Ebola virus (EBOV) is a negative-sense, enveloped, filamentous RNA virus that causes Ebola virus disease (EVD), a severe, zoonotic hemorrhagic disease with high case fatality rates.

Subsequent studies post-20132016 West African outbreak have elucidated its taxonomy, structure, replication, pathogenesis, and resulted in specific vaccines and monoclonal antibody treatments.
Taxonomy and Classification of the Ebola Virus
- Family / Order
-Family: Filoviridae; Order: Mononegavirales.
- Genus and species
-Genus: Ebolavirus (also called Orthoebolavirus in updated taxonomy).
-Six recognized ebolaviruses: Zaire ebolavirus (EBOV), Sudan virus (SUDV), Bundibugyo virus (BDBV), Taï Forest virus (TAFV), Reston virus (RESTV), Bombali virus (BOMV); human outbreaks largely due to EBOV, SUDV, BDBV.
- Biosafety and threat classification
-Category A biothreat agent; handled in BSL-4 facilities.
Structure and Morphology of the Ebola Virus
- Virion shape and size
-Filamentous, thread-like particles; diameter ~80–100 nm; length typically <1 μm but can extend to >20 μm, with extreme length polymorphism and polyploid particles containing up to ~22 genome copies.
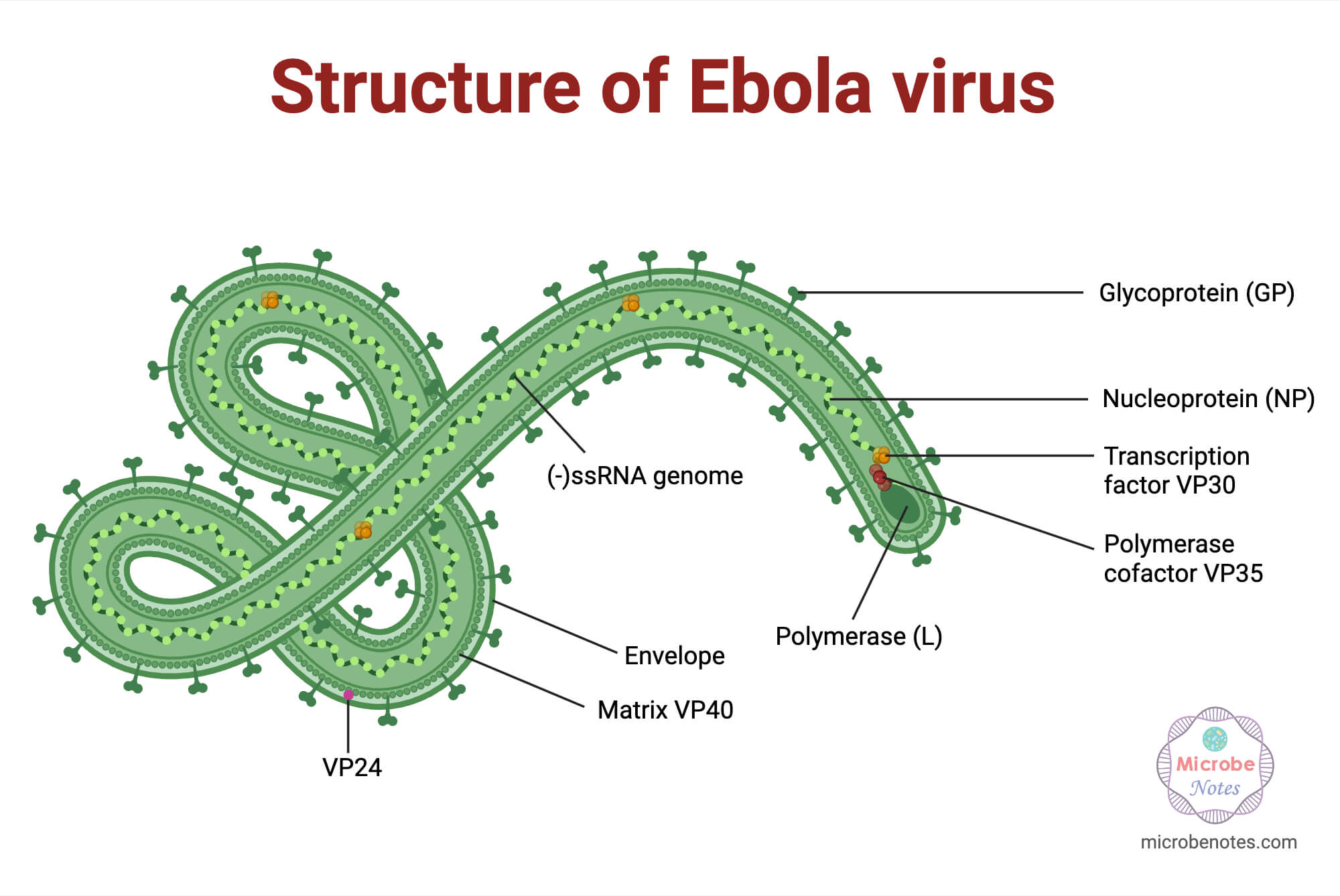
- Envelope and surface
-Lipid envelope of cellular origin with glycoprotein (GP) peplomers (~7–10 nm spikes) embedded in the bilayer.
- Internal organization
-Central, helically arranged nucleocapsid (NC): negative-sense RNA associated with NP, VP35, VP30, and L.
-Matrix layer between envelope and NC contains VP40 and VP24.
- Defective/empty particles
-Empty virions lacking a genome and small-diameter particles have been reported, likely impacting immunogenicity and particle heterogeneity.
