- Grocott-Gomori’s Methenamine Silver (GMS) stain is a histological stain that is used majorly for the identification of carbohydrates in fungal microorganisms.
- This staining method was named after György Gömöri, a physician from Hungary, who developed the staining methodology.
- Its initial application to assess missing tissues and diseases in the liver and the rectum (Nadworny, Wang, Tredget, & Robert, 2010) and then used for the identification of Pneumocystis jiroveci, a fungus known to cause an opportunistic infection called pneumocytosis, in immunocompromised and immunosuppressed patients.
- In comparison to other stains like Periodic Acid-Schiffs stain and Gridley Stain, Goromi has a higher sensitivity to for detecting fungi and other polysaccharide-rich microorganisms in paraffin prepared sections.
- Gomori’s methenamine-silver nitrate and chromic acid comprise the major reagents used in conventional Grocott stain
- It has also been used for the identification of fungi in tissue sections.
- Its application in Histology makes it ideal for the detection and demonstration of fungi in aspirates, tissues, and smears.
Objectives
- To demonstrate the presence of fungi in a given sample.
- To demonstrate the presence of Pneumocystis jiroveci and Histoplasma spp.
Principle of Grocott-Gomori’s Methenamine Silver Staining
The fungal cell wall is composed of polysaccharides which on interaction with chromatin Acid, undergo oxidation to form aldehydes. This is demonstrated by the reduction of the alkaline-hexamine-silver complex.
Grocott’s alkaline hexamine-silver solution undergoes reduction to form precipitates of silver ions making the cell wall of the fungi appear black a reaction is known as argentaffin reaction (the ability of cells to reduce the silver solution to metallic silver forming a black tissue element). Argentaffin cells are found of the epithelial lining of the lungs, the intestines, and melanin. This reaction drives the outcome of the result of the stain.
The staining methodologies are of two types:
- The conventional method at room temperature
- Microwave method
Reagents
Chromium trioxide solution, Sodium Bisulfite solution, Silver nitrate solution, methenamine solution, Borax solution, Gold chloride solution, sodium thiosulfate solution, Light green stock solution.
Preparation of solution
NOTE: These Reagents normally come in Kits for manual preparation and some solutions are already prepared
- Chromic Acid(1.4%): Chromium trioxide 4g + 100ml Distilled water
- Silver solution: 3% Methanamine/hexamine 23ml + 5% Silver Nitrate 1.25ml + 5% Borax(Sodium tetraborate) 3ml +25ml distilled water
- 3.2% Sodium Chloroaurate (Yellow gold chloride): Gold chloride 1.0g + 500ml Distilled water
- 4.2 % Sodium thiosulphate (Hypo): Sodium thiosulphate 2.0g + 100 ml Distilled water
- Working Light Green Stock Solution: 10ml of 1% light Green in 1% Acetic Acid + 40ml Distilled water
Note: Prepare the solutions depending on the concentrations you need to staining.
Procedure of Grocott-Gomori’s Methenamine Silver Staining
Conventional Procedure
- Hydrate sections with distilled water.
- Then oxidize the section with 4% aqueous chromic acid at room temperature and leave for 1 hr
- Wash in water for a few seconds
- Treat the sections with 1% sodium metabisulphite for 1 min
- Wash in smoothly running tap water for 3 mins
- Rinse thoroughly in distilled water
- Put the slides in pre-heated working silver solution in a water bath at 60°C for 15 to 20 mins until the section turns yellowish-brown (Check microscopically to see fungi turned dark brown).
- Rinse well in distilled water
- Add to the sections 0.2% gold chloride and leave for 2 mins
- Rinse well in distilled water
- Treat the sections with 2% sodium thiosulphate for 2 mins
- Wash with smoothly running tap water for 5 mins
- add the counterstain into the light green for 15 sec
- Rinse the excess light green solution off the slide with alcohol
- Dehydrate, clear and mount.
Microwave Procedure
- Hydrate the sections to distilled water.
- Place slides in 40 ml 1. 4% aqueous chromic acid in a loosely covered plastic Coplin jar.
- Microwave at 150 Watt for 2 min 30 sec
- Dip the slides up and down in the Coplin jar and let them stand for an extra 2 min
- Wash in smoothly running tap water 30 sec
- Add 3.2% sodium metabisulphite to the sections and leave for 10 sec and wash with tap water for 30 sec.
- Preheat the working silver solution at 450 watts for 60 sec
- and rinse the slide thoroughly with distilled water then place the slides sections in the hot silver solution.
- Microwave the sections at 150watt for 30 sec
- Dip slides up and down in the Coplin jar and allows to stand for another 1 minute. Rinse with distilled water and check under the microscope. Note: Dip back the slides in the silver solution if they are not stained sufficiently and check every 1 minute until the fungi stains dark brown.
- Tone the sections with 3.2% gold chloride 30 sec and rinse with distilled water.
