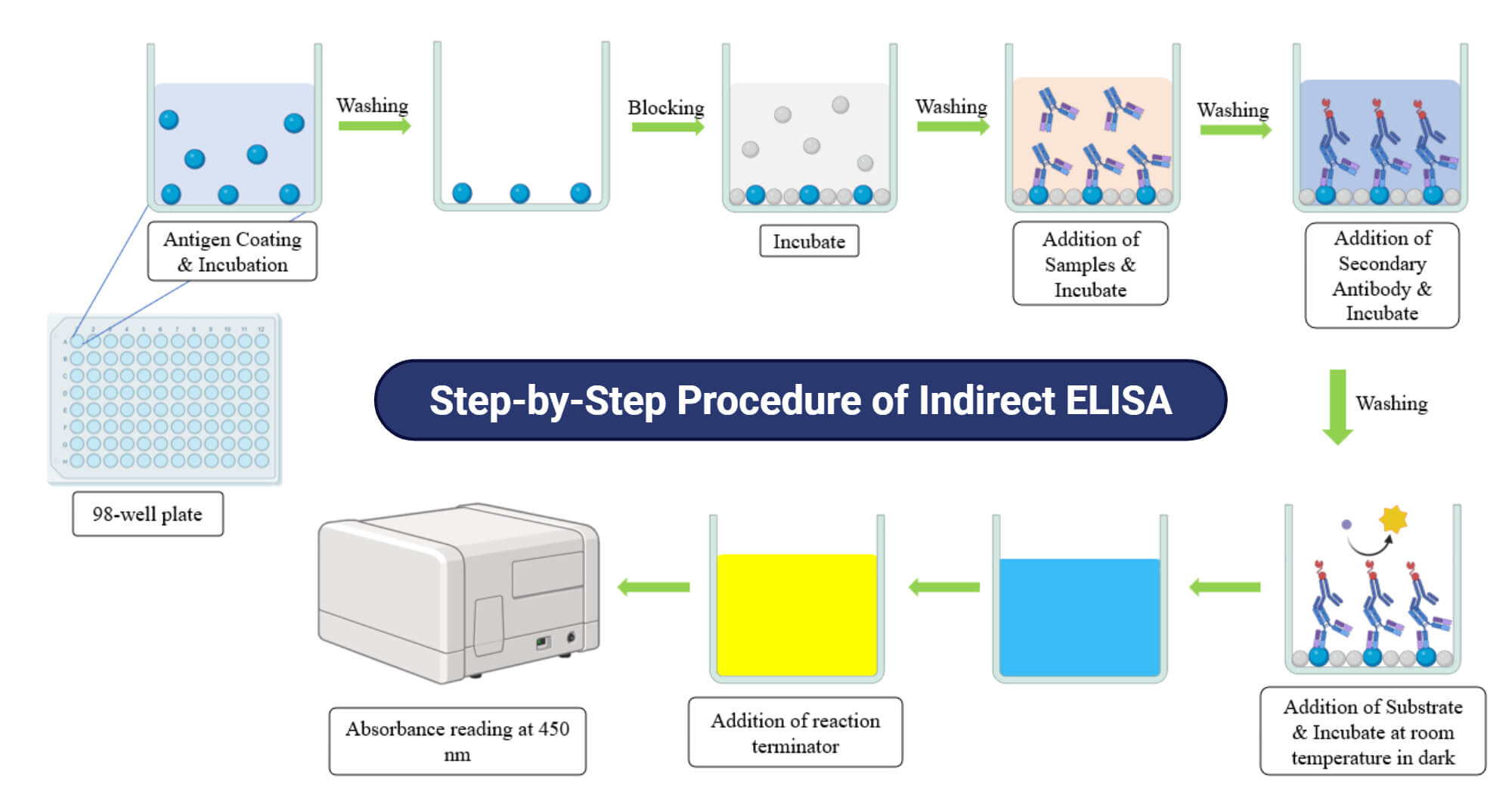
Step-by-Step Procedure of Indirect ELISA
- Preparation of antigen: Firstly, the known target antigen is diluted in coating buffer (e.g., PBS (Phosphate Buffer Saline)).
The optimum concentration needs to be found out by the titration technique with a known antibody.
