Crimean-Congo hemorrhagic fever virus (CCHFV) is a highly pathogenic, widespread, tick-borne virus with complex molecular biology and great public health significance.

Taxonomy and Classification of Crimean-Congo Hemorrhagic Fever Virus (CCHFV)
- Order: Bunyavirales
- Family: Nairoviridae
- Genus: Orthonairovirus
- Species: Crimean-Congo hemorrhagic fever orthonairovirus (CCHFV)
- Negative-sense, single-stranded RNA virus with tri-segmented genome (S, M, L).
- Genetically the most diverse known arbovirus; up to ~20% nucleotide divergence in S and ~31% in M segment between strains.
- Seven main phylogenetic lineages based on the S segment (Africa 1–3, Asia 1–2, Europe 1–2), reflecting strong geographic structure.
- WHO and many national agencies list CCHFV as a priority/high-consequence pathogen because of high lethality, epidemic potential, and lack of licensed countermeasures.
Structure and Morphology of Crimean-Congo Hemorrhagic Fever Virus (CCHFV)
- Enveloped, roughly spherical virions, ~80–100 nm in diameter
- Envelope derived from host Golgi/ER membranes and studded with two major surface glycoproteins Gn and Gc, mediating attachment and membrane fusion.
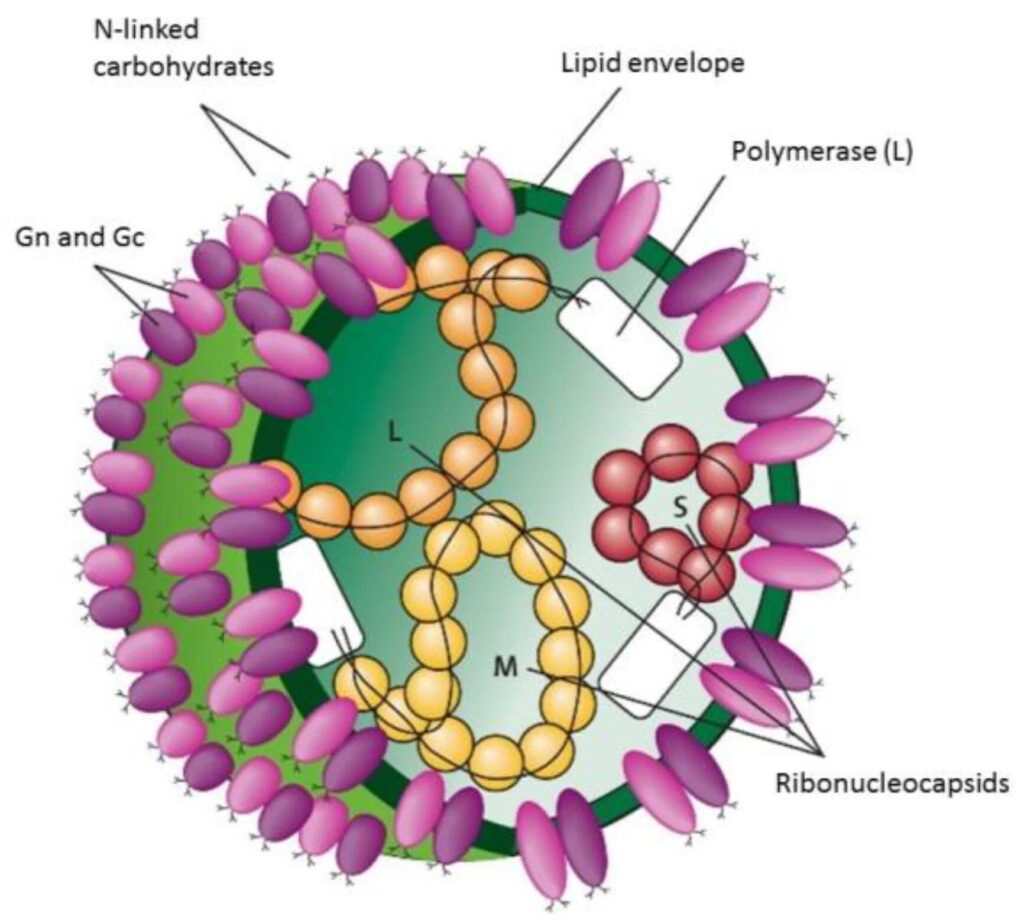
- Evidence suggests accessory glycoprotein GP38 can also be virion-associated, although its precise localization and role remain unclear.
- Internally, a helical ribonucleoprotein (RNP) complex formed by genomic RNA tightly coated with nucleoprotein (NP) and associated with the L polymerase.
- CCHFV virions are pleomorphic; ultra-thin EM shows dense core surrounded by a lipid bilayer with surface projections corresponding to glycoprotein spikes.
