Human cytomegalovirus (HCMV), also known as human herpesvirus 5, is a ubiquitous β herpesvirus with a large double-stranded DNA genome of approximately 235–250 kb.
HCMV has the ability to persist in the host by latent infection of myeloid lineage cells.
- Global seroprevalence: ~60% in high-income countries and >90% in many low- and middle-income regions.
- Usually asymptomatic in immunocompetent hosts, but:
-Leading infectious cause of congenital sensorineural hearing loss and neurodisability.
-Major opportunistic pathogen in solid-organ and hematopoietic stem-cell transplant (HSCT) recipients and advanced HIV.
- High genetic complexity underpins broad cell tropism, extensive immune evasion, and resistance development.
The impact of HCMV on public health is thought to be due to direct organ disease and also to indirect effects on graft survival, cardiovascular disease, immune aging, and possibly autoimmunity and cancer.
Taxonomy and Classification of Human Cytomegalovirus (HCMV)
- Family: Herpesviridae
- Subfamily: Betaherpesvirinae
- Genus: Cytomegalovirus
- Species: Human cytomegalovirus (HCMV; HHV-5).
Key taxonomic/biological features:
- Betaherpesvirus traits:
-Slow lytic replication and marked cytomegaly of infected cells.
-Latency in myeloid progenitors/monocytes with reactivation upon differentiation and inflammation.
- Strict species specificity: each mammalian host harbors a co-evolved CMV with unique “private gene” families driving host-adapted immune evasion.
- A prototype of the genus and the largest known human DNA virus.
Structure and Morphology of Human Cytomegalovirus (HCMV)
Virion size: ~200–300 nm; icosahedral nucleocapsid ≈130 nm, T=16, 162 capsomers (12 pentons, 150 hexons).
- Nucleocapsid: proteinaceous shell enclosing linear dsDNA genome.
- Tegument: amorphous but partially ordered protein layer:
-Major proteins: pp65 (UL83), pp71 (UL82), pp150 (UL32), involved in immune modulation, transactivation, and virion assembly.
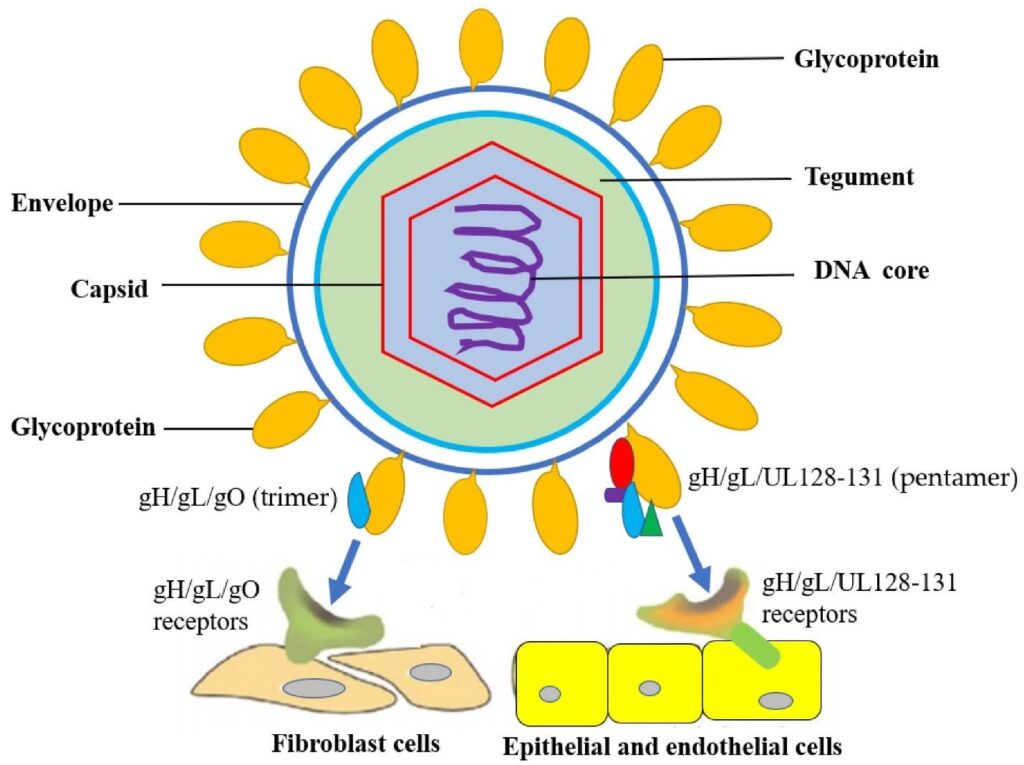
- Envelope: host-derived lipid bilayer with abundant glycoproteins and complexes critical for entry and tropism:
-gB (UL55) trimer – primary fusogen and dominant neutralizing antibody target.
