Gel Permeation Chromatography (GPC) is a liquid chromatography method used to separate and analyze molecules based on their hydrodynamic volume or size in solution. It is used to determine the molecular weight distribution and average molecular weight values of polymers.
GPC is a type of size-exclusion chromatography (SEC). The concept of SEC was first introduced in 1955 by Lathe and Ruthven, who demonstrated the separation of molecules according to size. GPC was later developed in the early 1960s by John C. Moore, who introduced the use of cross-linked polystyrene gels to separate polymers in organic solvents. This method was later commercialized by Jim Waters through Waters Corporation, leading to its widespread use in the polymer industry and research.
Principle of Gel Permeation Chromatography
GPC works on the principle of size exclusion, where molecules are separated by their size in solution. This separation occurs in a column packed with porous gel particles as a stationary phase that acts as a molecular sieve.
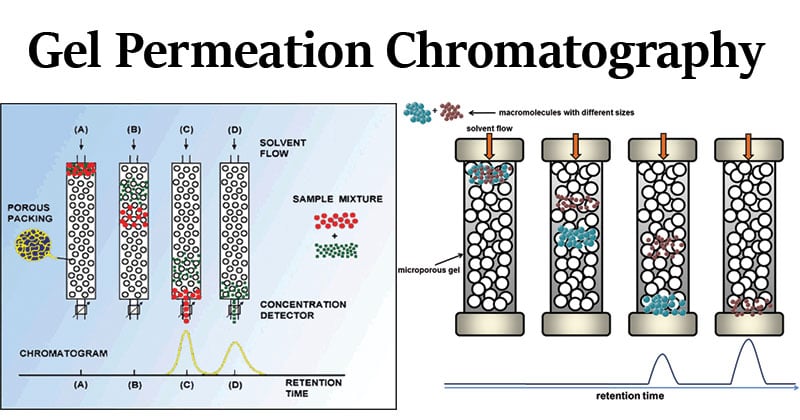
The sample is first dissolved in a suitable solvent and injected into a flowing mobile phase that passes through the column. The sample mixture contains molecules of different sizes, which interact differently with the porous stationary phase. The size of the molecule controls the separation. Larger molecules cannot enter the pores of the stationary phase, so they move faster to elute first. Smaller molecules can enter the pores, so they take a longer time to elute from the column. The time taken by a molecule to elute from the column is called retention time. So, the retention time of each molecule is related to its molecular size.
The eluted molecules are detected using a suitable detector, and the detector response is recorded as a plot called a chromatogram. This plot is compared with a calibration curve obtained from standard polymers and is used to determine the molecular weight distribution of the sample. Molecular weight distribution describes how polymer chains of different molecular weights are distributed within a sample, and it is an important parameter to understand the properties of polymers.
Components of Gel Permeation Chromatography
Pump: The pump system delivers the mobile phase and sample through the GPC system at a constant flow rate. High-pressure pumps are used to maintain uniform flow through the column to avoid pressure fluctuations.
Injector: The injector introduces the sample into the flowing solvent stream without disrupting the continuous flow. Loop injectors are often used. Autosamplers are also widely used. Autosamplers load samples automatically to the injection valve and improve the speed when analyzing many samples.
Column: The column is the main separation component that is packed with the stationary phase. Columns with different lengths and diameters are available for use. Many GPC systems have column ovens to maintain a controlled temperature. This is important for solvents with higher viscosities and samples that are insoluble at lower temperatures.
Stationary Phase: The stationary phase contains semi-permeable and porous polymer gel beads made from cross-linked polymer or inorganic materials that have defined pore sizes. Large molecules stay outside the pores, while smaller ones can enter some or all of them. Commonly used stationary phases are polystyrene-divinylbenzene, silica gel, dextran, agarose gel, and acrylamide gel.