Genome Organization and Proteins of Crimean-Congo Hemorrhagic Fever Virus (CCHFV)
Segmented genome: three negative-sense ssRNA segments with complementary termini forming pan-handle–like structures.
S segment (~1.7 kb)
-Encodes nucleoprotein (NP), a highly conserved, RNA-binding protein forming super-helical homo-oligomers that encapsidate the viral RNA.
-In many strains, a small opposite-sense ORF encodes NSs, an interferon antagonist typical of bunyaviruses; its expression and significance in CCHFV remain incompletely defined.
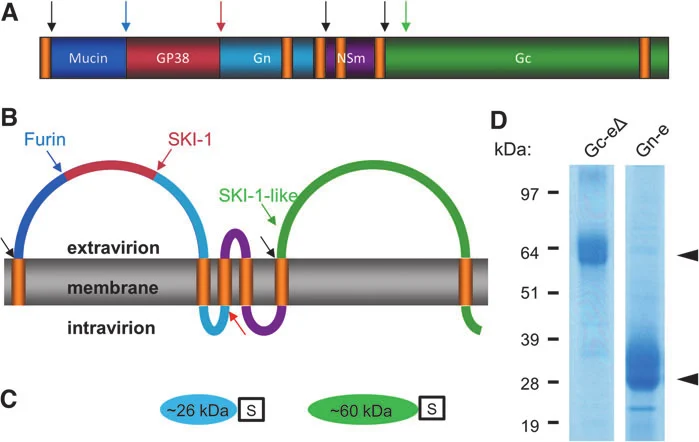
M segment (~5.3 kb)
-Encodes a polyprotein glycoprotein precursor (GPC).
-Co- and post-translational processing by host signal peptidases, SKI-1/S1P and other proteases yields:
- Mucin-like domain (MLD): heavily glycosylated, potentially shielding virions from neutralization.
- GP38 and GP85/160 intermediates: GP38 is a secreted/virion-associated accessory glycoprotein implicated in immune modulation and as a protective antigen.
- Gn and Gc: structural envelope glycoproteins important for receptor binding, tropism, and fusion.
- NSm: a non-structural glycoprotein that may influence assembly and budding; function still being clarified.
L segment (~12 kb; unusually large for bunyaviruses)
-Encodes L protein, containing:
- RNA-dependent RNA polymerase (RdRp) catalytic core needed for replication/transcription.
- N-terminal ovarian tumour (OTU)–like protease, a viral deubiquitinase/deISGylase that counteracts innate immune signaling by cleaving ubiquitin/ISG15 from host proteins.
- High amino-acid conservation in NP and L (≥95%) but much lower in glycoproteins (≤75% identity), contributing to antigenic diversity and immune escape.
Replication Cycle of Crimean-Congo Hemorrhagic Fever Virus (CCHFV)
Viral Entry and Attachment
- CCHFV initiates infection by binding to host cell surface receptors; candidate receptors include DC-SIGN and nucleolin, though precise receptors remain incompletely defined.
- The virus enters cells primarily via clathrin-mediated endocytosis, a pH-dependent process requiring acidification of late endosomes to trigger fusion.
- The viral envelope glycoprotein Gc undergoes conformational changes upon acidification, mediating fusion of the viral and endosomal membranes, releasing ribonucleoprotein complexes (RNPs) into the cytoplasm.
Transcription and Translation
- Once in the cytoplasm, the viral RNA-dependent RNA polymerase (L protein) performs cap-snatching, cleaving 5′ capped fragments from host mRNAs to prime viral mRNA synthesis.
- Viral mRNAs are transcribed from each of the three negative-sense RNA segments (S, M, L) and translated by host ribosomes:
-The S segment encodes nucleoprotein (NP) that encapsidates viral RNA.
-The M segment encodes a glycoprotein precursor (GPC), processed into Gn, Gc, GP38, mucin-like domain, and NSm proteins.
-The L segment encodes the multifunctional L protein with polymerase and OTU protease activities.