- Add 4.2% of Sodium thiosulphate to the section for 1 min and wash with tap water for 15 sec.
- Counterstain the section with the light green solution for 15 sec.
- Rinse off the light green solution with alcohol
- Dry, and Cover.
Results
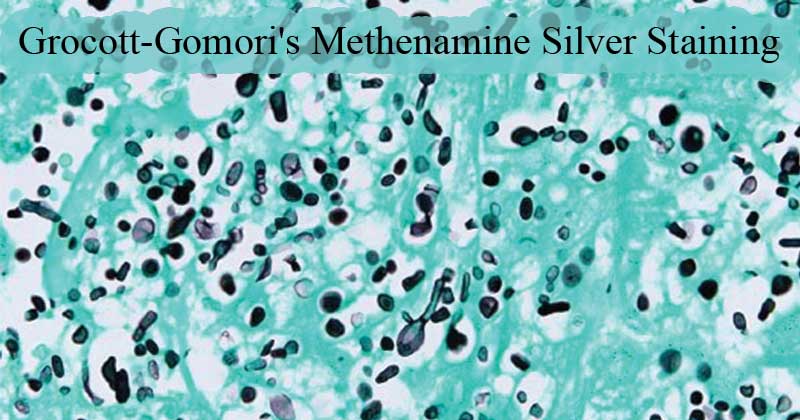
Figure: Fungal organisms are highlighted by the Grocott methenamine silver special stain. Hyphae are 4-6 um wide; septate fungal hyphae with nonparallel walls, non-dichotomous branching, and terminal bulbous dilations. Source: DOI: 10.1177/1040638716640313
- Fungi, Pneumocystis jirevoci, Histoplasma spp stain black
- Inner parts of mycelia and hyphae stain pink-red/ rose
- Leishmania spp, Toxoplasma spp – negative
- Mucin stains dark grey
- The background takes up the light green and stains pale green
Interpretation
The fungal species will stain black due to the reduction process of the silver nitrate solution. Silver Nitrate solution after reduction forms silver ions which are black in color, thus producing a black stain for fungal cells.
The fungal mycelium and hyphae, stain rose pink/pink-red, while the mucin stains dark grey also due to the reduction of silver nitrate to silver ions. the background will appear pale green by taking up the light green solution.
CAUTION
- Wear gloves, goggles and laboratory coats. Keep hot uncapped solutions under the fume hood. Avoid contact and inhalation of dyes and chemicals.
- Chromic acid is corrosive to skin and mucous membranes and Highly toxic affecting the Kidneys and itas also very carcinogenic
- Sodium metabisulfite ais Toxic if ingested and can cause Gastrointestinal distress and irritability. It also Irritates the skin, eye and the mucous membrane.
- Silver nitrate is an irritant to the skin and eyes.
- The oxidizer if ingested caused production of a violent gastrointestinal discomfort and it is possibly carcinogenic since its a tumorigenic agent.
- Sodium thiosulfate is toxic in ingestion and it causes stomach, skin, eyes and respiratory tract irritation.
- Light Green SF Yellowish is considered to be a possible carcinogen.
Applications of Grocott-Gomori’s Methenamine Silver Staining
- used to identify fungal cells in tissue sections
- used to differentiate fungal cells from bacterial cells
- also used to identify yeast-like fungi such as Pneumocystis jiroveci
Advantages
- It is an aggressive stain that fixes the stained sections firmly and can be used or future reference and observation.
- Tt can differentiate various fungal morphologies of fungi.
Limitations
- The chemical reagents can cause irritations of the skin, gastrointestinal tract if inhaled.
- The reagents also have carcinogenic effects.
Reference
- Grocott, R.G. 1955 , A stain for fungi in tissue sections and smears. American Journal of Clinical Pathology, V25, p975
- Alturkistani, Hani & Tashkandi, Faris & Mohammedsaleh, Zuhair. (2015). Histological Stains: A Literature Review and Case Study. Global Journal of Health Science. 8. 10.5539/gjhs.v8n3p72.
- http://www.ihcworld.com/_protocols/special_stains/grocott_methenamine_ellis.htm
- https://www.clinisciences.com/autres-produits-186/grocott-methenamine-silver-stain-351002431.html
- https://webpath.med.utah.edu/HISTHTML/MANUALS/GMS.PDF
- https://www.researchgate.net/post/Which_stains_can_be_used_for_Staining_Fungi
- https://www.coursehero.com/file/p342qaq/Grocotts-methenamine-silver-staining-protocol-Principle-Chromic-acid-oxidation/
- https://www.reference.com/science/nitric-acid-stain-skin-yellow-9475502370e787b5
- https://quizlet.com/185455989/histology-stains-flash-cards/
- http://pesticideinfo.org/Detail_Chemical.jsp?Rec_Id=PC33841

I read your post about the Gomori Grocott stain. I have a question about how you dispose of the stain waste, especially since some are classified as carcinogenic. I appreciate your post; it has been very helpful in understanding the technique.