Table 1: Structural Components and Locations
| Component | Location / Role |
| GP | Envelope spikes; receptor binding, fusion, tropism |
| VP40 | Matrix: budding, virion assembly |
| VP24 | Matrix/inner envelope; assembly, IFN antagonism |
| NP | Nucleocapsid; RNA encapsidation |
| VP35 | Polymerase cofactor; IFN antagonism |
| VP30 | Transcription activator |
| L | RNA-dependent RNA polymerase |
Genome Organization and Proteins of the Ebola Virus
- Genome
-Non-segmented, linear, negative-sense RNA ~18–19 kb.
-3′ leader and 5′ trailer regions contain cis-acting signals for transcription/replication; 5′ trailer and L gene are characterized as critical for polymerase function.
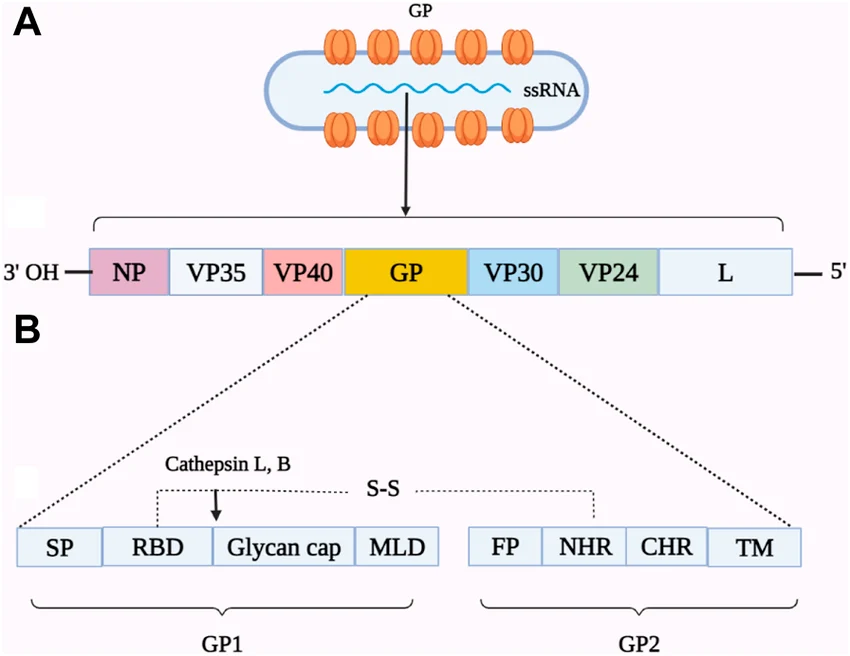
- Gene order (3′→5′)
-NP – VP35 – VP40 – GP – VP30 – VP24 – L.
- Major proteins and selected functions
-NP: encapsidates RNA; nucleocapsid formation; required for replication/transcription.
-VP35: polymerase cofactor; essential for replication/transcription; potent type I and III IFN antagonist via sensing and signaling blockade.
-VP40: primary matrix protein; drives virion assembly and budding; determines particle morphology.
-GP: mediates attachment, receptor binding (e.g., NPC1), macropinocytosis, and membrane fusion; main target of neutralizing antibodies and licensed monoclonals.
-VP30: transcription activator; phosphorylation state at Ser29 regulates switch between transcription and replication.
-VP24: secondary matrix protein; contributes to assembly; blocks STAT1 nuclear import, inhibiting IFN signaling.
-L: RNA-dependent RNA polymerase; catalyzes transcription and replication.
- Epitranscriptomic features
-m⁶A modifications in the 3′ leader region antagonize ISG20 exonuclease activity via YTHDF1 recruitment, enhancing replication and helping evade innate immunity.
Replication Cycle of the Ebola Virus
Genome and Core Proteins
- Non-segmented, negative-sense RNA (~19 kb) encoding NP, VP35, VP40, GP, VP30, VP24, L.
- NP encapsidates the genome, forming the nucleocapsid that serves as a template for transcription/replication.
- L polymerase with cofactor VP35 carries out RNA synthesis; VP30 is a key transcription activator whose phosphorylation state controls the switch between transcription and replication.
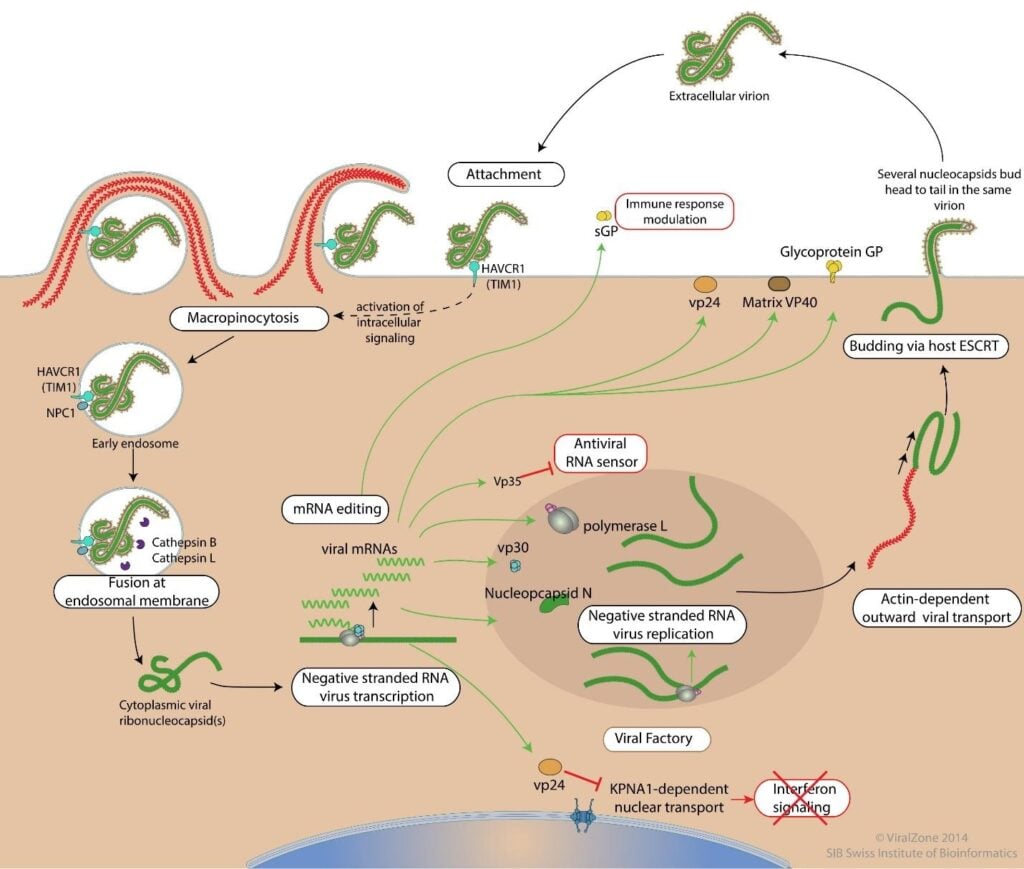
Entry and Uncoating
- Attachment: GP1,2 binds cell-surface carbohydrate receptors and phosphatidylserine (PS) receptors; PS in the viral envelope engages PS receptors (e.g., AXL).