-Trimeric complex: gH/gL/gO – essential for fibroblast entry (via PDGFRα).
-Pentameric complex: gH/gL/UL128-130-131 – required for epithelial, endothelial, and placental trophoblast entry (via NRP2, THBD, and others).
The structural features facilitate highly regulated and cell-type-specific entry mechanisms and inform vaccine and antibody development.
Genome Organization and Proteins of Human Cytomegalovirus (HCMV)
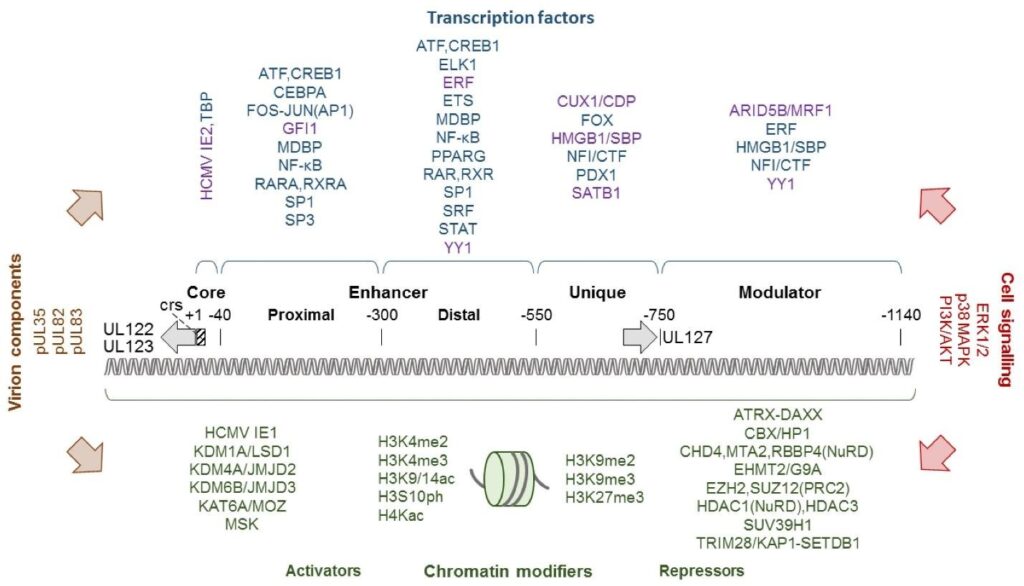
Genome type: linear dsDNA, class E herpesvirus architecture TRL–UL–IRL–IRS–US–TRS.
- Length ≈235–250 kbp; largest among human herpesviruses.
Main organizational features:
- Two unique regions:
-UL (unique long) and US (unique short), each flanked by internal and terminal inverted repeats (IRL/TRL, IRS/TRS).
- Coding capacity:
-≈170 canonical ORFs; however, ribosome profiling reveals >700 translated ORFs and >280 translationally active viral transcripts, indicating extensive use of alternative start sites and small ORFs.
-≥4 major lncRNAs (RNA1.2, 2.7, 4.9, 5.0) and at least 16 pre-miRNAs yielding -≥26 mature miRNAs, regulating host apoptosis, IFN responses, and antigen presentation.
-~40–45 core herpesvirus genes for replication and virion assembly (DNA polymerase, helicase-, primase, capsid, portal).
-Large complement of accessory/host-adaptation genes (RL11, US6, US12, US22 families, UL18, UL40, UL141, vIL-10, etc.) dedicated to immune evasion, cell tropism, and modulation of cell death.
Temporal gene expression cascade:
- Immediate-early (IE) – IE1 (UL123), IE2 (UL122):
-Activated from the major immediate-early promoter (MIEP).
-Reprogram chromatin, antagonize intrinsic silencing (Daxx/ATRX), and transactivate E genes.
- Early (E) – DNA replication machinery (UL54 DNA polymerase, UL44 processivity factor, helicase-primase), nucleotide metabolism, anti-apoptotic proteins.
- Late (L) – structural proteins (capsid, tegument, glycoproteins), assembly/egress factors.
Replication Cycle of Human Cytomegalovirus (HCMV)
Human cytomegalovirus has a complex replication cycle with tightly regulated stages that allow it to infect a wide range of cell types, achieve a latent infection, and reactivate under certain conditions. HCMV has a large double-stranded DNA genome of about 236 kbp with more than 165 genes that include replication and immune evasion genes.
