Glycoconjugates are complex biomolecules formed by the covalent attachment of carbohydrates to proteins or lipids and are essential for cellular structure and interactions. Categorized primarily into glycoproteins, proteoglycans, and glycolipids, these molecules are synthesized through enzymatic glycosylation in the ER and Golgi apparatus.

By forming the glycocalyx on the cell surface, they act as critical mediators for molecular recognition, immune signaling, and cell-cell adhesion. Beyond structural support in the extracellular matrix, glycoconjugates dictate protein folding, stability, and solubility. Their immense structural diversity allows them to serve as a biological coding system, where aberrant expression often correlates with malignancy and metabolic disorders.
Three Major Classes: Glycoproteins, Glycolipids, and Proteoglycans
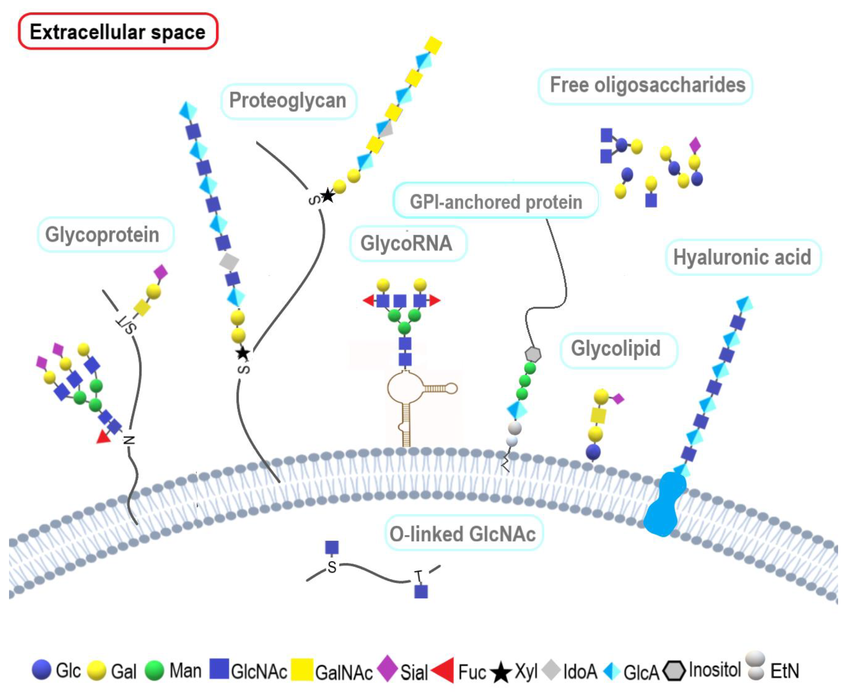
Glycoproteins
Glycoproteins are essential molecules consisting of a protein backbone covalently bonded to branched carbohydrate chains known as oligosaccharides. These sugars are typically attached via N-linkages to asparagine or O-linkages to serine and threonine. Primarily located on the outer surface of cell membranes or secreted into biological fluids, they act as critical identity tags. Their functions are diverse, ranging from cell-to-cell recognition and signaling to serving as receptors for hormones and viruses. In the immune system, most antibodies and major histocompatibility complex (MHC) molecules are glycoproteins. Additionally, they provide structural support and lubrication, as seen in the mucins of the digestive tract.
Glycolipids
These are vital membrane molecules composed of a carbohydrate group covalently bonded to a hydrophobic lipid tail, such as a sphingosine or glycerol backbone. Situated primarily on the outer lamella of the lipid bilayer, these amphipathic molecules extend their sugar chains into the extracellular space to form part of the glycocalyx, a dense network covering the exterior of the cell. They are fundamental for membrane stability, forming hydrogen bonds with the surrounding water to maintain structural integrity. Functionally, glycolipids serve as vital markers for cell-to-cell recognition and signaling for example, they determine human ABO blood types and act as receptors for toxins and pathogens. In the nervous system, specific glycolipids like cerebrosides and gangliosides are crucial for insulating nerve fibers and facilitating neurodevelopment.
Proteoglycans
A unique subclass of glycoproteins characterized by an exceptionally high carbohydrate content, often exceeding 95% of their total mass. They consist of a central core protein covalently linked to one or more glycosaminoglycans (GAGs) long, unbranched polysaccharide chains made of repeating disaccharide units. Because GAGs are heavily sulphated and negatively charged, they are highly hydrophilic and attract large volumes of water. This creates a hydrated, gel-like matrix that is essential for structural support and shock absorption in connective tissues like cartilage. Beyond their mechanical role in the extracellular matrix, proteoglycans also act as dynamic reservoirs for growth factors and participate in vital cell signalling processes that regulate tissue repair and development.