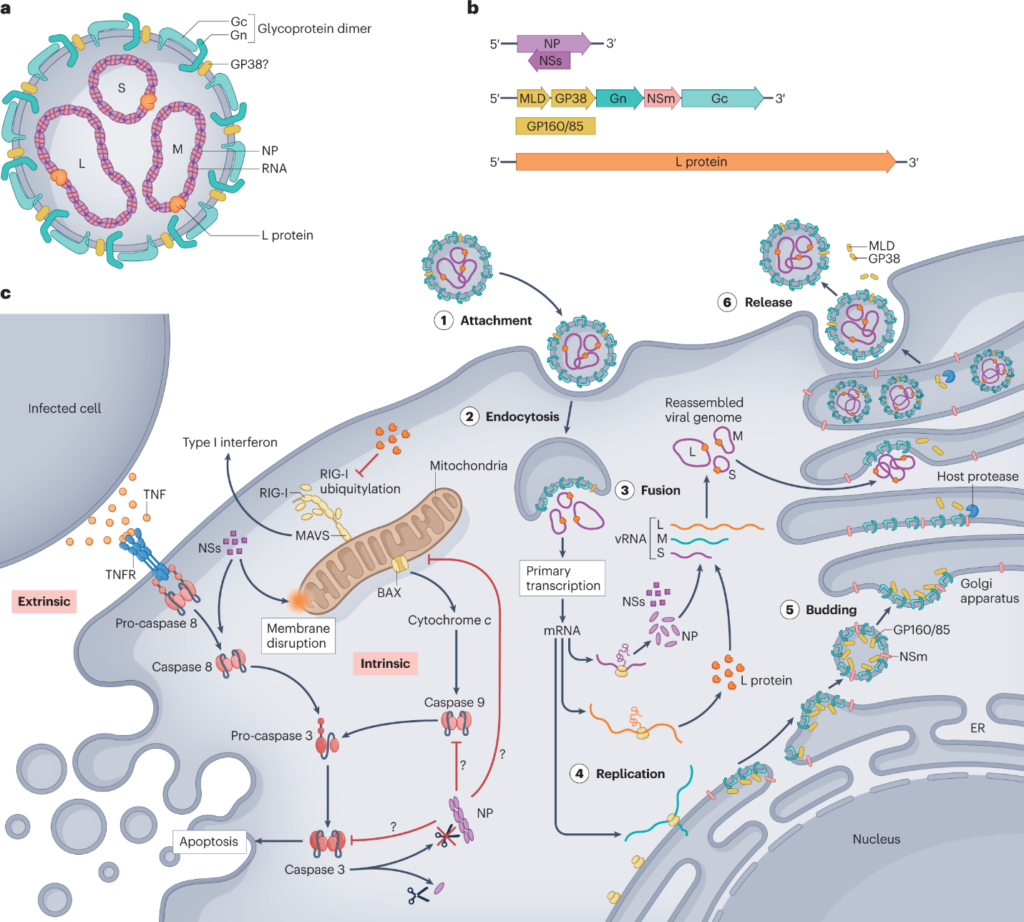
Genome Replication
- The virus switches from transcription to replication mode to produce full-length antigenomic RNAs serving as templates for progeny genomes.
- NP oligomerizes around nascent RNA strands, forming RNPs essential for genome stability and packaging.
Assembly and Egress
- Newly formed RNPs migrate to the Golgi or ER-Golgi intermediate compartments, where Gn and Gc accumulate.
- Virion assembly occurs by budding into intracellular membranes enriched with glycoproteins; mature virions are transported via secretory vesicles to the cell surface for release by exocytosis.
Host Metabolic Reprogramming During Replication
- Multi-omics studies reveal CCHFV infection induces metabolic shifts in host cells, notably upregulation of oxidative phosphorylation and alterations in glycolysis and glutaminolysis pathways critical for efficient viral replication.
- Pharmacological inhibition of glycolysis or glutaminolysis significantly reduces viral replication in vitro, highlighting potential antiviral targets.
Pathogenesis and Host Immune Response to Crimean-Congo Hemorrhagic Fever Virus (CCHFV)
Viral Tropism and Tissue Damage
- CCHFV infects multiple cell types, including monocytes/macrophages, dendritic cells, endothelial cells, hepatocytes, and possibly renal tubular cells.
- Infection leads to widespread viral dissemination with high titers in liver and spleen; histopathology shows hepatocellular necrosis, splenic lymphoid depletion, endothelial damage, hemorrhages, and multi-organ involvement.
Innate Immune Evasion and Dysregulation
- The virus employs its OTU protease domain within the L protein to antagonize innate immunity by deubiquitinating key signaling molecules involved in RIG-I-like receptor pathways, delaying type I interferon responses critical for early control.
- Delayed or suppressed interferon induction allows rapid viral replication, leading to high viremia associated with severe disease outcomes.
Cytokine Storm and Immunopathology
- Severe cases exhibit a cytokine storm, characterized by elevated pro-inflammatory cytokines such as TNF-α, IL-6, IL-8, IL-10, IFN-γ inducible protein 10 (IP-10), GM-CSF, and MCP-1, among others.
- Excessive cytokine release contributes to endothelial activation/dysfunction, causing increased vascular permeability, vasodilation, hypotension, disseminated intravascular coagulation (DIC), shock, multi-organ failure, and death.
