What is Cysticercosis?
Cysticercosis refers to the tissue infection caused by the metacestode, or larval stage, of Taenia solium. It is acquired by the accidental ingestion of T. solium eggs. It is endemic to many parts of the developing world, including Latin America, Asia, and sub-Saharan Africa. Cysticercosis is the most common intra-orbital parasitic infection and is observed in 13-46% of infected individuals. The larvae of T. solium gets into tissues such as muscle and brain, and form cysts there (these are called cysticerci).
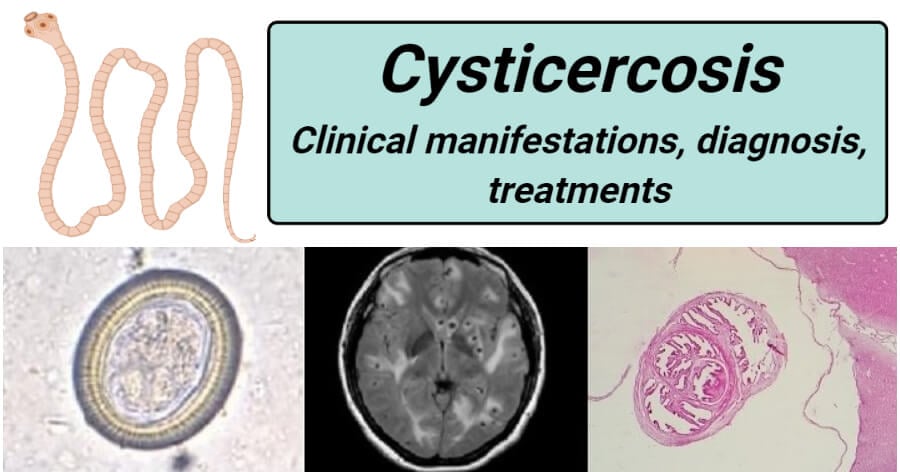
Figure: Below Left: Taenia egg at a high magnification of 400x. When consumed by humans, Taenia solium eggs can lead to cysticercosis, including a serious condition known as neurocysticercosis. Below Center: A radiographic image of the brain of a patient who has neurocysticercosis; the small dark spots within the brain are larval cysts of T. solium. Below Right: A cross-section through a T. solium cyst from a human brain tissue specimen, stained with hematoxylin and eosin (H&E). (Credit (Below L to R): Westchester Medical Center, PHIL, DPDx.)
Clinical manifestations
The incubation period is variable, the infected people may remain asymptomatic for a year. The symptoms can occur months to years after infection, usually when the cysts start dying. When cysts die, the brain or other tissue around the cyst may swell. Its clinical effects vary depending on the site of larval lodging, larval burden, and host reaction. The clinical features depend on the site affected
a. Subcutaneous cysticercosis
- It present as small, movable, painless modules that are commonly noticed in the arms or chest.
- After a few months or even years, the nodules become swollen, tender, and inflamed, and then they gradually disappear.
b. Muscular cysticercosis
- It appears as dot-shaped or ellipsoidal calcification.
- In this cysticercosis, three types of clinical manifestations have been described: the myalgic type; the mass-like, pseudotumor, or abscess-like type; and the rare pseudohypertrophic type, depending upon the pathogenesis.
c. Neurocysticercosis
- It is the most common and serious form of cysticercosis infection in the brain due to mass effects and inflammation induced by degeneration of cysticerci and release of antigens.
- Neurocysticercosis is endemic in most Latin American countries, sub-Saharan Africa, and large regions of Asia, including the Indian subcontinent, most of southeast Asia, and China.
- It is observed in 60-90% of infected patients.
- Cysts localized within the brain may range anywhere from 4-20 mm in length, but most commonly average between 8-10 mm.
- Seizures and headaches are the most common symptom of neurocysticercosis and occur in 70-90% of infected patients.
other symptoms of Neurocysticerosis include dizziness, involuntary muscle movement, intracranial hypertension, dementia, hydrocephalus(excess fluid around the brain), psychiatric disturbances, meningoencephalitis, transient paresis, behavioral disorders aphasia, and visual disturbances. - It is considered the second most common cause of intracranial space-occupying lesion (ICSOL) after Tuberculosis in India.
- Brain cysts cause the most morbidity, with the brain being the most common location for cysts (60 to 90 percent of all cases).
d. Ocular cysticercosis
- This condition is present in 20% of cases.
- In the case of ocular cysticercosis, cysts are found in the vitreous humor, subretinal space, and conjunctiva.
- The condition may present an as blurred vision or loss of vision, iritis, uveitis, and palpebral conjunctivitis
- In extreme cases, the infection may cause complete detachment of the retina.