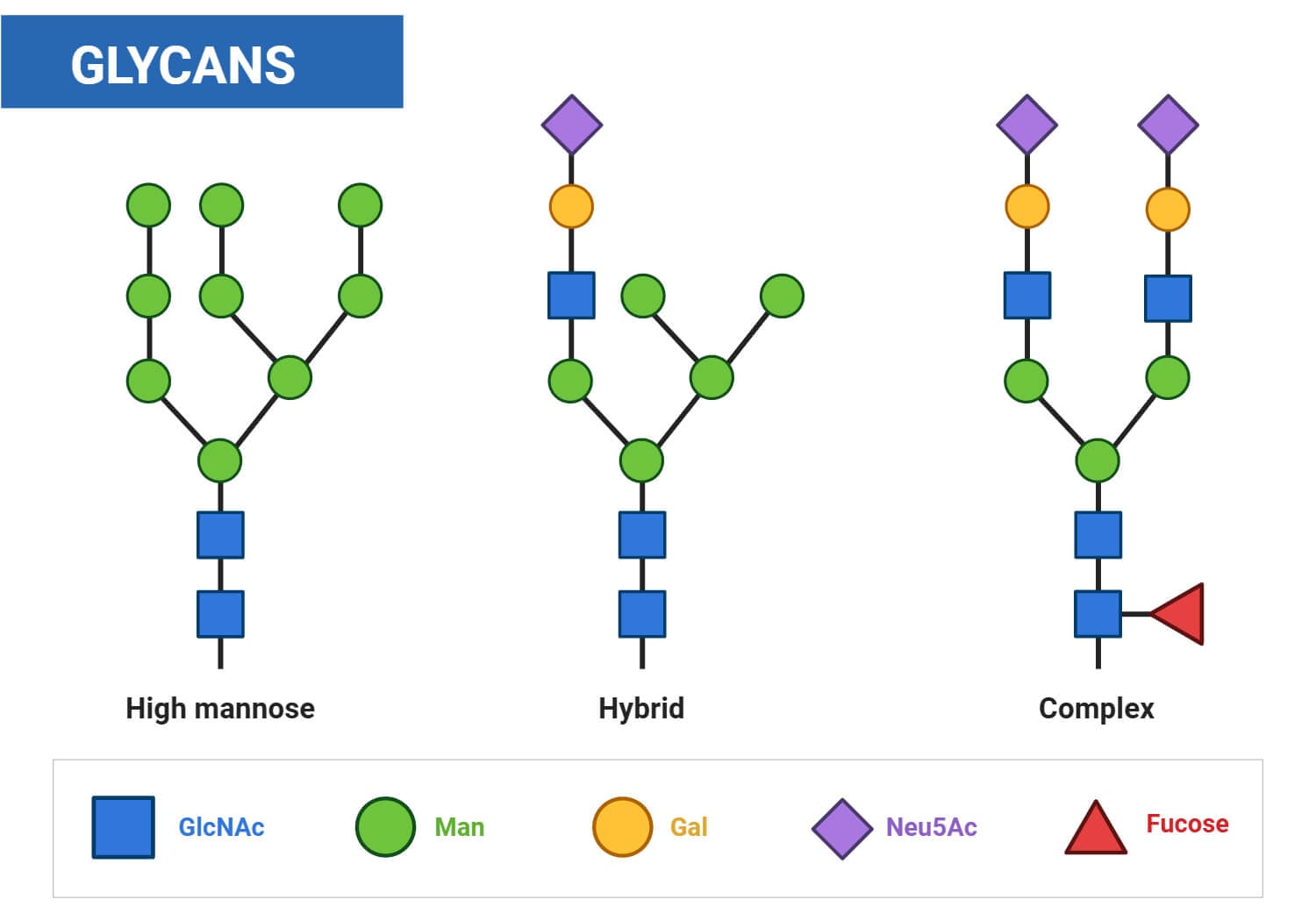
Glycosylation Explained: N-linked vs. O-linked Modifications
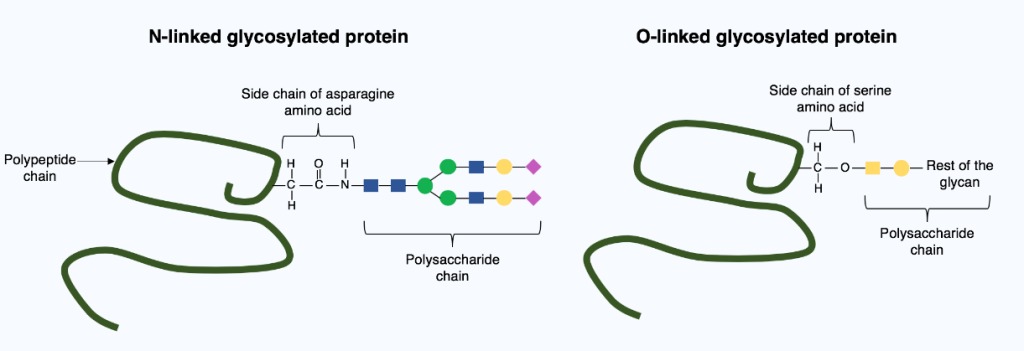
Glycosylation is a post-translational modification that facilitates complex molecular programming, thereby expanding the proteome’s functional range. The covalent integration of carbohydrate moieties (glycans) into polypeptide chains is a highly regulated process that dictates protein folding kinetics, thermodynamic integrity, and downstream trafficking. The eukaryotic glycoproteome is fundamentally shaped by two divergent biosynthetic trajectories: N-linked and O-linked glycosylation. While both processes involve the covalent conjugation of glycan moieties to polypeptide backbones, they are mechanistically delineated by their site-specific stereochemistry, discrete subcellular compartmentalization, and the rigorous enzymatic hierarchies that govern their assembly.
https://commons.wikimedia.org/wiki/File:Glycosylation_of_a_polypeptide.png
N-Linked
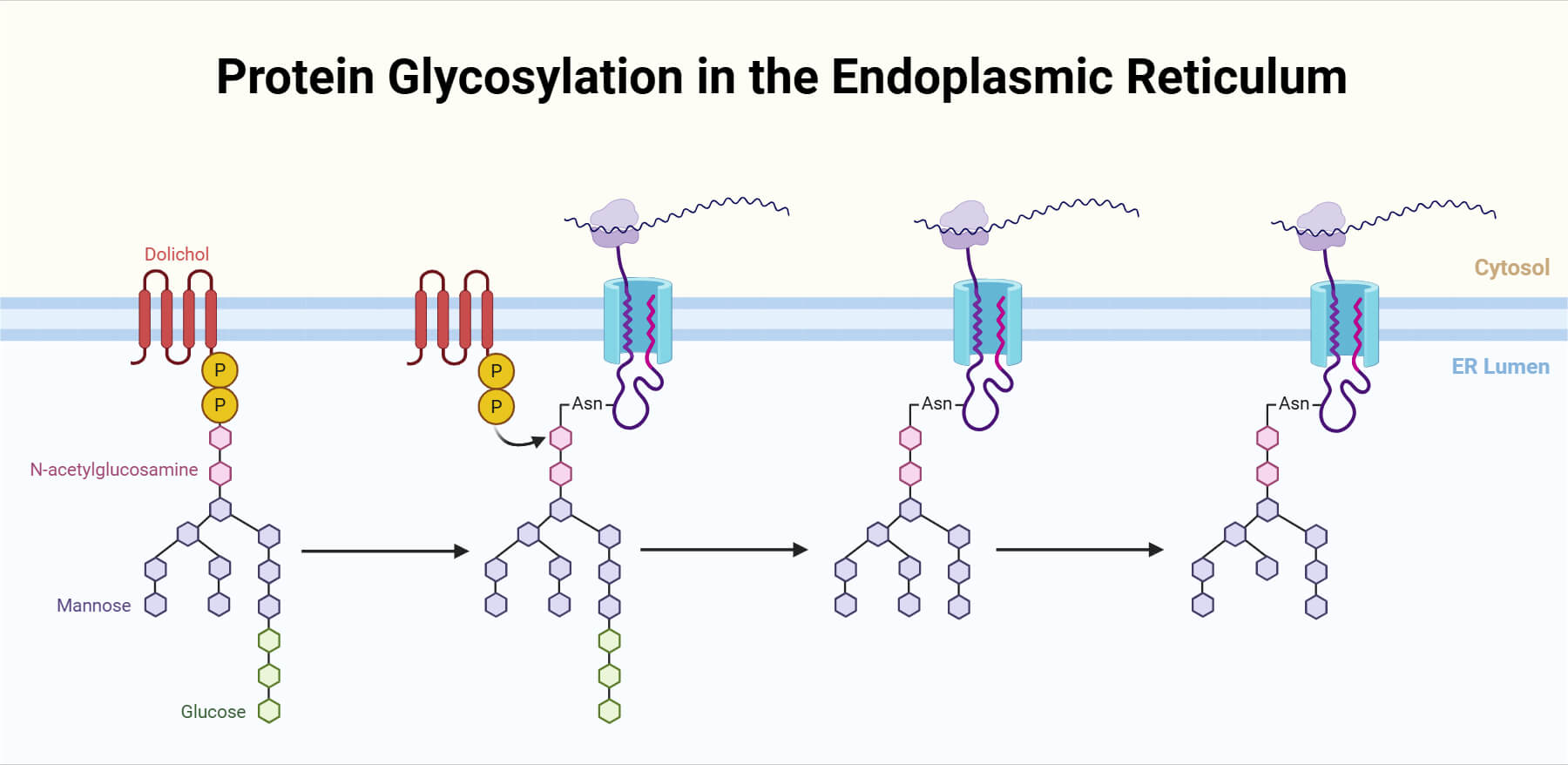
N-linked glycosylation is defined as the co-translational transfer of a pre-assembled, 14-sugar oligosaccharide precursor to the amide nitrogen of an asparagine residue. This high-fidelity process is strictly regulated by the consensus sequon Asn-X-Ser/Thr which is a specific sequence of amino acids in a protein that serves as a