Adaptive Immune Response Dynamics
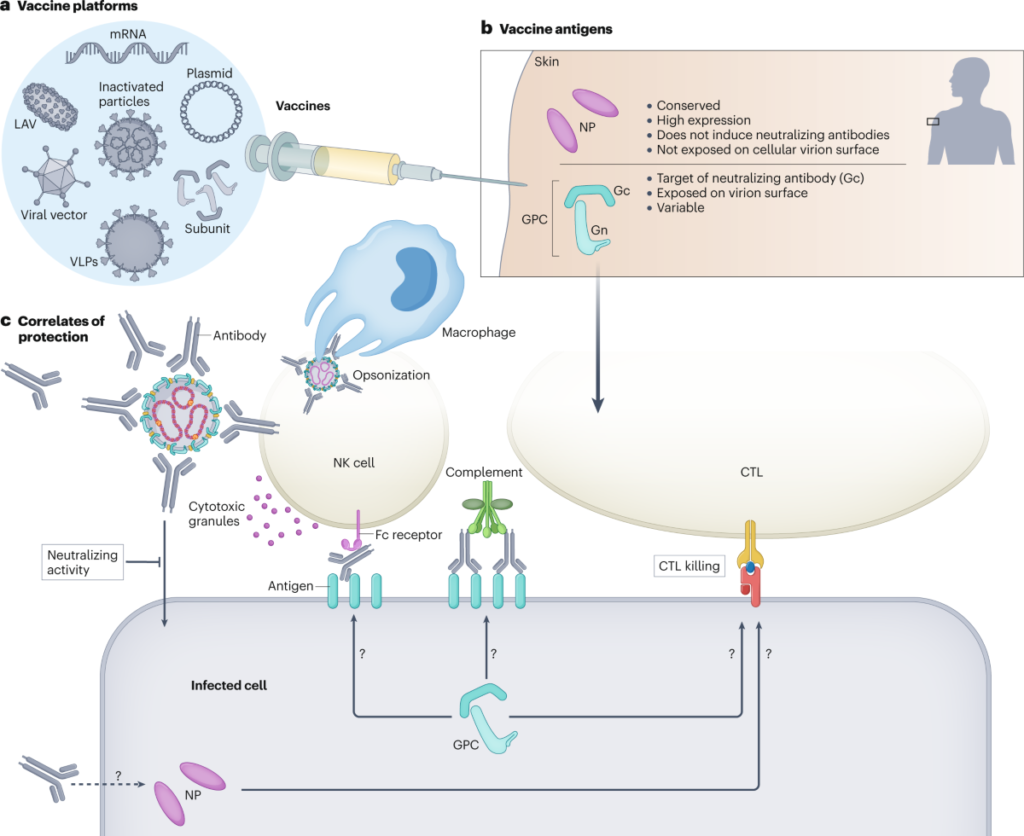
- Humoral immunity develops with IgM appearing around day 5–7 post-onset, followed by IgG; neutralizing antibodies mainly target glycoproteins Gn/Gc/GP38, but early antibody responses may be weak or delayed in fatal cases.
- Robust CD8+ T-cell responses targeting NP and glycoproteins correlate with viral clearance; however, severe disease is associated with lymphocyte apoptosis/depletion and impaired T-cell activation.
Immunopathogenic Mechanisms Summary: Two Main Theories
-Direct Endothelial Infection: Virus infects endothelial cells, causing direct cytopathic effects leading to vascular leakage.
-Immune-Mediated Damage: Excessive immune activation causes cytokine-mediated endothelial dysfunction without necessarily requiring direct infection of the endothelium.
Animal Models Insights
- STAT1 knockout mice lacking interferon signaling develop fatal disease mimicking human CCHF with fever, thrombocytopenia, leukopenia, elevated liver enzymes, systemic viral spread, and cytokine storm, confirming the critical role of interferon response in pathogenesis control
Epidemiology and Transmission of Crimean-Congo Hemorrhagic Fever Virus (CCHFV)
- Geographic distribution
-Endemic across Africa, the Middle East, Central and South-Western Asia, and Southern/Eastern Europe.
-Emerging/establishing in Western Europe, Spain has reported ≥17 autochthonous human cases since 2013 and widespread viral circulation in ticks and wildlife.
-Tick and serologic evidence in France and other countries without recognized human cases suggests silent circulation and risk of emergence.
- Vectors and enzootic cycle
-Maintained in a tick–vertebrate–tick cycle, mainly involving hard ticks of the Hyalomma genus (vectors and reservoirs).
-Additional tick genera (Rhipicephalus, Dermacentor, Amblyomma, Ornithodoros) can harbor and transmit CCHFV in some settings.
-Vertical (transovarial) and transstadial transmission in ticks allow long-term maintenance.
- Animal hosts
-Domestic ruminants (cattle, sheep, goats) and some wildlife (hares, wild ungulates) develop brief viremia without clinical disease, acting as amplifying hosts.
-Birds generally show no disease but can transport infected ticks across long distances, shaping geographic spread.