Laboratory diagnosis
Diagnosis is often difficult due to the nonspecific nature of symptoms associated with cysticercosis. Therefore, proper diagnosis of the diseases is most often based on a combination of clinical, serological, and epidemiological data.
1. Microscopy
- Samples: Biopsy
- The definitive diagnosis of cysticercosis is by biopsy of the lesion and its microscopic examination to show the invaginated
scolex with suckers and hooks.
2. Imaging Methods
a. X-ray
- Calcified cysticerci can be detected by radiography of subcutaneous tissue and muscles, particularly in the buttocks and thigh.
- X-ray of the skull many demonstrate cerebral calcified cyst and intracranial lesions in the neurocysticercosis.
b. Computed tomography (CT)
- CT scan of the brain is the best method for detecting dead calcified cysts.
- The cysticercal lesions appear as small hypodensities (ring or disc-like) with a bright central spot.
c. Magnetic resonance imaging (MRI)
- MRI scan of the brain is more helpful in the detection of noncalcified cysts and ventricular cysts.
- It also demonstrates spinal cysticerci.
* However, the high costs associated with these radiologic methods greatly restrict the availability and/or accessibility of these tests in most underdeveloped countries where the disease is endemic.
3. Serology
a. Antibody detection
- Samples: cerebrospinal fluid (CSF) and serum
- Anticysticercus antibodies in serum or cerebrospinal fluid (CSF) can be detected by ELISA and enzyme-linked immunoelectrotrasfer blot (EITB) tests.
- Native antigens: The enzyme-linked immunoelectrotransfer blot (EITB) assay with an affinity-purified lentil-lectin glycoprotein fraction (LLGP), used as antigens, is the gold-standard for Neurocysticercosis serodiagnosis. It has a Sensitivity of 90 % and specificity of 50-70%.
- However, EITB is not effective in the detection of antibodies when only one cyst is present.
- The ELISA, while not as sensitive, is technically simpler and is therefore used extensively in clinical settings.
b. Antigen detection
- Samples: cerebrospinal fluid (CSF) and serum
- The antigen can be detected in serum and CSF by ELISA, using monoclonal antibodies.
- Detection of antigens in serum or CSF indicates recent or viable infection.
- Indirect immunofluorescence assay (IFA)is an antigen detection method for Neurocysticercosis using monoclonal antibodies.
4. DNA detection
- Samples: CSF, biopsy
- Amplification protocols are the most used methods (PCRs) and are mainly based on the sequence of repetitive DNA, ribosomal DNA, mitochondrial DNA, and Antigen 2 (Ag2) gene.
- The amplification of pTsol9 repetitive sequence, in both conventional and real-time PCR, and the seminested-HDP2-PCR, have already been used in Neurocysticercosis case identification.
- It shows a relevant sensitivity and excellent specificity in neurocysticercosis diagnosis.
5. Others
- Ocular cysticercosis can be made out by ophthalmoscopy
- Eosinophilia usually occurs in the early stage of cysticercosis but is not constant
- ElevatedCSF protein level in neurocysticercosis.
- CSF may show lymphocytosis- Mononuclear pleocytosis is frequent
- Glucose levels may be mild to moderate low.
- Cell counts rarely exceed 300/mm3
Treatments
- For cysticercosis, excision is the best method, wherever possible.
- Asymptomatic neurocysticercosis requires no treatment.
- For symptomatic cases, Praziquantel and albendazole are the two anticysticercal drugs used to treat patients diagnosed with cysticercosis in the brain and skeletal muscles.
- Doses: Praziquantel (50 mg/kg in 3 divided doses for 20–30 days ), albendazole(400 mg twice daily for 30 days).
- Corticosteroids may be given along with praziquantel or albendazole to reduce the inflammatory reactions caused by the dead cysticerci.
- In addition, antiepileptic drugs should be given until the reaction of the brain has subsided.
- Surgical removal of cysts from infected tissues is possible and, prior to the development of anticysticercal drugs, was the primary means of treatment. However, the invasiveness and high risk of complications associated with surgery make this method less favorable to treatment with chemotherapeutic agents.
Prophylaxis
- Beef and pork to be eaten by man should be subjected to effective inspection for cysticerci in the slaughterhouse.
- Avoidance of eating raw or undercooked beef and pork. The critical thermal point of cysticercus is 56°C for 5 minutes.
- Maintenance of clean personal habits and general sanitary measures.