- Internalization: Macropinocytosis, followed by trafficking through acidic endosomes.
- In endolysosomes, GP is cleaved by cathepsins; cleaved GP1 binds NPC1, and GP2 mediates fusion to release nucleocapsid into the cytosol.
Transcription and Replication
- Primary transcription in the cytoplasm produces viral mRNAs from the 3′ leader promoter, using NP-encapsidated genome as a template.
- VP30 promotes transcription; phosphorylation of VP30 (notably at Ser29) weakens VP30–VP35 interaction, reducing transcription and shifting the balance toward replication.
- Inclusion bodies (VIBs/IBs) formed by NP and other proteins act as secondary sites for transcription and replication.
- High-resolution structures show the 3′ leader RNA adopts a bent conformation in the L–VP35–RNA complex, essential for de novo replication initiation.
Regulation by Viral and Host Factors
- VP35 acts as a polymerase cofactor and binds AKIP1, activating PKA–CREB1; CREB1 is recruited into VIBs and supports replication.
- VP35 also binds unanchored K63-linked polyubiquitin chains; this interaction promotes polymerase activity, and small molecules disrupting VP35–Ub reduce EBOV replication in cells and mice.
- Host RNA-binding protein RBM4 directly binds the 3′ leader “CU-rich” elements, suppresses viral mRNA production, and upregulates antiviral cytokines, thereby inhibiting replication.
- EBOV GP is heavily O-glycosylated in its mucin-like domain; specific GalNAc-transferases (especially GalNAc-T1) control subsets of O-glycosylation sites. Disrupting O-glycan initiation/elongation reduces viral particle production and progeny titers.
Assembly and Budding
- As genome replication increases, nucleocapsids assemble and are transported to the plasma membrane.
- VP40, the major matrix protein, drives budding; GP in the envelope and fully assembled nucleocapsid form filamentous virions, which can be polyploid, containing multiple genome copies end-to-end.
- Empty, genome-negative particles are also produced.
Pathogenesis and Host Immune Response to the Ebola Virus
Cell and Tissue Tropism
- Early infection targets macrophages and dendritic cells; EBOV has broad tropism except for lymphocytes.
- Recent work shows susceptibility of adipocytes, potentially adding a pro-inflammatory reservoir.
- Skin is an important target: viral RNA and infectious virions accumulate in dermal stromal and immune cells and around hair follicles; infection is patchy, with high local viral loads but minimal local inflammation.
- Primary vaginal epithelium models show robust infection, disruption of the epithelial barrier, and virus penetration to the lamina propria, with apical shedding over at least 7 days.
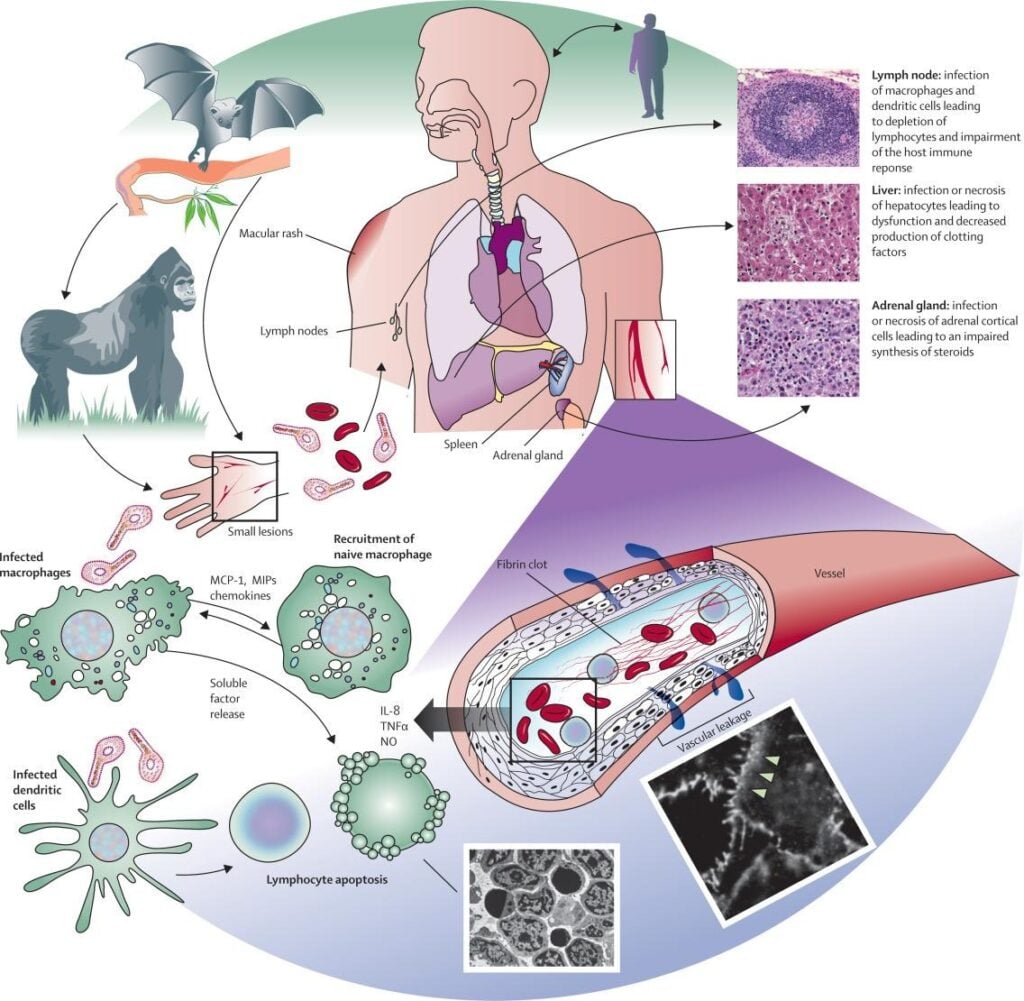
Innate Immune Evasion and Dysregulation
- VP35 and VP24 (and other proteins) block type I interferon signaling, enabling uncontrolled viral replication.