- Human transmission routes
-Tick bites (especially Hyalomma spp.) on agricultural workers, herders, abattoir workers, and veterinarians.
-Contact with blood or tissues of viremic livestock during slaughter or veterinary procedures.
-Nosocomial and household transmission via exposure to blood/body fluids, aerosol-generating procedures, or inadequate PPE.
- Burden and trends
-Global case-fatality rate (CFR) pooled estimate: ~11.7% in acute infections, with higher CFRs in low-income countries and some outbreaks up to 30–60%.
-Seroprevalence in humans varies widely (4–12% overall; much higher in occupationally exposed groups); animal past-infection seroprevalence is around 12%.
-Climate change, land-use changes, livestock movements, and the expansion of Hyalomma ticks in Europe likely increase the risk of emergence into new areas.
Clinical Manifestations of Crimean-Congo Hemorrhagic Fever Virus (CCHFV)
Incubation Period & Initial Presentation
- Incubation ranges from 1–3 days after a tick bite up to 9 days; longer (~5–6 days) after exposure to infected blood/tissues.
Clinical Phases
Pre-Hemorrhagic (Prodromal) Phase (2–7 days):
- Sudden onset high fever (>39°C), severe headache (retro-orbital), myalgia (especially back/limbs), arthralgia.
- Gastrointestinal symptoms: nausea/vomiting/diarrhea/abdominal pain are common.
- Other signs: photophobia, conjunctival injection/flushing; relative bradycardia may be noted early on.
Hemorrhagic Phase (Days 3–7):
- Petechiae progressing to ecchymoses; mucosal bleeding, including epistaxis, gingival bleeding; gastrointestinal hemorrhage (hematemesis/melena).
- Severe cases develop multi-site bleeding, including hematuria/vaginal bleeding/CNS hemorrhage, leading to confusion/coma/shock.
- Laboratory findings: thrombocytopenia (<50 ×10^9/L), leukopenia or leukocytosis; prolonged PT/aPTT; elevated D-dimer/fibrin degradation products consistent with DIC-like coagulopathy; markedly raised AST/ALT/LDH/CK indicating hepatic injury.
Convalescent Phase:
- Recovery usually begins after day 10 if the patient survives the acute phase; fatigue/myalgia may persist for weeks/months.
- Some patients develop prolonged IgM positivity up to one-year post-infection, indicating sustained immune activation or antigen persistence.
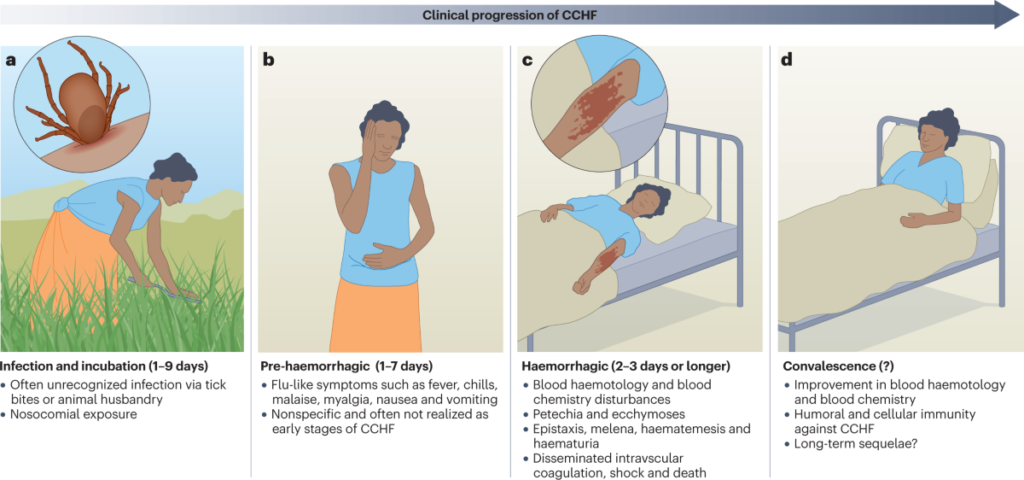
Table 1: Severity Spectrum & Prognostic Indicators
| Clinical Feature | Association |
| High initial viral load | Severe disease & fatal outcome |
| Profound thrombocytopenia | Bleeding risk |
| Elevated liver enzymes | Hepatic involvement |
| Elevated pro-inflammatory cytokines | Cytokine storm & poor prognosis |
| CNS involvement | Increased mortality |
| Older age | Worse outcomes |
Subclinical Infections & Epidemiological Considerations
- A substantial proportion of infections are asymptomatic or mild but underreported due to nonspecific symptoms resembling other febrile illnesses.
- Serosurveys indicate seroprevalence rates ranging from 4% up to >20% in endemic regions among at-risk populations such as farmers/slaughterhouse workers, suggesting frequent subclinical exposure.