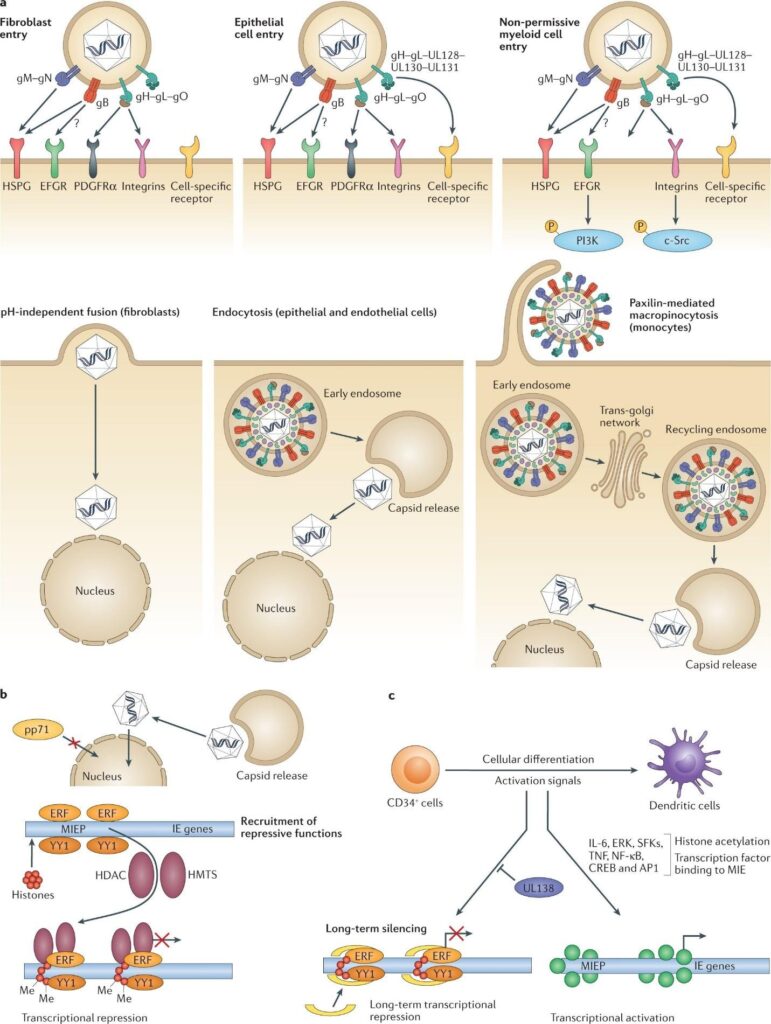
Attachment and Entry:
-Initial attachment occurs via interactions between viral glycoproteins and host cell surface heparan sulfate proteoglycans.
-Entry mechanisms are cell-type dependent: in fibroblasts, HCMV enters primarily through direct fusion at the plasma membrane mediated by the gH/gL/gO trimer complex engaging PDGFRα receptors; in epithelial, endothelial, and placental trophoblast cells, entry occurs via endocytosis followed by low-pH-triggered fusion facilitated by the pentameric gH/gL/UL128-131 complex interacting with receptors such as neuropilin-2 (NRP2) and thrombomodulin (THBD).
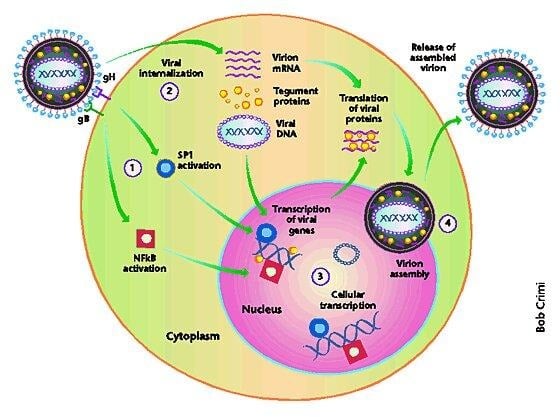
Uncoating and Nuclear Delivery:
-Following fusion or endocytosis, the capsid is transported along microtubules to the nuclear pore complex.
-Viral DNA is injected into the nucleus while tegument proteins like pp71 translocate to the nucleus to counteract intrinsic antiviral defenses by degrading repressive factors such as Daxx and ATRX, thereby activating the major immediate early promoter (MIEP).
Transcriptional Cascade:
The viral gene expression follows a temporal cascade:
- Immediate-Early (IE) genes (e.g., IE1/IE2) are expressed first to initiate transcriptional activation of early genes and modulate host chromatin.
- Early (E) genes encode proteins essential for viral DNA replication, including DNA polymerase (UL54), processivity factor (UL44), helicase-primase complex, and nucleotide metabolism enzymes.
