Introduction
Epsilometer test (E- test) is defined as the “exponential gradient” method that determines the antimicrobial resistance of the microorganism. It is a cost-effective tool that has been developed to provide a direct quantification of antimicrobial susceptibility of microorganisms. It is a quantitative method that applies both the dilution of antibiotics and the diffusion of antibiotics into the medium. More precisely, the test combines a disk diffusion pattern with the determination of Minimum Inhibitory Concentration (MIC) which is considered as the lowest concentration of an antimicrobial that inhibits the visible growth of a microorganism. The E-test uses rectangular plastic strips with the predefined, continuous and exponential gradient of antibiotic concentration (one side of the strip contains the antimicrobial agent concentration gradient and the other side of the strip contains a numeric scale that indicates the drug concentration).
Objectives
- To determine the MIC of fastidious, slow-growing or nutritionally deficient micro-organisms.
- To detect and confirm low levels of resistance of a specific antimicrobial phenotype resistance.
- To determine and demonstrate the MIC value of the given antibiotic to given micro-organism.
Principle of E test
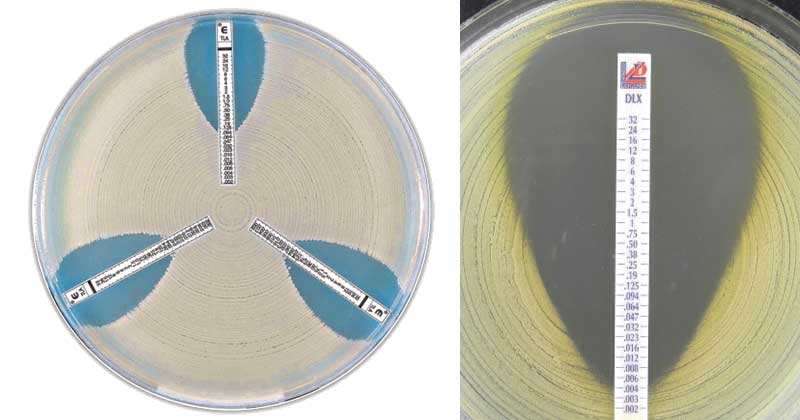
When the culture of the bacterial suspension is done on the Mueller Hinton Agar (MHA) using the sterile cotton swab. The e test strip is then placed on to an inoculated agar plate. After placing the strip on the agar surface, there is an immediate release of antibiotics from the plastic carrier surface into the agar surface. After incubation, bacterial growth becomes visible on the plate, and symmetrical inhibition ellipse along the strip is seen. The MIC value is read from the scale in terms of µg/ml where the ellipse edge intersects the strip as seen on the plate.
Procedure
- Inoculum Preparation
- Remove the E-test package from the freezer (-20°C) at least 30 minutes before required.
- Emulsify 3 or 4 individual test strain colonies and transfer to a tube of saline.
- Compare turbidity to that in the 0.5 McFarland standards. Adjust turbidity of inoculum in order to match the standard.
- Inoculation in Muller Hinton Agar
- Dip a sterile cotton swab into the inoculums and rotate the swab against the inside of the tube above the fluid level to remove excess liquid before swabbing.
- Streak the swab over the entire surface of the agar plate by rotating the plate approximately 60o.
- Complete inoculation by running the swab around the rim of the agar.
- Leave the lid of the plate agar for 5 minutes to allow any excess moisture to be absorbed before applying strips.
- Application of E-test strips:
- Open the E-test package and take a strip out using sterile forceps.
- Apply strips to the agar surface using forceps.
- Place the strip with the ‘E end’ at the edge of the plate and with the scale visible (i.e. facing upwards).
- Incubate Plates at 37°C for 18-24 hrs.
Result and Interpretation
Image Source: ETEST® Telavancin and Rapid Test Methods Ltd
- After the incubation, the tests are read by viewing the strips from the top of the plate, a symmetrical inhibition ellipse is produced and seen on the plate.
- Read MIC at the intersection of the lower part of the ellipse-shaped growth inhibition area with the test strip. If a MIC value is between twofold dilutions, always round up to the highest value.
- Read the MIC value at complete inhibition of all growth.
- MIC values of the bacteria should be interpreted as S (Susceptible), I (Intermediate) or R (Resistant) by comparing the breakpoint values of each antibiotic with the criteria recommended and given by CLSI guidelines.
Precautions
- Aseptic procedures and precautions against microbiological hazards should be used when handling bacterial specimens.
- E-test should be used strictly according to the procedures described.
- One should be very careful while placing the E strip on the plate containing the bacterial suspension to not touching the edge of the plate.
- Read results only if a good inhibition ellipse is visible.
- While doing or placing multiple strips on a single plate, the strips shouldn’t touch one another that would make the difference in the result interpretation of the given antibiotic.
Advantages of the E test
- It is very easy to perform, requires minimal training for test performance, and easy to execute.
- Contamination can be easily recognized.
- It is a less time-consuming and very convenient method of the determination of the Minimum Inhibitory Concentration (MIC) and applicable to a wide array of drugs and microorganisms.
- It is useful to detect some phenotypes resistance.
- It is an adequate method to detect potentially resistant strains to Amphotericin B.
- It helps to confirm or detect low-level or new resistance mechanisms.
- It can be used to investigate any synergistic potential of combination therapies.
Limitation of E test
- E-test is not suitable for Cryptococcus neoformans.
- For the usage of more than one antibiotic at a single time, it is imperative to know their interactions with each other and hard to know if they have an additive, synergistic, or antagonistic effect.
References
- Tille P.M (2014). Bailey and Scott’s diagnostic microbiology. Thirteen editions. Mosby, Inc., an affiliate of Elsevier Inc. 3251 Riverport Lane. St. Louis. Missouri 63043
- Biomerieux
- Joyce LF, Downes J, Stockman K, Andrew JH (1992). Comparison of five methods, including the PDM Epsilometer test (E test), for antimicrobial susceptibility testing of Pseudomonas aeruginosa. J Clin Microbiol. 30(10):2709–2713.
- https://www.jove.com/science-education/10512/antibiotic-susceptibility-testing-epsilometer-tests-to-determine-mic
- Sader HS, Pignatari ACC (1994). E Test: a novel technique for antimicrobial susceptibility testing. Sao Paulo Med. J. vol.112 no.4. Retrieved from: http://www.scielo.br/scielo.php?script=sci_arttext&pid=S1516-31801994000400003
Internet Sources
- 13% – https://microbeonline.com/e-test-epsilometer-test-principle-purpose-procedure-results-and-interpretations/
- 9% – https://microbiologynotes.com/e-epsilometer-test-principle-purpose-procedure-result-interpretation-with-precaution-advantages-and-disadvantages/
- 3% – https://pinardsflorist.com/epsilometer-test-94/
- 2% – https://www.ncbi.nlm.nih.gov/pubmed/11420333
- 2% – http://legacy.bd.com/ds/technicalCenter/inserts/L007393(11)(0706).pdf
- 1% – https://www.ncbi.nlm.nih.gov/pubmed/1324301
- 1% – https://www.dermnetnz.org/topics/laboratory-tests-for-bacterial-infections/
- 1% – https://www.biomerieux-diagnostics.com/etestr
- 1% – https://file.scirp.org/pdf/AiM_2013053114403142.pdf
- 1% – https://fas.org/irp/doddir/army/fm8-9.pdf
- <1% – http://www.chem.ucla.edu/~bacher/General/30BL/tips/TLC1.html

Relay ,thanks a lot it was very helpful and easy to understand.
Thanks a lot it was very helpful and easy to understand