Laboratory Diagnosis of Crimean-Congo Hemorrhagic Fever Virus (CCHFV)
Biosafety Considerations
- CCHFV is classified as a biosafety level 4 (BSL-4) pathogen due to its high lethality and potential for aerosol transmission; all diagnostic procedures involving live virus or infectious materials require BSL-4 containment.
- Clinical specimens (blood, serum, tissues) must be handled with strict infection control measures to prevent laboratory-acquired infections.
Diagnostic Methods Overview
- Early and accurate diagnosis is critical for patient management and outbreak control.
- Diagnostic approaches include:
- Molecular detection (RT-PCR)
- Serological assays (IgM/IgG ELISA, antigen-capture ELISA)
- Virus isolation (limited to reference labs)
- Emerging technologies such as biosensors and next-generation sequencing are under development.
Molecular Detection: RT-PCR and Real-Time RT-PCR
- The gold standard for acute-phase diagnosis is reverse transcription polymerase chain reaction (RT-PCR) targeting conserved regions of the S segment RNA genome.
- Real-time RT-PCR assays provide rapid, sensitive, and quantitative detection of viral RNA from blood or serum during the viremic phase, typically within the first 7–10 days after symptom onset.
- Assays must be designed to detect diverse CCHFV strains due to high genetic variability; multiplex or pan-lineage primers improve coverage.
- Viral load quantification correlates with disease severity and prognosis.
Serological Testing: ELISA and Immunofluorescence Assays
- IgM antibodies appear approximately 5–7 days post-symptom onset; IgG antibodies develop later and persist for years, indicating past exposure.
- Commercially available IgM/IgG ELISAs are widely used for retrospective diagnosis and epidemiological studies but have limited utility in early acute infection due to delayed seroconversion.
- Antigen-capture ELISAs detect viral proteins in blood but are less commonly used than molecular methods due to lower sensitivity and availability constraints.
Virus Isolation and Culture
- Virus isolation in cell culture (e.g., Vero E6 cells) remains the definitive diagnostic method but is restricted to specialized BSL-4 laboratories due to biohazard risks.
- Isolation is time-consuming and not practical for routine diagnostics but valuable for research, strain characterization, and antiviral susceptibility testing.
Emerging Diagnostic Technologies
- Biosensor-based platforms employing electrochemical, optical, or lateral flow immunoassays offer promise for rapid point-of-care testing with high sensitivity/specificity, but require further validation before widespread use
- Next-generation sequencing (NGS) enables comprehensive viral genome analysis, aiding outbreak investigation, strain typing, and detection of novel variants; however, it is resource-intensive
Diagnostic Algorithm Recommendations
- Suspected cases should undergo immediate molecular testing by RT-PCR. If negative, but clinical suspicion remains high beyond day 7 of illness, serology should be performed.
- Combined molecular and serological testing improves diagnostic yield across different disease stages.
Treatment and Antiviral Therapy for Crimean-Congo Hemorrhagic Fever Virus (CCHFV)
Supportive Care: The Cornerstone of Management
- No specific antiviral therapy is currently approved; treatment focuses on intensive supportive care, including:
-Fluid resuscitation to maintain hemodynamic stability.
-Correction of electrolyte imbalances.
-Oxygen supplementation as needed.
-Management of bleeding with transfusions of platelets, fresh frozen plasma, and cryoprecipitate based on coagulation parameters.
-Organ support in intensive care units for severe cases with multi-organ failure or shock.
Ribavirin: The Most Widely Used Antiviral Agent
- Ribavirin is a broad-spectrum nucleoside analogue that inhibits viral RNA synthesis; it has demonstrated in vitro activity against CCHFV
- Observational studies from Turkey, Iran, and other endemic regions suggest ribavirin may reduce mortality if administered early in the disease course; however, evidence remains inconclusive due to a lack of randomized controlled trials.
- Ribavirin dosing regimens vary widely; intravenous administration may be preferred in severe cases, but availability is limited in many settings.
- Side effects include hemolytic anemia and teratogenicity requiring careful monitoring during therapy.
Investigational Antiviral Agents and Future Prospects
- Favipiravir, a viral RNA polymerase inhibitor approved for influenza in some countries, shows superior efficacy compared to ribavirin in animal models of CCHF by reducing viral replication and improving survival rates.
- Other nucleoside analogues (e.g., T705 derivatives), protease inhibitors targeting the OTU domain of L protein, host-targeted agents modulating endocytosis or lipid metabolism pathways are under preclinical evaluation.
- Combination therapies targeting multiple viral or host factors may enhance antiviral efficacy while reducing resistance risk.
Immunotherapy Approaches
- Passive immunization using convalescent plasma or hyperimmune globulin has been attempted with variable success; timing relative to disease onset appears critical for benefit.