- Late (L) genes produce structural proteins for capsid assembly, tegument components, and envelope glycoproteins necessary for virion maturation.
- DNA Replication:
-Occurs in discrete nuclear replication compartments where rolling-circle-like mechanisms generate concatemers of viral genomes.
-Viral DNA packaging into preformed capsids is mediated by terminase complexes recognizing specific packaging signals.
Assembly and Egress:
-Capsid assembly occurs in the nucleus with subsequent primary envelopment at the inner nuclear membrane.
-De-envelopment at the outer nuclear membrane releases capsids into the cytoplasm, where tegumentation occurs within a virus assembly compartment formed by remodeling of Golgi and endoplasmic reticulum membranes.
-Secondary envelopment takes place in cytoplasmic vesicles before mature virions are released via exocytosis.
Latency and Reactivation:
-In myeloid progenitor cells and monocytes, initial lytic gene expression is rapidly silenced through epigenetic repression of MIEP, leading to latency establishment with restricted viral transcription.
-Reactivation is triggered by inflammatory signals, cellular differentiation into dendritic cells/macrophages, or stress pathways that remodel chromatin to permit full lytic gene expression and productive infection.
This complex replication strategy enables HCMV to maintain lifelong persistence while balancing productive infection with latency to circumvent immune clearance.
Pathogenesis and Host Immune Response to Human Cytomegalovirus (HCMV)
HCMV pathogenesis reflects a balance of direct cytopathic effects of lytic replication and intricate interactions with the host’s immune response, which may combat infection and contribute to disease.
Direct Pathogenic Effects
- Lytic replication causes tissue destruction, manifesting as pneumonitis, colitis, hepatitis, retinitis, encephalitis, myocarditis, nephritis, especially in immunocompromised hosts.
- In congenital infection, viral replication in the developing brain leads to microcephaly, ventriculomegaly, intracranial calcifications, sensorineural hearing loss, intellectual disability, and epilepsy; major causes of long-term neurodevelopmental impairment.
Immune Evasion Strategies
- HCMV encodes multiple proteins targeting innate immune sensing pathways: it inhibits TLR signaling (e.g., TLR2/9), cGAS-STING pathway activation, and RIG-I-like receptor signaling to suppress type I interferon production.
- Tegument protein pp65 antagonizes IFI16 inflammasome activation; UL36/UL37 inhibit apoptosis/necroptosis pathways, preserving infected cells.
- To evade natural killer (NK) cells: downregulation of classical HLA class I molecules is compensated by expression of UL18 (an HLA-I homolog engaging inhibitory receptors), UL40 peptides stabilizing HLA-E for NK inhibition; viral IL-10 homologs modulate cytokine milieu, suppressing NK activity.
Adaptive Immune Responses
- Robust CD8+ cytotoxic T lymphocyte responses target IE antigens; CD4+ T cells provide help for antibody production and cytotoxicity; “memory inflation” leads to large persistent CMV-specific T-cell populations critical for controlling latent reservoirs.
- Viral proteins US2/US3/US6/US11 interfere with antigen presentation by degrading or retaining MHC class I molecules; downregulation of co-stimulatory molecules impairs effective T-cell activation.
- Humoral immunity targets glycoproteins gB and pentameric complexes but does not confer sterilizing immunity due to cell-associated spread and immune evasion mechanisms.
Latency-Reactivation Dynamics
- Latency involves epigenetic silencing of MIEP in myeloid progenitors; low-level transcription of latency-associated transcripts maintains viral genome persistence without virion production.
- Reactivation is induced by inflammatory cytokines (TNFα), Toll-like receptor ligands during co-infections or tissue injury; differentiation into macrophages/dendritic cells relieves repression, allowing lytic gene expression resumption.
- In immunocompromised patients or aging individuals with immunosenescence, impaired immune surveillance permits uncontrolled reactivation, leading to end-organ disease.
Epidemiology and Transmission of Human Cytomegalovirus (HCMV)
HCMV is globally widespread, with seroprevalence varying widely based on socioeconomic factors:
- Seroprevalence ranges from ~60% in developed countries up to >90% in developing regions, influenced strongly by crowding, ethnicity, and education level.
- Primary infection typically occurs in early childhood via close contact with saliva or urine from young children who shed virus extensively; sexual transmission contributes significantly during adolescence/adulthood.
Transmission Routes
- Horizontal transmission: through saliva and urine exposure, especially among children in daycare settings; sexual contact via semen/cervical secretions facilitates adult spread.
- Vertical transmission: transplacental passage during pregnancy causes congenital infection; intrapartum exposure through birth canal secretions; postnatal transmission via breastfeeding also occurs, but usually less pathogenic than congenital infection.