- Single-cell profiling in non-human primates shows EBOV-infected monocytes downregulate STAT1 mRNA and interferon-stimulated genes, while upregulating pro-viral host factors (e.g., DYNLL1, HSPA5).
- VP35-PKA-CREB1 activation also upregulates coagulation-related genes (e.g., THBD, SERPINB2), linking replication to hemorrhagic manifestations.
Systemic Immune and Cell-Death Responses
- A cytokine storm with high pro-inflammatory cytokines damages immune and parenchymal tissues; EBOV infects the spleen, liver, kidneys, and lungs, contributing to multi-organ failure and vascular leak.
- Single-cell data show replacement of normal monocyte subsets by immature, proliferative monocytes with reduced antigen presentation; lymphocytes upregulate apoptosis genes and decline in abundance.
- Molecular analyses highlight EBOV-induced cell death pathways (apoptosis, possibly autophagy modulation) as central to tissue injury and suggest that targeting cell-death pathways could be therapeutic.
Humoral and Cellular Immunity
- EBOV GP uses epitope masking and secretion of truncated/soluble GP forms to subvert neutralizing antibody responses.
- Survivors develop virus-specific antibodies and T-cell responses; vaccines (e.g., rVSV-ZEBOV and others) elicit protective immunity, but challenges remain for cross-species and durable protection.
Epidemiology and Transmission of the Ebola Virus
Zoonotic Reservoirs and Animal Hosts
- Fruit bats are strongly implicated as natural reservoirs, with high filovirus diversity and mostly asymptomatic infection.
- Experimental infection of Angolan free-tailed bats shows high, disseminated EBOV replication and shedding without disease; EBOV uniquely crosses the placenta, infecting and persisting in fetal tissues, supporting vertical and horizontal transmission in bats.
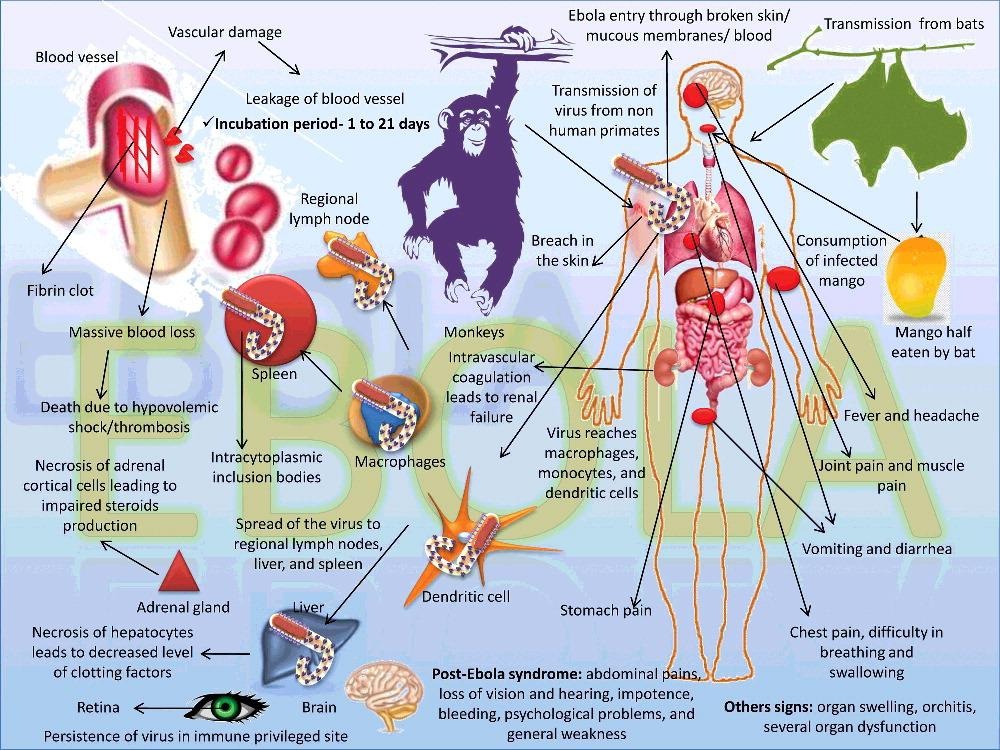
Human Transmission Routes
- Primary spillover from infected wildlife (bats or intermediate hosts) initiates outbreaks; subsequent spread is predominantly human-to-human via direct contact with blood or body fluids.