Mobile Phase: The mobile phase is the solvent used to dissolve the sample and move it through the column. The solvent must completely dissolve the sample. Organic solvents like tetrahydrofuran (THF), trichlorobenzene (TCB), toluene, chloroform, and dimethylformamide (DMF) are commonly used for the analysis of synthetic polymers. Additives can be used to stabilize the solution.
Calibration kits: They contain polymer standards with known molecular weights and narrow molecular weight distributions. These standards are used to create calibration curves that show the relation between retention time or elution volume and molecular weight. Some of the commonly used standards are polystyrene, polymethyl methacrylate (PMMA), and polyethylene glycol or oxide (PEG/PEO).
Detectors: They detect the components eluting from the column and convert the response into an electrical signal. Different detectors use different characteristics of molecules to detect their presence. The refractive index (RI) detector is the most commonly used detector in GPC as it responds to most polymers. Other detectors include UV detectors, evaporative light scattering detectors (ELSD), multi-angle light scattering detectors (MALS), and viscometers.
Data System: The data system processes and records the detector signals to generate a chromatogram. It uses the calibration curve and the chromatogram plot to produce molecular weight distribution curves, which are used to calculate average molecular weight values. Modern GPC software supports multi-detector analysis, where signals from multiple detectors are combined to obtain accurate molecular information about the sample.
Procedure or Steps of Gel Permeation Chromatography
System and Column Preparation
- All components of the system, including the pump, injector, column, and detectors, are checked for proper operation.
- A suitable column is selected based on the required molecular weight range and separation resolution.
- The column is washed and equilibrated with the mobile phase. The mobile phase and the solvent used in sample preparation must be the same.
Sample Preparation
- Sample preparation is an important step in GPC as poorly prepared samples can cause incomplete dissolution, clogged columns, and inaccurate molecular weight data.
- A well-prepared sample should be fully soluble, free of particulates, and compatible with the mobile phase.
- At first, the sample is fully dissolved in a suitable solvent.
- The solution is then filtered through a syringe filter to remove insoluble impurities or aggregates that could clog the column.
- The solution is also degassed to prevent bubble formation, which can interfere with detection.
Calibration
- GPC does not directly measure molecular weight, so calibration is needed to convert chromatographic data into molecular weight information.
- GPC uses standards with known molecular weights to calibrate the system.
- Calibration is done by injecting polymer standards under the same chromatographic conditions used for sample analysis.
- A calibration curve is constructed by plotting log molecular weight versus retention time or elution volume.
- This curve is stored in the data system and used during analysis to calculate molecular weight distributions and averages.
- Calibration must be repeated when columns, solvents, or operating conditions are changed.
Sample Injection
- A fixed volume of the prepared sample is injected into the flowing mobile phase, which carries the sample into the GPC column.
- Suitable injection volume must be used to avoid overloading.
Separation
- After sample injection, the sample moves through the column and interacts with the porous stationary phase inside the column based on hydrodynamic volume.
- Smaller molecules enter the pores and take longer paths to elute later, while larger molecules are excluded from the pores and elute faster through the column.
Detection
- After separation, the molecules pass through the detector that measures the amount of material eluting from the column and converts it into electrical signals.
- GPC systems use detectors including RI, UV absorbance, ELSD, light scattering detectors, and viscometers.
- Multiple detectors can also be used in the same GPC instrument, which provides more accurate and detailed information.
Data Analysis
- The detector signals are recorded as a chromatogram by the data system.
- A chromatogram is a plot of detector response versus time or volume, where each peak represents molecules within a specific size range.
- The chromatogram data is compared to the calibration curve.
- This comparison is used to determine the molecular weight distribution and averages like number-average molecular weight (Mn), weight-average molecular weight (Mw), Z-average molecular weight (Mz), and polydispersity index (PDI).
- These are important parameters that affect the physical properties of polymers, such as tensile strength, elasticity, brittleness, melt viscosity, impact strength, and adhesive properties.
Factors Affecting Gel Permeation Chromatography
- The selection of the stationary phase material and its pore size is an important factor in GPC. The gel must have a suitable pore size distribution that matches the molecular weight range of the sample. Specific molecular weight ranges need specific pore ranges.