- Iatrogenic transmission: blood transfusions (especially non-leukoreduced products), solid organ transplantation (donor-to-recipient transmission), and hematopoietic stem cell transplantation represent important routes, particularly affecting immunocompromised hosts.
Epidemiological Impact
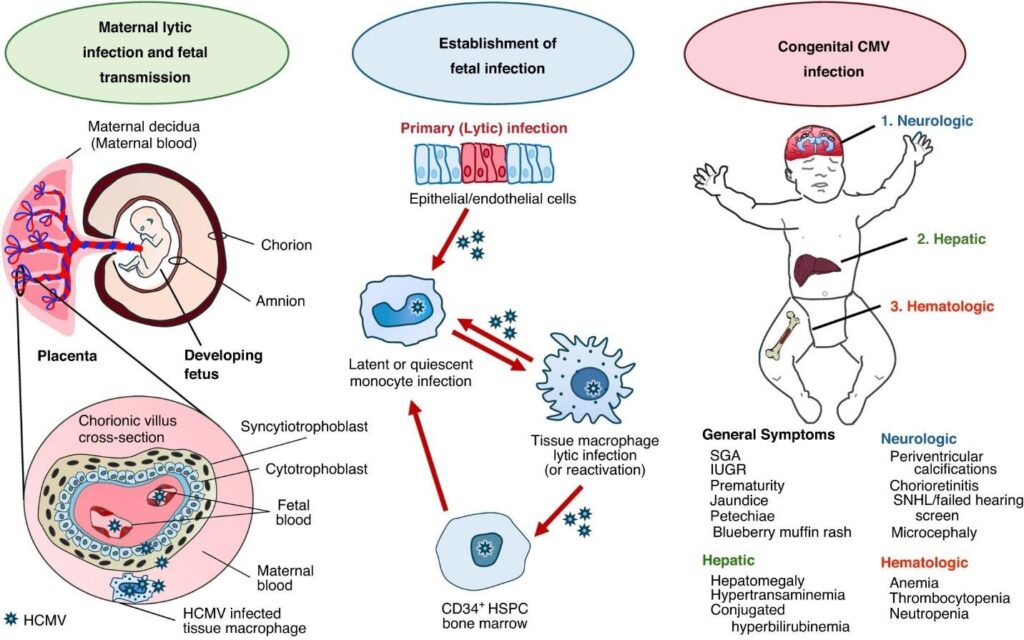
- Congenital CMV affects approximately 0.5–2% of live births worldwide, causing significant morbidity, including hearing loss and neurodevelopmental disabilities, even among asymptomatic neonates at birth.
- Immunosuppressed populations, such as transplant recipients, experience high rates of CMV reactivation, leading to morbidity/mortality despite antiviral prophylaxis strategies. Monitoring viral load kinetics guides preemptive therapy initiation, improving outcomes.
Clinical Manifestations of Human Cytomegalovirus (HCMV)
The spectrum of CMV infection varies from asymptomatic to severe disease, depending on the host’s immune status and age.
Immunocompetent Individuals:
- Often asymptomatic or mild mononucleosis-like syndrome with fever, malaise, lymphadenopathy, and atypical lymphocytosis.
- In infants and children, postnatal CMV can cause liver involvement (hepatitis), hematological abnormalities (anemia, thrombocytopenia), and gastrointestinal symptoms; neurological and pulmonary involvement is more common in older children.
- CMV anterior uveitis (AU) is increasingly recognized in immunocompetent adults, especially in Asia, characterized by recurrent hypertensive iritis resembling Posner-Schlossman syndrome or chronic inflammation mimicking Fuchs uveitis; elevated intraocular pressure may lead to glaucoma if untreated.
Congenital CMV (cCMV):
- Leading non-genetic cause of sensorineural hearing loss and neurodevelopmental disabilities worldwide.
- Symptomatic neonates (~10–15%) present with microcephaly, intracranial calcifications, hepatosplenomegaly, thrombocytopenia, chorioretinitis, jaundice, and growth restriction.
- Asymptomatic infants at birth may develop late-onset sequelae such as hearing loss or cognitive impairment.
Immunocompromised Hosts:
- Solid organ transplant (SOT) and hematopoietic stem cell transplant (HSCT) recipients are at high risk for CMV syndrome (fever, malaise, cytopenia) and end-organ diseases, including pneumonitis, colitis, hepatitis, nephritis, myocarditis, and retinitis.
- CMV retinitis is the most common ocular manifestation in people living with HIV/AIDS with low CD4 counts (<50 cells/μL); it typically begins peripherally and progresses centrally, causing retinal necrosis (“pizza” appearance), leading to vision loss if untreated.