- Virus is present in saliva, stool, semen, and other secretions; transmission occurs via direct touch, contaminated fomites, droplets, and possibly aerosols in specific settings.
- Skin: infectious virions appear on skin surfaces late in infection; skin supports local replication before viremia, and hair-follicle involvement suggests a route for virus egress. Abraded skin exposed to EBOV GP–bearing virus leads to local epidermal infection and systemic dissemination in mice, requiring PS receptor AXL for efficient entry.
- Sexual transmission: persistent EBOV in semen of male survivors has been linked to outbreak flare-ups; human vaginal epithelium models confirm susceptibility and sustained apical shedding, with associated vaginitis-like inflammation.
Clinical and Outbreak Patterns
- Incubation typically lasts 2–21 days (often 8–10); early symptoms include fever, headache, myalgia, followed by vomiting, diarrhea, and in a subset, hemorrhage.
- Case fatality rates vary (~25–90%), strongly correlated with viral load.
- Most outbreaks occur in Central and East/Central Africa (DRC, Sudan, Uganda, Gabon), with the 2014–2016 West Africa epidemic highlighting rapid spread and global impact.
Table 2: Transmission from Skin and Mucosae: Key Insights
| Site / Route | Key Findings on EBOV Dynamics |
| Skin (general) | Increasing viral RNA and infectious virions; patchy foci, low local inflammation |
| Hair follicles | Dermal/follicular infection suggests a route for shedding to the surface |
| Abraded skin entry | Topical exposure infects keratinocytes and can cause systemic spread; AXL is required |
| Vaginal epithelium | High-dose infection, full-thickness penetration, 7-day apical shedding, inflammatory cytokines |
Clinical Manifestations of Ebola Virus Disease (EVD)
EVD is a multiphase illness that evolves from non-specific febrile symptoms to severe gastrointestinal disease and, in many patients, multi-organ dysfunction and shock.
Incubation and Early Phase
- Incubation period
-2–21 days (mean ~8–12 days) after exposure.
- Early, non-specific symptoms (first 3–5 days of illness)
-Sudden onset:
- Fever or subjective feverishness
- Profound fatigue and weakness
- Myalgia and arthralgia
- Headache, chills.
-Often indistinguishable from malaria, typhoid, Lassa fever, and other tropical infections; co-infection with Plasmodium spp. is common and worsens prognosis.
Gastrointestinal and Systemic Phase
- Timing
-Typically, days 3–7 after symptom onset, paralleling rising viral load.
- Predominant features
-Profuse watery diarrhea (up to 5–10 L/day in severe cases).
-Repetitive vomiting, abdominal pain, anorexia.
-Severe volume depletion:
- Orthostatic symptoms, tachycardia
- Hypotension if not aggressively resuscitated.
- Laboratory correlates in this phase
-Rising aminotransferases (AST often higher than ALT).
-Progressive hypoalbuminemia, hyponatremia, hypokalemia, hypocalcemia, and hypomagnesemia reflect GI loss and capillary leak.
-Lymphopenia, thrombocytopenia, and evolving coagulopathy.
Severe Disease, Hemorrhagic and Multi-Organ Dysfunction
- Clinical progression in highly viraemic patients
-Onset of multiple organ dysfunction syndrome (MODS):
- Shock (vasodilatory and hypovolemic components)
- Acute kidney injury: oliguria/anuria; some require renal replacement therapy.
- Respiratory failure with hypoxemia; 30–50% may need supplemental O₂ or mechanical ventilation in high-resource settings.
-Hemorrhagic manifestations (not universal but prognostically important):
- Mucosal bleeding, GI bleeding, petechiae, ecchymoses, and oozing from venipuncture sites.
-Metabolic acidosis and marked transaminitis peak around day 7–10 after symptom onset.
-High viral loads (>10⁷ genome copies/mL) correlate with poor outcomes.
Recovery Phase and Post-Ebola Sequelae
- Survivor profile
-Often present with lower peak viraemia and less extensive organ dysfunction.
-Clearance of viremia around 2–3 weeks after symptom onset in intensive-care settings.
- Post-Ebola Syndrome
-Persistent symptoms: fatigue, arthralgia, headache, neurocognitive complaints, depression/anxiety.
-Ocular complications (uveitis), meningoencephalitis, and other organ-specific inflammatory syndromes linked to viral persistence in immune-privileged sites even after apparent recovery.
-Viral RNA and sometimes infectious virus are detectable in semen, ocular fluid, and CSF for months to years; associated with relapse and sexual transmission events.
Laboratory Diagnosis of EVD
Laboratory confirmation is essential because clinical presentation overlaps with many tropical infections. Diagnostic strategies must be tailored to the phase of illness, viral load, and biosafety constraints.
Indications and Case Definition
- When to test
-Compatible illness (acute fever + GI symptoms ± hemorrhage) plus epidemiologic risk (contact with a case, funeral exposure, residence/travel in outbreak area).
- Co-testing
-In endemic settings, simultaneous testing and/or empirical treatment for malaria, Lassa fever, and bacterial infections is recommended, as co-infections are common and alter prognosis.
Specimen Types and Biosafety
- Preferred specimen
-Whole blood or plasma collected in an EDTA tube from symptomatic patients.
- Timing
-Viral RNA may be undetectable by RT-PCR in the first 1–3 days after symptom onset; repeat testing is mandatory when suspicion remains high.
- Biosafety
-Samples must be handled under strict PPE with protocols to inactivate the virus before downstream assays, especially in resource-limited settings.
Molecular Diagnostics
- Current gold standard for acute EVD.
- Targets typically conserved gene regions (e.g., L, NP, VP40).
- High sensitivity and specificity after day 3 of illness; cycle-threshold (Ct) values inversely reflect viral load and correlate with prognosis.
- Widely used platforms:
-Commercial kits (e.g., RealStar® assays targeting L gene) with analytical sensitivity ~471 copies/mL for EBOV and higher limits for SUDV.