- Coating of antigen: 100 µl of antigen is added to the wells and incubated at 4 °C overnight or at 37 °C for 1-2 hrs. for adsorption of antigen in plate wells.
- Washing of wells: After incubation, the washing buffer (PBS with 0.01- 0.05 % Tween-20 (PBST)) is used to remove the unbound antigen in the wells. This step is repeated 2-3 times.
- Blocking of wells: 50-100 µl of blocking buffer (BSA or PBS with 1% skim milk (PBSM)) is added to the wells to block the remaining unbound sites of the well surface. It is incubated for 1-2 hrs. at 37 °C and washed with PBST for 3 times.
Samples to be used need to be diluted to avoid signal overloading before the sample addition step.
- Addition of Sample: 100 µl of samples diluted in dilution buffer (PBSM) are added to the wells and incubated for 1-2 hrs. at 37 °C. Then, again, the wells are washed to remove unbound samples.
Note: The industrially prepared antibody against a specific antigen is used as a positive control, and healthy samples can be used as a negative control. And PBS can be used for blank.
- Addition of Secondary antibody: 100 µl of diluted secondary antibody conjugated with enzyme is added to the wells and incubated for 1-2 hrs. at 37 °C.
The secondary antibody conjugated with an enzyme to be used needs to be diluted in the prescribed dilution ratio, as mentioned in the manual.
- Addition of Substrate: Substrate solution (e.g., TMB) is added to the wells and incubated for 10- 30 minutes at room temperature in dark conditions.
- Reaction terminator and reading the results: Finally, the reaction terminator solution (e.g., 1M HCl) is added to stop enzyme substrate reaction. Within 1-2 minutes of addition, an absorbance reading is taken at 450 nm wavelength.
The obtained absorbance is proportional to the amount of antibody present in the samples. The amount of antibody can be detected by plotting a standard curve of known primary antibody.
The Role of Primary and Secondary Antibodies in Indirect ELISA
In indirect ELISA, the primary antibody is an antibody produced in the host body against a specific antigen. Secondary antibody is the one that recognizes the primary antibody.
For positive control, a monoclonal antibody produced in the industry by immunizing the model organism with a specific antigen is used as the primary antibody, while for samples, the antibody is only produced if the individual is infected with any specific infection.
The secondary antibody is produced by immunizing the primary antibody in another host model, like if the primary antibody is produced in a mouse, the secondary antibody is produced by immunizing other host models, such as a goat or rabbit. It plays a major role in indirect ELISA as it binds only with the primary antibody, but not with the target antigen. So, the secondary antibody will only bind if the individual is infected, i.e., if the sample contains primary antibody. This enzyme-linked secondary antibody then reacts with the substrate and produce colored product. The colored product represents the presence of the primary antibody in the sample, and the quantity of antibody can be determined by taking an absorbance reading.
Substrate Addition and Signal Detection in Indirect ELISA
The substrate solution is a chemical that reacts with the enzyme to produce a product. In ELISA, the substrate solution differs according to the signals it produces and the detection process. There are mainly three types of substrates: colorimetric, chemiluminescent, and fluorogenic substrates. They all have different reader instruments. But usually, colorimetric substrates and a spectrophotometric device are used in ELISA, due to the affordability and easy visualization of the colored product they produce. There are various colorimetric substrate solutions for ELISA. Some of the common substrates are TMB (3,3′,5,5′-tetramethylbenzidine) and pNPP (p-Nitrophenyl Phosphate, Disodium Salt). The use of substrate mainly depends on what enzyme-linked secondary antibody is being used in the assay. Also, the signals they produce are recorded at different wavelengths.
For example, TMB is used for secondary antibody conjugated with HRP (Horseradish Peroxidase) while pNPP is used for antibody conjugated with AP (Alkaline Phosphatase), and signal detection is measured at 450 nm and 405 nm, respectively. This is because the end product compound in each case, exhibits maximum absorbance peak in those wavelengths.
Data Analysis and Result Interpretation of Indirect ELISA
In indirect ELISA, result interpretation can be done simply by visualizing the production of the color product at the end of the assay. For example, if TMB substrate is used, it first converts colorless substrate to a blue color compound, which then is terminated by acid (e.g., HCl), and a yellow-colored product is formed. The higher the intensity of the colored product, the higher the analyte concentration. Therefore, the formation of colored products distinguishes analytes containing samples from other samples.