- Gastrointestinal CMV disease affects both immunocompromised and immunocompetent patients, but is more severe in the former; symptoms include abdominal pain, diarrhea, GI bleeding. Diagnosis often requires histopathology due to nonspecific clinical/endoscopic features
Laboratory Diagnosis of Human Cytomegalovirus (HCMV)
The diagnosis of CMV infection is based on a combination of molecular tests, antigen detection, serological tests, culture, and histopathological examination.
Molecular Testing:
- Quantitative PCR (qPCR) detecting CMV DNA in plasma or whole blood is the gold standard for diagnosing active infection and monitoring viral load in transplant recipients and other high-risk groups.
- PCR on body fluids (e.g., cerebrospinal fluid for CNS disease), bronchoalveolar lavage for pneumonitis, or tissue biopsies enhances diagnostic sensitivity for end-organ disease.
- Saliva or urine PCR within the first three weeks of life is preferred for congenital infection screening due to high sensitivity and ease of collection.
Antigen Detection:
- pp65 antigenemia assay detects viral protein in leukocytes; useful for rapid diagnosis but less sensitive than PCR; still used in some centers, especially for HSCT patients.
Serology:
- IgG serostatus determines prior exposure; IgM positivity combined with low IgG avidity suggests recent primary infection important in pregnancy risk assessment.
- Not useful alone for diagnosing active disease.
Culture:
- Shell vial culture accelerates virus isolation from clinical specimens, but has largely been replaced by molecular methods due to longer turnaround times.
Histopathology:
- “Owl’s eye” intranuclear inclusions on tissue biopsy remain the diagnostic gold standard for invasive disease. Immunohistochemistry staining improves sensitivity.
Emerging Diagnostics:
- Measurement of CMV-specific T-cell immunity by ELISPOT or flow cytometry-based assays helps predict risk of reactivation post-transplantation and guide therapy decisions.
Treatment and Antiviral Therapy for Human Cytomegalovirus (HCMV)
The decision to treat depends upon the immune status of the host, the severity of the disease, the location of the infection, and the presence of resistant mutations. The primary indications of antiviral treatment are symptomatic immunocompromised patients and symptomatic congenital infections.
Licensed Antiviral Agents
Ganciclovir / Valganciclovir:
- First-line agents inhibit viral DNA polymerase after phosphorylation by UL97 kinase. Valganciclovir offers oral bioavailability.
- Used for induction therapy followed by maintenance/prophylaxis in transplant recipients. Also, standard treatment for symptomatic cCMV infants (oral valganciclovir recommended for up to 12 months, improving hearing/neurodevelopmental outcomes).
- Adverse effects: myelosuppression (neutropenia), nephrotoxicity.
Foscarnet:
- A pyrophosphate analogue inhibiting DNA polymerase directly without requiring phosphorylation. Used mainly for ganciclovir-resistant infections or intolerance. Nephrotoxicity limits use.
Cidofovir / Brincidofovir:
- Nucleotide analogues active against resistant strains but limited by toxicity profiles; cidofovir causes nephrotoxicity; brincidofovir has better safety, but GI side effects restrict use.
Letermovir:
- Targets viral terminase complex UL56/UL89; approved for prophylaxis in CMV-seropositive HSCT recipients with a favorable safety profile. Resistance mutations are emerging, but with less cross-resistance with polymerase inhibitors.
Maribavir:
- UL97 kinase inhibitor blocking viral DNA encapsidation/egress rather than polymerase activity; approved for refractory/resistant CMV infections post-transplantation with a distinct resistance profile from ganciclovir/foscarnet.
Treatment Strategies
- Congenital CMV: Oral valganciclovir recommended for symptomatic neonates up to one year; no current evidence supports treatment of asymptomatic infants, though ongoing studies aim to clarify this area.
- Immunocompromised Hosts: Preemptive therapy guided by viral load monitoring reduces overt disease incidence; universal prophylaxis is used variably depending on transplant type/risk factors. Combination antiviral therapy (>14 days) improves outcomes, especially in gastrointestinal disease, regardless of immune status.
- CMV Retinitis: Early initiation of systemic antivirals is critical to prevent vision loss; adjunctive therapies include intravitreal ganciclovir injections or implants; emerging immunotherapies such as adoptive T-cell transfer show promise with reduced toxicity/mortality compared to antivirals alone.
Antiviral Resistance
- Resistance arises mainly via mutations in the UL97 kinase gene, conferring ganciclovir resistance or the UL54 DNA polymerase gene, causing multidrug resistance, including foscarnet/cidofovir resistance. Letermovir resistance linked to UL56 mutations occurs less frequently but requires vigilance during prophylaxis/treatment failures.
