- The plasma membrane, also known as the cell surface membrane or plasmalemma, defines the boundary of the cell.
- They are a special type of membranes which are lipid structures that separate the cell from its environment.
- In composition, it is a phospholipid bilayer with embedded proteins that enclosing every living cell.
- It serves some specific functions such as controlling the flow of nutrients and ions into and out of the cells, mediating the response of a cell to external stimuli (a process called signal transduction), and interacting with bordering cells.
The Fluid Mosaic Model of Biomembranes
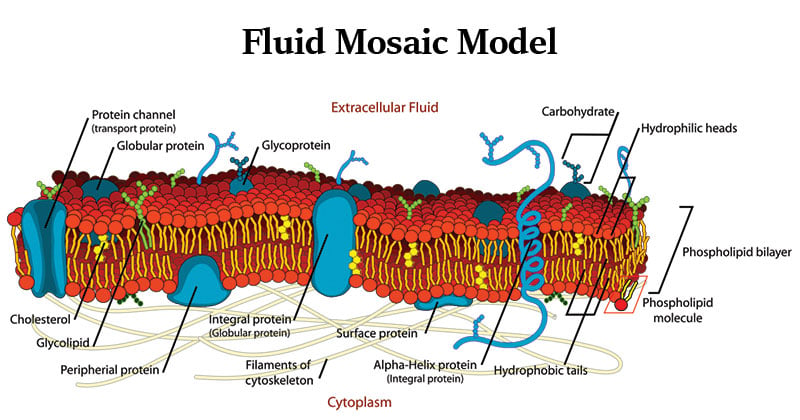
- The fluid mosaic model describes the structure of the plasma membrane as a mosaic of components —including phospholipids, cholesterol, proteins, and carbohydrates—that gives the membrane a fluid character.
- Membranes are impermeable to most polar or charged solutes, but permeable to nonpolar compounds; they are 5 to 8 nm (50 to 80 Å) thick and appear trilaminar when viewed in cross-section with the electron microscope.
- The combined evidence from electron microscopy and studies of chemical composition, as well as physical studies of permeability and the motion of individual protein and lipid molecules within membranes, led to the development of the fluid mosaic model for the structure of biological membranes.
- A bilayer of phospholipids about 3 nm thick provides the basic architecture of all cellular membranes with membrane proteins giving each cellular membrane its unique set of functions.
- Thus the plasma membrane consists of a lipid bilayer containing embedded and peripheral proteins. The major component of membranes is, however, lipids.
- Individual phospholipids can move laterally and spin within the plane of the membrane, giving the membrane a fluid-like consistency similar to that of olive oil.
- Membrane lipids are strongly amphipathic molecules with a polar hydrophilic “head group” and a polar hydrophobic “tail.”
- The polar head group attached to two hydrophobic fatty acid tails; the head group faces the aqueous environment, the fatty acid tails the interior of the bilayer.
Figure: Fluid mosaic model of a cell membrane, Source: Wikipedia.
- In membranes, they are primarily held together by the hydrophobic effect and weak Van der Waals forces and are therefore mobile relative to each other. This gives membranes a more or less fluid quality.
- Non- covalent interactions between phospholipids, and between phospholipids and proteins, also lend strength and resilience to the membrane.
- At the same time, the hydrophobic core of the bilayer prevents the unassisted movement of water-soluble substances from one side to the other.
- Integral membrane proteins (transmembrane proteins) span the bilayer and often form dimers and higher-order oligomers.
- Lipid-anchored proteins are tethered to one leaflet by a covalently attached hydrocarbon chain.
- Peripheral proteins associate with the membrane primarily by specific non- covalent interactions with integral membrane proteins or membrane lipids.
- Proteins in the plasma membrane also make extensive contact with the cytoskeleton.
- Lipids and proteins are mobile within the membrane. If they are not fixed in place by special mechanisms, they float within the lipid layer as if in a two-dimensional liquid; biological membranes are therefore also described as being a “fluid mosaic”.
- The carbohydrate moieties attached to some proteins and lipids of the plasma membrane are exposed on the extracellular surface of the membrane.
References
- Lodish, H. F., Berk, A., Kaiser, C., Krieger, M., Scott, M. P., Bretscher, A., Ploegh, H. L., Matsudaira, P. T. (2008). Molecular cell biology. New York: W.H. Freeman.
- Smith, C. M., Marks, A. D., Lieberman, M. A., Marks, D. B., & Marks, D. B. (2005). Marks’ basic medical biochemistry: A clinical approach. Philadelphia: Lippincott Williams & Wilkins.
- Koolman, J., & Röhm, K.-H. (2005). Color atlas of biochemistry. Stuttgart: Thieme.
- Alberts, B. (2004). Essential cell biology. New York, NY: Garland Science Pub.
- https://www.mheducation.co.uk/he/chapters/9780071102087.pdf
- http://www.nslc.wustl.edu/courses/bio101/cruz/Organelles/Organelle.htm

How can i seperate its properties from its functions and structure