But sometimes, due to a technical error, such as a handling error, damaged reagents during the assay, the colored product might not be produced even if the sample contains the analytes. Hence, further testing of reagents and precise pipetting needs to be done.
In the ELISA assay, test samples along with positive and negative controls are used to validate the test, and a blank is used to reduce background absorbance. Also, for high accuracy and minimum error, the samples are run in replicates (duplicates or triplicates). Then average readings of the samples, controls, and blank need to be taken for final data analysis. Usually, the final mean of samples is calculated by deducting the negative control mean and 3 times its standard deviation from the positive control mean. These data analyses can be done with software such as GraphPad Prism, MS Excel, etc.
A standard curve of different known concentrations of analytes is required for the quantification of analytes in test samples. A standard curve is constructed by plotting known concentrations of analytes in x-axis and the absorbance obtained in y-axis. On the basis of it, the absorbance obtained from the samples can be interpolated to find the concentration of the analyte in the respective samples.
Advantages of Indirect ELISA
High Sensitivity: More than one enzyme-linked antibody can bind to an analyte-specific antibody, such that it amplifies the signal, resulting in increased sensitivity compared to direct ELISA.
Greater Flexibility: A single secondary antibody can be used for the detection of multiple primary antibodies from the same host species. Therefore, the assay is flexible as well as cost-effective.
Limitations of Indirect ELISA
Long and complex Protocol: Additional incubation and washing steps for the secondary antibody increase the time and complexity of the assay.
Increased chance of cross reactivity: While a specific antigen binds a specific antibody, sometimes cross-reaction between antigen and antibody of different infections recognizing the similar epitopes can occur. Also, since the same secondary antibody can be used for multiple primary antibodies from the same host species, there is a high risk of cross-reactivity and false positive results.
Applications of Indirect ELISA
Clinical diagnosis: Indirect ELISA is used to detect antibodies to infectious diseases, autoantibodies, or allergy testing in the samples.
Screening of blood: It can be used to screen the donated blood before its dispatch to the recipients. This can be helpful to prevent transmission of blood-borne infections.
Vaccine development and its efficacy: The immune response after vaccination can be measured through this ELISA assay to understand the effectiveness of the vaccine in individuals.
Laboratory research: It can be used in immune response evaluation through in house ELISA assay for research purposes on certain diseases.
Conclusion
In conclusion, indirect ELISA is a simple and highly sensitive serological testing technique, which has been used commercially for the diagnosis of various diseases, infections, and even allergy testing. It is a bit time-consuming, but it is also a reliable method that has been used for research-related work.
Although it has some disadvantages, it can be minimized by using a specific and unique antigen to avoid cross-reactivity and following the steps carefully to avoid any deviations in the data. The efficacy of the assay also depends upon proper storage of reagents, use of reagents in optimum concentration, and good handling practice of small-volume reagents.
References
- Alhajj, M., Zubair, M., Farhana, A. (2023). Enzyme Linked Immunosorbent Assay StatPearls- NCBI Bookshelf. https://www.ncbi.nlm.nih.gov/books /NBK555922/
- Boster Biological Technology (n.d.). ELISA Handbook [PDF]. Retrieved March 1, 2026, from https://www.bosterbio.com/ebooks
- Boston Industries, Inc. (2025, September 15). A Comprehensive Guide to Indirect ELISA (Enzyme-Linked Immunosorbent Assay). Retrieved March 1, 2026, from https://www.bostonind.com/blog/Guide-to-Indirect-ELISA
- Hayrapetyan, H., Tran, T., Tellez-Corrales, E., & Madiraju, C. (2023). Enzyme-Linked Immunosorbent Assay: Types and Applications. Methods in molecular biology (Clifton, N.J.),2612, 1–17. https://doi.org/10.1007/978-1-0716-2903-1_1
- Lin A. V. (2015). Indirect ELISA. Methods in molecular biology (Clifton, N.J.),1318, 51–59. https://doi.org/10.1007/978-1-4939-2742-5_5

please send pdf
i.e laboratory report sample about antibody antigen detection Newcastle