signature or target site for the attachment of a carbohydrate chain, (X is any amino acid except proline) and begins within the lumen of the endoplasmic reticulum (ER). The assembly of this precursor typically consisting of 2 N-acetylglucosamines, 9 mannoses, and 3 glucoses occurring on a specialized lipid carrier called dolichol pyrophosphate, which flips across the ER membrane to allow the enzyme oligosaccharyltransferase (OST) to catalyze the glycan attachment to the nascent polypeptide. As a Post-Translational Modification (PTM), N-linked glycosylation is indispensable for protein quality control, as the attached glycans act as molecular signals in the calnexin/calreticulin cycle to ensure proteins are correctly folded before exiting to the Golgi apparatus. Beyond folding, this PTM fundamentally dictates the protein’s thermodynamic stability, prevents unwanted intracellular aggregation, and provides essential ligands for cell-cell recognition and immune signalling, making it a master regulator of the functional glycoproteome.
O-linked glycosylation
O-linked glycosylation is defined as the post-translational, stepwise attachment of monosaccharides to the hydroxyl oxygen of serine or threonine residues. Unlike the N-linked pathway, these glycans are built incrementally, typically initiated by GalNAc within the Golgi apparatus after the protein has achieved its native fold. Crucially, this process lacks a strict consensus sequence; instead, site selection is governed by the protein’s secondary structure and the local concentration of proline residues. As a master PTM, O-glycosylation modulates biophysical properties by creating dense bottle-brush formations that provide lubrication and a robust proteolytic shield against degradation. Furthermore, these modifications act as vital identity markers, determining human ABO blood groups and serving as ligands for selections during leukocyte trafficking to facilitate precise intercellular communication.
Glycoproteins: Structure and Role in Immunity (Antibodies)
Glycoproteins are essential hybrid molecules consisting of carbohydrate chains covalently bonded to proteins, a structure that dictates their folding, stability, and biological activity. In the immune system, they function as the primary tools for recognition and defense, most notably as antibodies (immunoglobulins). These Y-shaped glycoproteins use their glycan components to maintain structural integrity and trigger effector functions, allowing the immune system to identify and neutralize pathogens. Beyond antibodies, glycoproteins on cell surfaces facilitate vital communication and self vs. non-self-recognition, ensuring that immune cells can accurately target invaders while sparing healthy tissue.