Emerging Therapies
- Immunotherapies, including adoptive transfer of CMV-specific T cells, are increasingly used, especially in post-HSCT refractory cases, showing improved survival and reduced drug toxicity risks compared to prolonged antiviral use alone.
- Novel agents targeting host factors essential for viral replication or CRISPR/Cas-based gene editing approaches are under preclinical investigation, aiming at overcoming resistance issues and improving safety profiles.
Prevention and Control of Human Cytomegalovirus (HCMV)
General and Clinical Measures
- Rigorous hand hygiene and avoidance of saliva/urine exposure from young children, especially for seronegative pregnant women.
- Safe sex practices, screening, and appropriate processing (e.g., leucoreduction) of blood products.
- In transplantation:
-D/R serostatus-guided risk stratification.
-Universal prophylaxis vs pre-emptive therapy strategies using viral load monitoring.
Vaccine Development
In spite of over 50+ years of research, there still isn’t a licensed HCMV vaccine. However, it’s agreed upon that an effective vaccine is “within reach.”
- Targets and rationales:
-gB: induces binding/neutralizing antibodies; first-generation subunit vaccines achieved ~50% efficacy in some trials.
-Pentameric complex: critical for epithelial/placental tropism; strong target for neutralizing antibodies.
-T-cell antigens (pp65, IE1/2) to drive robust cellular responses.
- Platforms under active evaluation:
-Live-attenuated, vectored, subunit (gB ± pentamer), DNA and mRNA vaccines.
- Barriers:
-Profound immune evasion, incomplete correlates of protection, species-specificity limiting animal models, and challenges in designing trials powered for congenital outcomes.
- Even partially effective vaccines (reducing maternal primary infection or viral load) are projected to yield major public-health benefits.
Conclusion
Human cytomegalovirus (HCMV) is a highly adapted beta-herpesvirus with a large, complex genome. This genome is characterized by extraordinary coding potential to manipulate host immunity, cell death, and tissue tropism. HCMV is known to establish a latent infection in myeloid cells, reactivates in response to immune perturbation, and is capable of dissemination to all organs of the body. This makes it a primary cause of congenital infection and opportunistic infection in immunocompromised patients.
Some of the current research in human cytomegalovirus is focusing on certain key issues, such as the cell biology of infection, tissue-specific host immunity, and genetic diversity, which is useful in developing future antiviral strategies, host-targeting antiviral strategies, cellular immunotherapy, and vaccines against human cytomegalovirus infection. It is of great importance to integrate this with clinical investigation to translate current mechanistic understanding to control of this infection in vulnerable populations.
Reference
- Adamson, C. S., & Nevels, M. M. (2020). Bright and Early: Inhibiting Human Cytomegalovirus by Targeting Major Immediate-Early Gene Expression or Protein Function. Viruses, 12(1), 110. https://doi.org/10.3390/v12010110
- Becker, S., Fink, A., Podlech, J., Reddehase, M., & Lemmermann, N. (2022). Host-Adapted Gene Families Involved in Murine Cytomegalovirus Immune Evasion. Viruses, 14. https://doi.org/10.3390/v14010128
- Bottino, P., Pastrone, L., Curtoni, A., Bondi, A., Sidoti, F., Zanotto, E., Cavallo, R., Solidoro, P., & Costa, C. (2023). Antiviral Approach to Cytomegalovirus Infection: An Overview of Conventional and Novel Strategies. Microorganisms, 11. https://doi.org/10.3390/microorganisms11102372
- Chen, S.-J., Wang, S.-C., & Chen, Y.-C. (2022). Challenges, Recent Advances and Perspectives in the Treatment of Human Cytomegalovirus Infections. Tropical Medicine and Infectious Disease, 7(12), 439. https://doi.org/10.3390/tropicalmed7120439
- Grgic, I., & Gorenec, L. (2024). Human Cytomegalovirus (HCMV) Genetic Diversity, Drug Resistance Testing and Prevalence of the Resistance Mutations: A Literature Review. Tropical Medicine and Infectious Disease, 9. https://doi.org/10.3390/tropicalmed9020049