-Cartridge-based systems (e.g., Cepheid platform) offering near-patient molecular testing with sensitivity ~232 copies/mL.
Isothermal Amplification
- LAMP (Loop-mediated isothermal amplification)
-Detects EBOV RNA at a single temperature; reported analytical sensitivity as low as 1 copy/reaction, compatible with point-of-care deployment.
-Real-world performance data still limited; not yet a stand-alone replacement for RT-PCR.
Antigen-Detection Assays and Rapid Tests
- Immunoassays (ELISA and RDTs)
-Detect viral proteins (VP40, GP, NP) in blood or sometimes other fluids.
-Sensitivity:
- VP40-based: 57.4–93.1%
- GP-based: 53–88.9%
- NP-based: ~85% compared with RT-PCR.
-Sensitivity is highest at high viral loads; specificity is generally good.
-WHO-endorsed RDTs are useful for rapid triage and field use, but none reach the ≥99% sensitivity threshold; any negative RDT in a suspected case requires confirmatory molecular testing.
Serology and Ancillary Tests
- Antibody detection (IgM/IgG ELISA)
-Useful for retrospective diagnosis, serosurveys, or confirming past infection; less useful in early acute stages.
- Routine clinical laboratory findings
Hematology:
- Leukopenia with lymphopenia, thrombocytopenia.
Biochemistry:
- Elevated AST/ALT (often AST>ALT), hyperbilirubinemia in some, and rising creatinine in kidney injury.
Coagulation:
- Prolonged PT/aPTT, high D-dimer, low fibrinogen consistent with DIC.
- These tests support severity assessment and guide supportive care, but are non-specific for EVD.
Point-of-Care and Decentralized Testing
- The 2014–2016 West Africa epidemic catalyzed:
-Deployment of field laboratories, mobile RT-PCR platforms, and validated RDTs close to treatment units.
-Emphasis on ASSURED criteria (Affordable, Sensitive, Specific, User-friendly, Rapid/robust, Equipment-free, Deliverable) by WHO for RDT development.
- Expanded decentralized capacity has improved:
-Turn-around time for diagnosis
-Infection-prevention decisions
-Patient triage and outbreak control.
Prevention and Control of Ebola Virus Disease (EVD)
- Wash your hands with soap and water or use an alcohol-based hand sanitizer.
- Avoid contact with bats, nonhuman primates, and dead wild animals, as these can act as sources of Ebola virus transmission.
- Clean and disinfect frequently touched surfaces regularly to reduce contamination.
- Do not handle or consume bush meat from wild animals, as it may carry the virus.
- Disinfect contaminated clothing, bedding, and other personal items that may have come into contact with infected body fluids.
- Avoid direct contact with people infected with Ebola virus disease and their body fluids, including blood, saliva, vomit, urine, feces, and sweat.
- Avoid direct contact with the bodies of people who died from Ebola, and follow safe burial procedures.
- Wear gloves and appropriate protective clothing when caring for sick individuals or handling potentially infected animals.
Early prevention, infection control, and community awareness are essential to limiting the spread of Ebola virus disease.

Treatment and Antiviral Therapy for the Ebola Virus
Optimal management combines aggressive supportive/critical care with virus-specific therapies, mainly monoclonal antibodies, plus vaccination and infection-control measures at the population level
General Principles of Management
Early diagnosis + early intervention markedly improve survival.
Core components:
- Prompt isolation and PPE use
- Hemodynamic stabilization and organ support
- Management of complications (e.g., DIC, renal failure, secondary infections)
- Early administration of specific antivirals, particularly mAbs, in EBOV (Zaire) outbreaks.
Supportive and Critical Care
Fluid and Electrolyte Management
Massive GI fluid losses (5–10 L/day in critically ill) require:
- Aggressive oral and IV rehydration, sometimes guided by invasive or non-invasive hemodynamic monitoring.
- Frequent correction of hyponatremia, hypokalemia, hypocalcemia, and hypomagnesemia to prevent arrhythmias and neuromuscular complications.
In high-resource settings, individualized fluid and vasoactive therapy, plus nutritional support, is standard.
Organ Support
- Renal failure
-Up to one-third of severely ill patients develop oliguria/anuria; continuous renal replacement therapy is often required where available.
- Respiratory failure
-Hypoxemia requiring high-flow oxygen, non-invasive ventilation, or mechanical ventilation in 30–50% of critically ill.
- Coagulopathy and bleeding
-Support with blood products (packed RBCs, FFP, platelets), vitamin K, and, in some settings, antifibrinolytics or heparin depending on DIC profile.
- Aggressive supportive care in Europe/US reduced mortality to about 18.5%, compared with ~40–70% in African outbreaks.
Virus-Specific Therapies
Monoclonal Antibody (mAb) Therapies (Zaire ebolavirus)
Ebanga™ (mAb114) and Inmazeb™ (REGN-EB3)
- Both are human or human-derived mAbs targeting EBOV GP; FDA approved in 2020 based on the PALM trial.
PALM trial findings
- Randomized trial in DRC comparing REGN-EB3, mAb114, ZMapp and remdesivir in confirmed EVD.
- REGN-EB3 and mAb114 significantly reduced mortality compared with ZMapp and remdesivir, particularly when given early and at lower viral loads.
Dosing
- Single high-dose IV infusion, facilitating use in outbreak settings.
Safety
- Generally, well-tolerated; infusion reactions and hypersensitivity can occur but are manageable.
- Key clinical implications
mAbs are now the standard of care for EBOV (Zaire) EVD in settings where available, administered alongside optimized supportive care.
Limitations:
- Virus species-specific (ineffective against SUDV/BDBV).
- Large molecular size limits penetration into immune-privileged sites, so it may not fully eradicate the persistent virus.