- Monoclonal antibodies targeting glycoproteins Gn/Gc or accessory proteins like GP38 show protective effects in animal models but remain experimental without clinical approval yet.
Adjunctive Therapies
- Corticosteroids have been used empirically to modulate cytokine storm but carry risks of immunosuppression; no consensus exists on their routine use, given limited data
- Anti-coagulants are generally contraindicated during active hemorrhage but may be considered cautiously once bleeding stabilizes, if thrombotic complications arise.
Prevention and Control of Crimean-Congo Hemorrhagic Fever Virus (CCHFV)
Individual-Level Preventive Measures
- Avoidance of tick bites through:
-Use of insect repellents containing DEET or permethrin-treated clothing.
-Wearing long sleeves/pants when working outdoors in endemic areas.
-Regular body checks for ticks after outdoor activities with prompt removal using fine-tipped tweezers, minimizing tick saliva exposure risk.
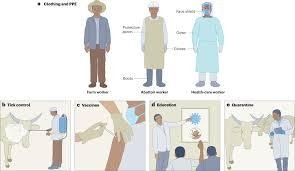
Occupational Safety Practices
- High-risk groups include farmers, abattoir workers, and veterinarians who should:
-Wear gloves, gowns, and eye protection when handling animals or animal products potentially infected with CCHFV.
-Implement safe slaughtering practices, minimizing blood splashes/exposure.
-Use appropriate personal protective equipment (PPE) during veterinary procedures involving blood/tissues from livestock known or suspected to be viremic
Healthcare Infection Prevention and Control (IPC)
- Early identification and isolation of suspected/confirmed cases are essential to prevent nosocomial transmission via contact with blood/body fluids or aerosol-generating procedures.
- Standard precautions plus contact/droplet precautions should be applied routinely; airborne precautions are recommended during aerosol-generating interventions such as intubation or suctioning.
- Proper disposal/decontamination of sharps and contaminated materials reduces the risk of healthcare worker infections.
Public Health Surveillance & Vector Control
- Integrated surveillance systems monitoring human cases alongside tick populations and animal reservoirs enable early warning of outbreaks and geographic expansion risk assessment.
- Tick control strategies include acaricide application on livestock pastures where feasible; pasture management reducing tick habitats can lower vector density, though ecological impacts must be considered carefully.
Community Education & Risk Communication
- Awareness campaigns targeting at-risk populations emphasize recognition of symptoms, tick bite prevention measures, safe handling practices around livestock/blood products, and early healthcare-seeking behavior.
Vaccine Development Status
- No licensed vaccine currently exists despite decades-long efforts; several candidates are under development, including:
-Inactivated whole-virus vaccines have been used historically in Eastern Europe with limited data on efficacy/safety profiles.
-Nucleic acid vaccines encoding glycoproteins showing promising immunogenicity in preclinical models requiring CD8+ T-cell responses for protection.
-Viral vector-based vaccines (e.g., MVA vectors expressing GPC).
-Multi-epitope subunit vaccines targeting NP/Gn/Gc/GP38 proteins aiming at broad immune coverage across diverse strains.
Conclusion
Crimean-Congo hemorrhagic fever virus is a genetically diverse and high-consequence agent that circulates in complex tick and vertebrate cycles. The virus causes considerable morbidity and mortality in humans. Since around 2015, there have been considerable advances in the molecular biology of the virus, its immune evasion strategies, immunopathogenesis, and epidemiology worldwide. This has led to the establishment of sophisticated animal models and the development of antiviral and vaccine approaches against the virus.
However, the clinical management of the disease is still essentially supportive in nature. The use of ribavirin is debatable, and no vaccine or antiviral drugs against the virus are yet available. There is a need for enhanced One Health surveillance for the virus, vector control measures, infection prevention and containment strategies, and accelerated evaluation of promising medical countermeasures against the increasing threat of the expanding tick-borne virus.