Structure of Glycoproteins in Antibodies
Structurally, an antibody is a Y-shaped monomeric unit built from four polypeptide chains: two identical heavy (H) chains and two identical light (L) chains, held together by disulfide bridges. The tips of the Y contain variable (V) regions that serve as specific antigen-binding sites, while the stem consists of constant (C) regions that define the antibody’s functional class. The glycoprotein nature of the molecule stems from N-linked glycosylation, where oligosaccharide chains are chemically bonded to the protein backbone. In the IgG isotype, a vital, conserved glycosylation site is located at the Asn-297 residue within the CH2 domain of the Fc (tail) region. These complex, biantennary carbohydrate structures are tucked between the heavy chains, playing a decisive role in maintaining the antibody’s three-dimensional shape and ensuring the correct orientation of the Fc region for immune signaling.
Role: The carbohydrate component of an antibody functions essentially as a molecular switch that dictates the immune system’s effector response. While the protein’s variable regions are responsible for recognizing and docking onto a pathogen, it is the glycan chains located on the Fc region that communicate the instruction to the rest of the immune system. Specifically, these sugars are indispensable for the antibody’s ability to bind to Fc receptors found on immune cells, such as macrophages and natural killer (NK) cells. These binding triggers critical defence mechanisms like phagocytosis (engulfing the pathogen) or Antibody-Dependent Cellular Cytotoxicity (ADCC).
Glycolipids: Cell Surface Markers and the ABO Blood Group System
Glycolipids serve as vital membrane components, consisting of a carbohydrate head group covalently linked to a lipid tail. Primarily situated in the outer leaflet of the plasma membrane, their sugar chains extend into the extracellular environment to facilitate critical recognition and signaling processes.
Structure and Function as Cell Surface Markers
At a structural level, a glycolipid is composed of a hydrophobic tail, usually a sphingosine or glycerol backbone attached to fatty acids that anchors the molecule within the cell membrane. Extending outward from this base is a polar oligosaccharide chain, which contributes to the glycocalyx, a sugary coating on the cell’s exterior.
https://en.wikipedia.org/wiki/Glycolipid
Functionally, these carbohydrate chains act as biological ID tags. Because the sequence and branching of these sugars are highly unique, they allow the immune system to identify self-cells versus foreign invaders. Additionally, these markers are essential for cell-cell adhesion, ensuring tissues remain cohesive, though they can unfortunately also serve as docking points for viruses and bacteria.
The ABO Blood Group System
A primary example of glycolipids acting as surface markers is the ABO blood group system. The antigens that characterize different blood types are actually specific sugar sequences attached to glycolipids on the surface of red blood cells. Every individual begins with a foundational precursor known as the H antigen.
https://www.creative-biolabs.com/anti-glycan-antibodies/glycosylation-influences-blood-type.htm
Blood type is determined by the activity of enzymes called glycosyltransferases, which attach a specific terminal sugar to this H-chain:
- Type A: The enzyme adds N-acetyl galactosamine.
- Type B: The enzyme adds D-galactose.
- Type AB: Both enzymes are active, resulting in a mixture of both A and B sugar chains.
- Type O: The enzyme is inactive, leaving the H antigen in its basic, unmodified state.
Proteoglycans: The Backbone of the Extracellular Matrix (ECM)
Proteoglycans function as the fundamental regulatory scaffold of the extracellular matrix (ECM), characterized by a complex architecture of core proteins modified by covalently linked glycosaminoglycan (GAG) side chains. These GAGs specifically chondroitin, keratan, and heparan sulfate exhibit high polyanionic density due to their sulfate and carboxyl constituents. This biochemical profile generates an osmotic gradient that sequesters water, creating a pressurized, visco-elastic hydrogel. This mechanism is physiologically vital for providing compressive resilience in load-bearing tissues, such as articular cartilage.
In addition to their mechanical utility, proteoglycans serve as biochemical hubs. By tethering growth factors and cytokines, they prevent premature proteolysis and modulate the kinetics of ligand-receptor interactions. This sequestration directly influences cellular trajectories, including migration and differentiation. Moreover, specialized subsets like small leucine-rich proteoglycans (SLRPs), such as decorin, are indispensable for structural homeostasis; they physically dock onto collagen fibrils to regulate their assembly and spatial orientation. The precise synthesis and distribution of these molecules are mandatory for preserving both the physical durability and the signaling equilibrium of the interstitial environment.
Biological Functions: Cell Signaling and Cell-Cell Recognition
Glycoconjugates, spanning the molecular spectrum of glycoproteins, glycolipids, and proteoglycans, constitute the primary biochemical interface for cellular recognition and the regulation of complex signal transduction. Centrally positioned on the extracellular leaflet of the plasma membrane, their carbohydrate residues integrate into the glycocalyx, a dense, information-rich matrix that encodes cellular identity. These glycan sequences function as high-affinity ligands for lectins, driving critical physiological events such as the tethering and rolling of leukocytes during inflammatory responses and the precise spatial patterning of cells during morphogenesis. glycoconjugates facilitate the discrimination between self and non-self and govern targeted cellular migration.