Antivirals and Investigational Agents
Remdesivir and other nucleoside analogs
- Evaluated in PALM; remdesivir did not show a survival benefit over mAbs and is not preferred for acute EBOV (Zaire) EVD, though it retains interest for combination or other filoviruses.
Host-directed and repurposed drugs
- Multiple small molecules (e.g., favipiravir, brincidofovir, chloroquine, clomiphene, amiodarone, etc.) have shown in vitro or animal activity but limited or inconclusive clinical benefit so far.
- Advantages: potential oral administration, lower cost, better penetration into immune-privileged sites; however, none are currently licensed for EVD.
siRNA, PMOs, gene-expression inhibitors
- Small interfering RNAs and phosphorodiamidate morpholino oligomers targeting EBOV genes (e.g., TKM-Ebola, AVI-7537) showed promise in animals and early clinical experience, but development has lagged behind mAbs and vaccines.
Management of Viral Persistence and Sequelae
Challenges
- EBOV persistence in semen, ocular fluid, CNS, and other sanctuaries leads to:
- Late relapses, meningoencephalitis, uveitis
- Sexual transmission months after recovery.
- Therapeutic considerations
- mAbs may have limited penetration; combination regimens with small molecules may be required to fully clear persistent infection.
- Evidence remains sparse, and optimal regimens for relapse and post-EVD syndromes are an important research gap.
Vaccines and Their Interface with Clinical Management
Ervebo® (rVSV-ZEBOV)
- Live, recombinant VSV-vectored vaccine expressing EBOV GP; first FDA-approved EBOV vaccine (2019).
- Highly efficacious in ring vaccination strategies to prevent spread around confirmed cases.
- Other platforms (Ad26.ZEBOV/MVA-BN-Filo, etc.) have been approved by some regulators, but global access and logistics remain challenging.
- Vaccines do not directly treat acute EVD, but:
- Protect high-risk contacts and health-care workers
- Reduce caseload, easing pressure on treatment units and indirectly improving quality of care.
Evolving Standards and Future Directions
Clinical guidelines
- First evidence-based guidelines for specific EVD therapies were published in 2022, formalizing the use of mAbs and supportive care bundles.
Research priorities
- Broader-spectrum antivirals are effective against the Sudan virus and other ebolaviruses.
- Small-molecule agents with good tissue penetration and oral formulations for remote areas.
- Strategies to prevent and treat viral persistence and post-EVD sequelae.
Table 3: Therapeutic Modalities at a Glance
| Modality | Role in EVD Management |
| Aggressive supportive care | Core determinant of survival; manages MODS |
| mAb114 (Ebanga), REGN-EB3 | First-line specific therapy for EBOV (Zaire) |
| Remdesivir | Evaluated; less effective than mAbs in PALM. |
| Favipiravir, brincidofovir, etc. | Investigational/repurposed; limited clinical proof |
| siRNA/PMO-based drugs | Strong preclinical data; human evidence limited |
| Vaccines (Ervebo, etc.) | Prevention and ring vaccination, not treatment |
Conclusion
Ebola virus is a very dangerous filovirus with a pathogenesis characterized by effective replication, an advanced immune evasion (especially through the use of VP24/VP35 and m6A-mediated pathways), and systemic vascular and inflammatory damage. It has a non-segmented RNA genome that encodes seven multifunctional proteins. These proteins mediate a cytoplasmic replication cycle and establish therapeutic targets. Large epidemics, especially the epidemic in West Africa, led to the development of effective vaccines and monoclonal antibody treatments.
However, supportive care and strict measures of public health are still at the core. There are also ongoing concerns about species- and variant-specific virulence, immunological sequelae in survivors, development of antiviral drugs beyond mAbs, and equal access to vaccines and treatments in the affected regions.
References
- (2023). Replicative Cycle of Ebola Virus. Issue Zoonosis Volume 3. https://doi.org/10.47278/book.zoon/2023.119
- Abdul-Rahman, T., Lawal, L., Meale, E., Ajetunmobi, O., Toluwalashe, S., Alao, U., Ghosh, S., Garg, N., Aborode, A., Wireko, A., Mehta, A., & Sikora, K. (2023). Inequitable access to Ebola vaccines and the resurgence of Ebola in Africa: A state-of-the-art review. Journal of Medical Virology, 95. https://doi.org/10.1002/jmv.28986
- Almeida-Pinto, F., Pinto, R., & Rocha, J. (2024). Navigating the Complex Landscape of Ebola Infection Treatment: A Review of Emerging Pharmacological Approaches. Infectious Diseases and Therapy, 13, 21 – 55. https://doi.org/10.1007/s40121-023-00913-y
- Ayoubi, L., Mahmoud, O., Zakhour, J., & Kanj, S. (2024). Recent advances in the treatment of Ebola disease: A brief overview. PLOS Pathogens, 20. https://doi.org/10.1371/journal.ppat.1012038
- Cao, Z., Liu, C., Peng, C., Ran, Y., Yao, Y., Xiao, G., Li, E., Chen, Z., Chuai, X., & Chiu, S. (2023). Ebola virus VP35 perturbs type I interferon signaling to facilitate viral replication. Virologica Sinica, 38, 922-930. https://doi.org/10.1016/j.virs.2023.10.004