- Separation is also affected by the properties of the mobile phase. It must completely dissolve the polymer and minimize interactions with the stationary phase.
- The sample concentration and injection volume are also important factors to consider. It is important to optimize sample concentration and injection volume for accurate analysis. High sample concentration and large sample volumes can overload the column and cause peak broadening.
- Column conditions, including column design, flow rate, temperature, and pressure parameters, also affect the separation process.
Common Products and Manufacturers of Gel Permeation Chromatography
| Common Products | Manufacturers |
| Styragel columns, HSPgel Columns, Envirogel GPC Cleanup Columns, Shodex Columns, Ultrastyragel Preparative Columns, Ultrahydrogel Columns, 2414 Refractive Index (RI) Detector, DAWN MALS instrument | Waters Corporations |
| 1260 Infinity II GPC/SEC System, 1260 Infinity II Multidetector GPC/SEC System, 1290 Infinity II GPC/SEC System, GPC/SEC Software for OpenLab CDS | Agilent |
| Nexera Gel Permeation Chromatography System | Shimadzu |
| GPC-IR, GPC-QC | Polymer Char |
| EcoSEC High Temperature GPC System, EcoSEC Elite GPC System, TSKgel GPC Columns | Tosoh Corporation |

Applications of Gel Permeation Chromatography
- GPC is used to determine molecular weight averages and the overall molecular weight distribution of polymers. These are important parameters for polymer characterization.
- It has applications in quality control and product development in the polymer industry. It is used to monitor polymerization reactions and check stability.
- It is also used in pharmaceutical and biomedical research to study drug delivery systems like polymeric nanoparticles or microspheres.
- It has applications in the food industry to analyze polysaccharides and food gums to understand their structure and effect on texture and consistency.
- It is also used in environmental studies to analyze macromolecular components like humic substances or dissolved organic matter in water samples.
Advantages of Gel Permeation Chromatography
- GPC is a non-destructive method, as the separation occurs by size rather than chemical interaction.
- It provides accurate molecular weight data, which is important for both industry and research applications.
- It can separate different molecular weight fractions in complex mixtures such as polymers, monomers, oligomers, and additives.
- It can be combined with multiple detectors, like light scattering and viscometers, for more detailed analysis.
- It has applications in both small-scale identification and large-scale purification.
Limitations of Gel Permeation Chromatography
- GPC has a lower resolution compared to some other chromatographic methods. It is less effective for polymers with very similar molecular sizes, as it can only accurately separate peaks that differ by at least 10% in molecular weight.
- The accuracy of molecular weight data depends on proper calibration using standards.
- A calibration curve must be made for each polymer and solvent system, which can be repetitive and time-consuming.
- Samples must be fully dissolved and filtered before analysis. Some polymers need specific solvents or conditions to dissolve completely. So, it has high sample preparation requirements.
Gel Permeation Chromatography Troubleshooting and Safety Considerations
- Peak tailing can occur due to excessive dead volume, column degradation or unwanted interactions. Reducing tubing length, replacing or repairing columns, and using mobile phase additives can minimize this problem.
- Resolution problems can occur when the wrong column pore size is used. So, columns with a correct column pore size that matches the size distribution of the sample should be used.
- Peak broadening occurs due to large dead volume, high solvent viscosity, or large detector cell volume. This can be minimized by reducing injection volume, using a column oven for viscous solvents, and using a smaller detector cell volume if possible.
- Peak splitting occurs when the column is overloaded or due to a blocked frit or injection valve. This can be resolved by reducing sample concentration or injection volume, replacing blocked frits or column, or using inline filters to protect the column.
- Ghost peaks are caused by carryover from previous injection or sample adsorption. This can be avoided by allowing the previous sample to fully elute and adding buffer or changing solvent to reduce adsorption.
- Changes in retention time can occur due to unstable flow rates, changes in solvent composition, or interaction with packing. Solvent should be degassed to avoid bubble formation, and modifiers or additives can be used.