Functionally, glycoconjugates operate as regulators of transmembrane signaling rather than passive structural elements. Heparan sulfate proteoglycans (HSPGs), are essential co-receptors that sequester signaling ligands, such as Fibroblast Growth Factors (FGFs), and present them to cognate receptors. This ternary interaction is a thermodynamic prerequisite for receptor dimerization and the subsequent activation of intracellular phosphorylation cascades. Furthermore, the heterogeneous glycosylation of receptor proteins can sterically modulate ligand-binding kinetics, providing a mechanism for cells to fine-tune their sensitivity to environmental cues. Consequently, the structural integrity and diversity of glycoconjugates are indispensable for maintaining homeostatic signaling and the accuracy of intercellular communication.
The Sugar Code: How Lectins Decipher Glycan Information
Lectins are specialized non-enzymatic proteins defined by their highly selective carbohydrate-recognition domains (CRDs). Structurally, these proteins often exist as multimeric complexes, an architectural feature that facilitates the simultaneous binding of multiple sugar residues. This arrangement utilizes a network of hydrogen bonds and van der Waals interactions between specific amino acid side chains and the hydroxyl groups of the target glycan, ensuring high stereochemical specificity for distinct monosaccharide or oligosaccharide sequences.
The process of deciphering the sugar code involves the translation of complex glycan structural diversity into specific biological outcomes. Because individual glycan-lectin interactions are typically characterized by low affinity, the system relies on multivalency, or avidity. In this mechanism, the lectin acts as a molecular reader that recognizes clustered patterns of glycans on the cell surface. This spatial sensing allows lectins to distinguish subtle variations in glycan branching or terminal sugar modifications, effectively interpreting the glycan profile presented by the glycocalyx.
Once the sugar code is decoded through successful binding, the lectin triggers definitive cellular responses. This includes the orchestration of cell-cell adhesion, the initiation of transmembrane signalling cascades, and the sorting of glycoproteins during intracellular trafficking. By discriminating between these intricate glycan patterns, lectins convert chemical information into the physiological instructions necessary for immune surveillance, tissue development, and homeostatic regulation.
Clinical Significance: Glycoconjugates in Cancer Metastasis and Viral Entry
The clinical significance of glycoconjugates is underscored by their instrumental roles in oncogenic progression and viral pathogenesis. In the context of malignancy, the onset of aberrant glycosylation typically manifesting as hyper-branching of N-glycans and terminal sialylation constitutes a pathological molecular signature that drives metastasis. These structural alterations attenuate homotypic cell-cell adhesion and facilitate the hijacking of endogenous lectins, such as galectins, to promote tumour cell intravasation and the colonization of secondary niches. By altering the spatio-temporal regulation of the interstitial microenvironment, these glycoconjugates enable neoplastic cells to bypass immunological checkpoints and circumvent programmed cell death.
Mirroring these processes, the viral internalization pathways are dependent upon the structural integrity of the host’s glycocalyx. Pathogens, including the influenza virus and SARS-CoV-2, utilize specialized surface glycoproteins such as hemagglutinin or spike proteins to act as lectin-like probes that target specific sialic acid motifs or heparan sulfate sequences. This high-fidelity molecular recognition is a thermodynamic prerequisite for viral docking and the eventual fusion of the viral envelope with the plasma membrane. Given these dependencies, the biochemical modulation of glycan-lectin affinities has emerged as a critical therapeutic target, driving the development of glycan mimetics and competitive inhibitors aimed at arresting both metastatic dissemination and viral replication cycles.
Biotech Applications: Glycoengineering for Therapeutic Proteins
Glycoengineering serves as a highly precise method for refining therapeutic proteins by altering their carbohydrate profiles. Because the majority of biotherapeutics are glycoproteins, the specific arrangement of these sugars is a critical determinant of how the drug functions within the human body. By employing genetic tools to modify host cell pathways or utilizing enzymatic reactions to remodel glycans after production, researchers can significantly improve a protein’s pharmacokinetic profile. This includes enhancing metabolic stability, increasing solubility, and ensuring that the drug reaches its intended target without being prematurely cleared by the body.