- Vaccines aimed at preventing infection in pigs may play a role in efforts to control the spread of disease. Due to their typically short-life span (approximately one year), pigs do not require long-term immunity; therefore, vaccines that provide only short term resistance may be sufficient to prevent the spread of infection to humans. Additionally, the vaccination, rather than the confiscation, of pigs is often a more favorable alternative to local farmers.
- To date, the most effective vaccines have involved the expression of recombinant oncosphere antigens TSOL18 and TSOL45 in E. coli. TSOL18 appears to be more effective, inducing greater than 99% protection in the five vaccine trials undertaken thus far.
- Current efforts are focused on developing the methods necessary to make the vaccine widely available and successful on a practical scale. The use of recombinant vaccines in pigs, combined with anticysticercal chemotherapy in humans, seems to be the most effective approach in the battle against cysticercosis and appears to have the potential to control and/or eradicate the disease.
References and Sources
- Garcia, H. H., Nash, T. E., & Del Brutto, O. H. (2014). Clinical symptoms, diagnosis, and treatment of neurocysticercosis. The Lancet. Neurology, 13(12), 1202–1215. https://doi.org/10.1016/S1474-4422(14)70094-8
- Meena, D., Gupta, M., Jain, V. K., & Arya, R. K. (2016). Isolated intramuscular cysticercosis: Clinicopathological features, diagnosis and management – A review. Journal of clinical orthopaedics and trauma, 7(Suppl 2), 243–249. https://doi.org/10.1016/j.jcot.2016.06.016.
- Gómez-Morales, M. A., Gárate, T., Blocher, J., Devleesschauwer, B., Smit, G., Schmidt, V., Perteguer, M. J., Ludovisi, A., Pozio, E., Dorny, P., Gabriël, S., & Winkler, A. S. (2017). Present status of laboratory diagnosis of human taeniosis/cysticercosis in Europe. European journal of clinical microbiology & infectious diseases : official publication of the European Society of Clinical Microbiology, 36(11), 2029–2040. https://doi.org/10.1007/s10096-017-3029-1
- 3% – https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5653711/
- 3% – https://www.cdc.gov/parasites/cysticercosis/gen_info/faqs.html
- 2% – https://www.sciencedirect.com/science/article/pii/S0140673603141177
- 2% – https://www.researchgate.net/publication/8882611_DNA_Differential_Diagnosis_of_Taeniasis_and_Cysticercosis_by_Multiplex_PCR
- 2% – https://www.researchgate.net/publication/269178749_Clinical_symptoms_diagnosis_and_treatment_of_neurocysticercosis
- 2% – https://www.researchgate.net/publication/232235842_Cysticercosis_of_the_masseter
- 1% – https://www.sciencedirect.com/topics/medicine-and-dentistry/immunofluorescence-test
- 1% – https://www.researchgate.net/publication/284464659_Solitary_Cysticercosis_in_Eye_Literature_Review_and_A_Hypothesis_on_Transmission_of_Infection
- 1% – https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4119910/
- 1% – https://www.medicinenet.com/cysticercosis/article.htm
- 1% – https://www.aafp.org/afp/2007/0701/p91.html
- 1% – https://pubmed.ncbi.nlm.nih.gov/24554747/
- 1% – https://pubmed.ncbi.nlm.nih.gov/19358669/
- 1% – https://europepmc.org/article/MED/12932389
- <1% – https://www.sciencedirect.com/science/article/pii/S1471492209002839
- <1% – https://www.sciencedirect.com/science/article/pii/S1387265607130040
- <1% – https://www.sciencedirect.com/science/article/pii/S1045105619300545
- <1% – https://www.researchgate.net/publication/6631386_Conformation-sensitive_immunoassays_improve_the_serodiagnosis_of_solitary_cysticercus_granuloma_in_Indian_patients
- <1% – https://www.researchgate.net/publication/324530698_Clay_Nanoparticles_Elicit_Long-Term_Immune_Responses_by_Forming_Biodegradable_Depots_for_Sustained_Antigen_Stimulation
- <1% – https://www.researchgate.net/publication/232216530_Neurocysticercosis_A_Pictorial_Review
- <1% – https://www.healthtap.com/q/praziquantel-dosage/
- <1% – https://www.healthdirect.gov.au/magnetic-resonance-imaging-mri
- <1% – https://www.health.harvard.edu/diseases-and-conditions/preventing-the-spread-of-the-coronavirus
- <1% – https://newsnetwork.mayoclinic.org/discussion/infectious-diseases-a-z-will-eating-undercooked-pork-make-you-sick/
- <1% – https://en.wikipedia.org/wiki/Cysticercosis
- <1% – https://diseasesdic.com/neurocysticercosis-causes-diagnosis-and-treatment/