- Inconsistent molecular weight can result from column aging, pump drift, or new fittings. Regular recalibration using appropriate standards, routine system performance checks, and column replacement are necessary to ensure accurate molecular weight determination.
- Baseline drift or excessive noise can occur from column or detector contamination, bubbles in the detector, and temperature fluctuations. This can be resolved by flushing the column or detector to waste, using high-quality solvents and degassing them, and using a column heater or insulating tubing to maintain stable temperature conditions.
- Many GPC solvents are volatile, flammable, or toxic. They should be handled in a fume hood with personal protective equipment.
- High-pressure systems can cause leakage, so all fittings and connections should be properly tightened and regularly inspected.
- Waste solvents must be disposed of according to laboratory safety regulations. Solvents should be clearly labeled and disposed of.
Gel Permeation Chromatography Recent Advances and Innovations
- High-performance columns have been developed that provide higher resolution and better separation of polymers.
- Advances in column packing materials and particle design have improved separation performance.
- Detector technologies have also developed. Advanced detectors like multi-angle light scattering (MALS) and refractive index (RI) provide more accurate molecular weight information.
- Multi-detector systems have been developed that allow accurate and detailed characterization of polymers and macromolecules.
- High-temperature GPC instruments and columns are also used, which allow the analysis of polymers that only dissolve at high temperatures.
- Modern data analysis software and algorithms can process large amounts of data and combine signals from different detectors to perform accurate calculations.
Conclusion
Gel Permeation Chromatography (GPC) is an important method used for the separation and analysis of polymers. Unlike other chromatography methods that separate molecules based on chemical reactions, GPC separates molecules according to their size. This provides accurate information about molecular weight distribution, which is important for understanding polymer properties useful in industry and research applications.
Continuous improvements in column technology, detection systems, and data analysis have expanded the applications of GPC beyond polymer analysis. It is now widely used in the characterization of macromolecules and complex mixtures in pharmaceuticals, food science, and environmental research.
References
- Agilent Technologies. (2015, April 30). An Introduction to Gel Permeation Chromatography and Size Exclusion Chromatography (5990-6969EN). https://www.agilent.com/cs/library/primers/Public/5990-6969EN%20GPC%20SEC%20Chrom%20Guide.pdf
- Agilent Technologies. (2019, December 15). GPC/SEC Troubleshooting Guide (5994-1573EN). https://www.agilent.com/cs/library/posters/public/poster-GPC-Troubleshooting-Guide-5994-1573EN-agilent.pdf
- Beginner’s Guide to Size-Exclusion Chromatography (SEC). (n.d.). Waters. https://www.waters.com/nextgen/us/en/education/primers/beginners-guide-to-size-exclusion-chromatography.html
- GPC Systems. (n.d.). Tosoh Bioscience. https://www.separations.eu.tosohbioscience.com/products/chromatography-instruments/gpc-systems
- Measurement of Molecular Weight by using GPC method. (n.d.). https://www.shimadzu.com.sg/an/service-support/technical-support/liquide-chromatography/required_tools/gpc.html?utm
- Seng, C. (2025, June 28). Master Gel Permeation Chromatography of Polymers: setup and techniques. Jmscience B. https://blog.jmscience.com/master-gel-permeation-chromatography-of-polymers-setup-and-techniques/
- Understanding gel permeation chromatography techniques. (n.d.). Chrom Tech, Inc. https://chromtech.com/blog/understanding-gel-permeation-chromatography-techniques/
- Understanding Gel Permeation Chromatography: A Comprehensive Guide. (n.d.). TCA Lab / Alfa Chemistry. https://tcalab.alfa-chemistry.com/understanding-gel-permeation-chromatography-a-comprehensive-guide.html
- Waters Corporation. (2014). Gel permeation chromatography (GPC). https://www.waters.com/content/dam/waters/en/library/brochures/2014/waters-brochure-GelPermeationChromatography-720004331.pdf