Beyond basic stability, glycoengineering is a powerful tool for immunomodulation. Many non-human expression systems naturally attach sugar molecules that the human immune system recognizes as foreign, leading to potential adverse reactions. Through glycoengineering, these non-human signatures can be replaced with human-like glycan structures, drastically reducing immunogenicity. Furthermore, by optimizing the carbohydrate attachments on the protein backbone, researchers can amplify the drug’s ability to trigger specific immune responses. This level of molecular tailoring allows for the creation of superior biologics that are more potent, safer, and more effective for patient use.
Conclusion
The multifaceted landscape of glycoconjugates comprising glycoproteins, glycolipids, and proteoglycans functions as a sophisticated biological coding system indispensable for maintaining cellular homeostasis. These molecules establish the critical interface between the cell and its microenvironment, providing mechanical resilience within the extracellular matrix and defining antigenic identity, such as the ABO blood group system. The highly regulated mechanisms of N-linked and O-linked glycosylation are vital for ensuring protein folding fidelity and biological activity, while lectins act as molecular “readers” that translate these glycan patterns into coordinated immune signaling and cellular cascades.
The clinical implications of this glycomic framework are significant, as dysregulation within the glycoproteome is a primary driver of oncogenic metastasis and viral pathogenesis. However, the maturation of glycoengineering provides a high-precision methodology to recalibrate these pathways for therapeutic benefit. By strategically modifying carbohydrate profiles, biotechnological interventions can optimize the pharmacokinetic stability and reduce the immunogenicity of recombinant proteins. Ultimately, the ability to synthesize and manipulate these complex glycans is enabling the development of next-generation biologics that offer superior potency, safety, and targeted efficacy in the treatment of human disease.
References
- Glycosylation: Mechanisms, Biological Functions, and Clinical Implications Li, H., Gao, X., & Liu, N. (2024). Glycosylation: Mechanisms, biological functions and clinical implications. Signal Transduction and Targeted Therapy, 9(1), 1–32. https://doi.org/10.1038/s41392-024-01798-y
- Immunoglobulins (Antibodies) Justiz Vaillant, A. A., Jamal, Z., & Ramphul, K. (2023). Immunoglobulin. StatPearls Publishing. https://www.ncbi.nlm.nih.gov/books/NBK513460/
- ABO Blood Group Antigens Cooling, L. (2015). ABO blood group antigens and differential glycan expression: Perspective on the evolution of common human enzyme deficiencies. Human Immunology, 76(12), 934–946. https://doi.org/10.1016/j.humimm.2015.04.004
- Proteoglycans and the Extracellular Matrix Iozzo, R. V., & Schaefer, L. (2015). Proteoglycan form and function: A comprehensive nomenclature of proteoglycans. Matrix Biology, 42, 11–55. https://doi.org/10.1016/j.matbio.2015.02.003 (Reference for the provided PMC link: https://pmc.ncbi.nlm.nih.gov/articles/PMC7258702/)
- Lectins: Translators of the Sugar Code Ricci, D. G., Lanza, M., Atri, G. A., & de Oliveira, L. L. (2023). Lectins: Getting familiar with translators of the sugar code. International Journal of Molecular Sciences, 24(11), 9333. https://doi.org/10.3390/ijms24119333
- Metabolic Glycoengineering Applications Du, J., Meledeo, M. A., Wang, Z., Khanna, H. S., Paruchuri, V. D., & Yarema, K. J. (2009). Metabolic glycoengineering: Sialic acid and beyond. Glycobiology, 19(12), 1382–1401. https://doi.org/10.1093/glycob/cwp115 (Reference for the provided PMC link regarding Applications of Metabolic Glycoengineering)
- Glycoconjugates: Synthesis and Therapeutic Developments Frenkel-Pinter, M., Shmueli, M. D., Belakhov, V., Baasov, T., Segal, D., & Gazit, E. (2017). Glycoconjugates: Synthesis, functional studies, and therapeutic developments. Chemical Reviews, 117(15), 10349–10389. https://doi.org/10.1021/acs.chemrev.7b00104
- Glycan-Lectin Interactions in Cancer and Viral Infections Serna-Vargas, J. P., Magadán-Corpas, P., & Garcia-Vallejo, J. J. (2023). Glycan–Lectin interactions in cancer and viral infections and how to disrupt them. International Journal of Molecular Sciences, 24(21), 15820. https://doi.org/10.3390/ijms242115820