- Chakravarty, M., & Vora, A. (2020). Nanotechnology-based antiviral therapeutics. Drug Delivery and Translational Research, 11, 748 – 787. https://doi.org/10.1007/s13346-020-00818-0
- Fatima, K., Panwar, R., Varnika, .., Saini, P., Sudhanshu, .., Bhargava, R., Bhardwaj, K., Semwal, A., Semwal, A., Jabi, S., Maurya, V., Verma, R., & Gaurav, N. (2024). The Tactical Strategy, Readiness, Epidemiology, and Reaction to Potential Treatments for the Possibly Lethal Ebola Virus: A Review. UTTAR PRADESH JOURNAL OF ZOOLOGY. https://doi.org/10.56557/upjoz/2024/v45i73986
- Feldmann, H., & Geisbert, T. W. (2010). Ebola haemorrhagic fever. The Lancet, 377(9768), 849–862. https://doi.org/10.1016/s0140-6736(10)60667-8
- Hansen, F., Feldmann, H., & Jarvis, M. (2021). Targeting Ebola virus replication through pharmaceutical intervention. Expert Opinion on Investigational Drugs, 30, 201 – 226. https://doi.org/10.1080/13543784.2021.1881061
- He, F., Mélen, K., Kakkola, L., & Julkunen, I. (2019). Interaction of Ebola Virus with the Innate Immune System. Emerging Challenges in Filovirus Infections. https://doi.org/10.5772/intechopen.86749
- Jacob, S., Crozier, I., Fischer, W., Hewlett, A., Kraft, C., De La Vega, M., Soka, M., Wahl, V., Griffiths, A., Bollinger, L., & Kuhn, J. (2020). Ebola virus disease. Nature Reviews. Disease Primers, 6. https://doi.org/10.1038/s41572-020-0147-3
- Jain, S., Khaiboullina, S., & Baranwal, M. (2020). Immunological Perspective for Ebola Virus Infection and Various Treatment Measures Taken to Fight the Disease. Pathogens, 9. https://doi.org/10.3390/pathogens9100850
- Kasarla, R., Verma, A., Bhandari, N., & Pathak, L. (2024). Ebola Haemorrhagic Fever: An Overview. Janaki Medical College Journal of Medical Science. https://doi.org/10.3126/jmcjms.v12i01.65247
- Kotliar, D., Lin, A., Logue, J., Hughes, T., Khoury, N., Raju, S., Wadsworth, M., Chen, H., Kurtz, J., Dighero-Kemp, B., Bjornson, Z., Mukherjee, N., Sellers, B., Tran, N., Bauer, M., Adams, G., Adams, R., Rinn, J., Melé, M., Schaffner, S., Nolan, G., Barnes, K., Hensley, L., Mcilwain, D., Shalek, A., Sabeti, P., & Bennett, R. (2020). Single-Cell Profiling of Ebola Virus Disease In Vivo Reveals Viral and Host Dynamics. Cell, 183, 1383 – 1401.e19. https://doi.org/10.1016/j.cell.2020.10.002
- Letafati, A., Ardekani, O., Karami, H., & Soleimani, M. (2023). Ebola virus disease: A narrative review. Microbial pathogenesis, 106213. https://doi.org/10.1016/j.micpath.2023.106213
- Luthra, P., Naidoo, J., Pietzsch, C., De, S., Khadka, S., Anantpadma, M., Williams, C., Edwards, M., Davey, R., Bukreyev, A., Ready, J., & Basler, C. (2018). Inhibiting pyrimidine biosynthesis impairs Ebola virus replication through depletion of nucleoside pools and activation of innate immune responses. Antiviral Research, 158, 288–302. https://doi.org/10.1016/j.antiviral.2018.08.012
- Manu, M. (2018). Infectious characteristics of the Ebola virus. International Journal of Virology and AIDS, 5(1). https://doi.org/10.23937/2469-567x/1510044
- Mirza, M., Vanmeert, M., Ali, A., Iman, K., Froeyen, M., & Idrees, M. (2019). Perspectives towards antiviral drug discovery against the Ebola virus. Journal of Medical Virology, 91, 2029 – 2048. https://doi.org/10.1002/jmv.25357
- Nicastri, E., Kobinger, G., Vairo, F., Montaldo, C., Mboera, L., Ansunama, R., Zumla, A., & Ippolito, G. (2019). Ebola Virus Disease: Epidemiology, Clinical Features, Management, and Prevention. Infectious disease clinics of North America, 33 4, 953-976. https://doi.org/10.1016/j.idc.2019.08.005
- Rajappa, M. (2024). Ebola Epidemic: A Retrospective Review on Pathophysiology, History, Epidemiology, Diagnosis, Treatment, and Prevention (1976–2023). Journal of Communicable Diseases. https://doi.org/10.24321/0019.5138.202361
- Singh, R. K., Dhama, K., Malik, Y. S., Ramakrishnan, M. A., Karthik, K., Khandia, R., Tiwari, R., Munjal, A., Saminathan, M., Sachan, S., Desingu, P. A., Kattoor, J. J., Iqbal, H. M., & Joshi, S. K. (2017). Ebola virus – epidemiology, diagnosis, and control: threat to humans, lessons learnt, and preparedness plans – an update on its 40-year journey. Veterinary Quarterly, 37(1), 98–135. https://doi.org/10.1080/01652176.2017.1309474
- Taki, E., Ghanavati, R., Navidifar, T., Dashtbin, S., Heidary, M., & Moghadamnia, M. (2023). EbangaTM: The most recent FDA-approved drug for treating Ebola. Frontiers in Pharmacology, 14, 1083429. https://doi.org/10.3389/fphar.2023.1083429
- Yamaoka, S., & Ebihara, H. (2021). Pathogenicity and Virulence of Ebolaviruses with Species- and Variant-specificity. Virulence, 12, 885 – 901. https://doi.org/10.1080/21505594.2021.1898169
- Zhao, B., Cao, Z., Zhang, K., Xiao, S., Shen, X., Li, F., Wu, Y., Zhang, B., Wang, Q., Zhang, H., Chuai, X., & Chiu, S. (2025). m6A modification in the 3′-leader region of EBOV genome antagonizes ISG20 to facilitate viral replication.. International journal of biological macromolecules, 149804. https://doi.org/10.1016/j.ijbiomac.2025.149804

Very informative, thank you bringing everything together sir.
Thanks you so much sir
for giving usefull information and helping me to take a clear notes
Thanks for your well presented Dr
Thans dear professre
today I am very happy thad I used your impormation abute Ebula viru