- Griffiths, P., & Reeves, M. (2021). Pathogenesis of human cytomegalovirus in the immunocompromised host. Nature Reviews. Microbiology, 19, 759–773. https://doi.org/10.1038/s41579-021-00582-z
- Gugliesi, F., Coscia, A., Griffante, G., Galitska, G., Pasquero, S., Albano, C., & Biolatti, M. (2020). Where do we stand after Decades of Studying Human Cytomegalovirus? Microorganisms, 8. https://doi.org/10.3390/microorganisms8050685
- Gugliesi, F., Pasquero, S., Griffante, G., Scutera, S., Albano, C., Pacheco, S. F. C., Riva, G., Dell’Oste, V., & Biolatti, M. (2021). Human Cytomegalovirus and Autoimmune Diseases: Where Are We? Viruses, 13. https://doi.org/10.3390/v13020260
- Huang, E.-S., & Johnson, R. A. (2000). Human cytomegalovirus – no longer just a DNA virus. Nature Medicine, 6(8), 863–864. https://doi.org/10.1038/78612
- Krstanović, F., Britt, W., Jonjić, S., & Brizić, I. (2021). Cytomegalovirus Infection and Inflammation in the Developing Brain. Viruses, 13. https://doi.org/10.3390/v13061078
- Lawrence, S. M., Goshia, T., Sinha, M., Fraley, S. I., & Williams, M. (2024). Decoding human cytomegalovirus for the development of innovative diagnostics to detect congenital infection. Pediatric Research, 95(2), 532–542. https://doi.org/10.1038/s41390-023-02957-9
- Lee, C.-H., & Grey, F. (2020). Systems Virology and Human Cytomegalovirus: Using High Throughput Approaches to Identify Novel Host-Virus Interactions During Lytic Infection. Frontiers in Cellular and Infection Microbiology, 10. https://doi.org/10.3389/fcimb.2020.00280
- Liu, X., Liu, C., & Zhang, T. (2025). The Immunoregulatory Mechanisms of Human Cytomegalovirus from Primary Infection to Reactivation. Pathogens, 14. https://doi.org/10.3390/pathogens14100998
- Majewska, A., & Młynarczyk-Bonikowska, B. (2022). 40 Years after the Registration of Acyclovir: Do We Need New Anti-Herpetic Drugs? International Journal of Molecular Sciences, 23. https://doi.org/10.3390/ijms23073431
- Mihalić, A., Železnjak, J., Lisnić, B., Jonjić, S., Lisnić, V. J., & Brizić, I. (2024). Immune surveillance of cytomegalovirus in tissues. Cellular and Molecular Immunology, 21, 959–981. https://doi.org/10.1038/s41423-024-01186-2
- Permar, S., Schleiss, M., & Plotkin, S. (2025). A vaccine against cytomegalovirus: How close are we? The Journal of Clinical Investigation, 135. https://doi.org/10.1172/jci182317
- Preston, H., Casey, R., Ferris, E., Kerr-Jones, L., Jones, L., Latif, F., Clement, M., Aicheler, R., Wang, E., Stanton, R., & Fielding, C. (2025). Human Cytomegalovirus Immune Evasion of Natural Killer Cells: A Virus for All Seasons? Pathogens, 14. https://doi.org/10.3390/pathogens14070629
- Reeves, M. (2025). Cell Biology of Human Cytomegalovirus Latency: Implications for Pathogenesis and Treatment. Reviews in Medical Virology, 35. https://doi.org/10.1002/rmv.70063
- Scarpini, S., Morigi, F., Betti, L., Dondi, A., Biagi, C., & Lanari, M. (2021). Development of a Vaccine against Human Cytomegalovirus: Advances, Barriers, and Implications for Clinical Practice. Vaccines, 9. https://doi.org/10.3390/vaccines9060551
- Shang, Z., & Li, X. (2024). Human cytomegalovirus: Pathogenesis, prevention, and treatment. Molecular Biomedicine, 5. https://doi.org/10.1186/s43556-024-00226-7
- Venturini, C., & Breuer, J. (2025). Cytomegalovirus Genetic Diversity and Evolution: Insights into Genotypes and Their Role in Viral Pathogenesis. Pathogens, 14. https://doi.org/10.3390/pathogens14010050
- Xia, Y., Zhang, J., Shui, X., Wang, C., Chai, W., & Wang, C. (2026). Recent advances in human cytomegalovirus: A comprehensive review of pathogenic mechanisms, virus-host interactions, and antiviral strategies. Frontiers in Immunology. https://doi.org/10.3389/fimmu.2025.1636978
- Ye, L., Qian, Y., Yu, W., Guo, G., Wang, H., & Xue, X. (2020). Functional Profile of Human Cytomegalovirus Genes and Their Associated Diseases: A Review. Frontiers in Microbiology, 11. https://doi.org/10.3389/fmicb.2020.02104
- Yu, C., He, S., Zhu, W., Ru, P., Ge, X., & Govindasamy, K. (2023). Human cytomegalovirus in cancer: The mechanism of HCMV-induced carcinogenesis and its therapeutic potential. Frontiers in Cellular and Infection Microbiology, 13. https://doi.org/10.3389/fcimb.2023.1202138