References
- Balinandi, S., Mulei, S., Whitmer, S., Nyakarahuka, L., Cossaboom, C., Shedroff, E., Morales-Betoulle, M., Krapiunaya, I., Tumusiime, A., Kyondo, J., Baluku, J., Namanya, D., Torach, C., Mutesi, J., Kiconco, J., Pimundu, G., Muyigi, T., Rowland, J., Nsawotebba, A., … Klena, J. (2024). Crimean-Congo hemorrhagic fever cases diagnosed during an outbreak of Sudan virus disease in Uganda, 2022–23. PLOS Neglected Tropical Diseases, 18. https://doi.org/10.1371/journal.pntd.0012595
- Balinandi, S., Whitmer, S., Mulei, S., Nyakarahuka, L., Tumusiime, A., Kyondo, J., Baluku, J., Mutyaba, J., Mugisha, L., Malmberg, M., Lutwama, J., Shoemaker, T., & Klena, J. (2021). Clinical and Molecular Epidemiology of Crimean-Congo Hemorrhagic Fever in Humans in Uganda, 2013–2019. The American Journal of Tropical Medicine and Hygiene, 106, 88–98. https://doi.org/10.4269/ajtmh.21-0685
- Belobo, J.-T. E., Kenmoe, S., Kengne-Ndé, C., Emoh, C. P. D., Bowo-Ngandji, A., Tchatchouang, S., Wobessi, J. N. S., Mikangue, C. A. M., Tazokong, H., Bebey, S. R. K., Noura, E. A., Ka’e, A., Simo, R. E. G., Modiyinji, A. F., Ngongang, D., Che, E., Kenfack, S., Nzukui, N. D., Adjia, N. A., … Njouom, R. (2021). Worldwide epidemiology of Crimean-Congo Hemorrhagic Fever Virus in humans, ticks,̶ and other animal species, a systematic review and meta-analysis. PLoS Neglected Tropical Diseases, 15. https://doi.org/10.1371/journal.pntd.0009299
- Bernard, C., Holzmuller, P., Bah, M. T., Bastien, M., Combes, B., Jori, F., Grosbois, V., & Vial, L. (2022). Systematic Review on Crimean–Congo Hemorrhagic Fever Enzootic Cycle and Factors Favoring Virus Transmission: Special Focus on France, an Apparently Free-Disease Area in Europe. Frontiers in Veterinary Science, 9. https://doi.org/10.3389/fvets.2022.932304
- Celina, S., Italiya, J., Tekkara, A. O., & Černý, J. (2025). Crimean-Congo haemorrhagic fever virus in ticks, domestic, and wild animals. Frontiers in Veterinary Science, 11. https://doi.org/10.3389/fvets.2024.1513123
- Dai, S., Dèng, F., Wáng, H., & Ning, Y. (2021). Crimean-Congo Hemorrhagic Fever Virus: Current Advances and Future Prospects of Antiviral Strategies. Viruses, 13. https://doi.org/10.3390/v13071195
- Eslava, M., Carlos, S., & Reina, G. (2024a). Crimean-Congo Hemorrhagic Fever Virus: An Emerging Threat in Europe with a Focus on Epidemiology in Spain. Pathogens, 13(9), 770. https://doi.org/10.3390/pathogens13090770
- Eslava, M., Carlos, S., & Reina, G. (2024b). Crimean-Congo Hemorrhagic Fever Virus: An Emerging Threat in Europe with a Focus on Epidemiology in Spain. Pathogens, 13. https://doi.org/10.3390/pathogens13090770
- Frank, M., Weaver, G., & Raabe, V. (2024a). Crimean-Congo Hemorrhagic Fever Virus for Clinicians—Diagnosis, Clinical Management, and Therapeutics. Emerging Infectious Diseases, 30, 864–873. https://doi.org/10.3201/eid3005.231648
- Frank, M., Weaver, G., & Raabe, V. (2024b). Crimean-Congo Hemorrhagic Fever Virus for Clinicians—Epidemiology, Clinical Manifestations, and Prevention. Emerging Infectious Diseases, 30, 854–863. https://doi.org/10.3201/eid3005.231647
- Frank, M., Weaver, G., & Raabe, V. (2024c). Crimean-Congo Hemorrhagic Fever Virus for Clinicians—Virology, Pathogenesis, and Pathology. Emerging Infectious Diseases, 30, 847–853. https://doi.org/10.3201/eid3005.231646
- Gholizadeh, O., Jafari, M., Zoobinparan, R., Yasamineh, S., Tabatabaie, R., Akbarzadeh, S., Amini, P., Eslami, M., Yousefi, B., & Dadashpour, M. (2022). Recent advances in treatment of Crimean-Congo hemorrhagic fever virus: A concise overview. Microbial Pathogenesis, 105657. https://doi.org/10.1016/j.micpath.2022.105657
- Hamidinejad, M., Ghaleh, E. G., Farzanehpour, M., Bolandian, M., & Dorostkar, R. (2021). Crimean-Congo hemorrhagic fever from the immunopathogenesis, clinical, diagnostic, and therapeutic perspective: A scoping review. Asian Pacific Journal of Tropical Medicine, 14, 254–265. https://doi.org/10.4103/1995-7645.315899
- Hawman, D., & Feldmann, H. (2018). Recent advances in understanding Crimean–Congo hemorrhagic fever virus. F1000Research, 7. https://doi.org/10.12688/f1000research.16189.1
- Hawman, D., & Feldmann, H. (2023). Crimean–Congo haemorrhagic fever virus. Nature Reviews. Microbiology, 21, 463–477. https://doi.org/10.1038/s41579-023-00871-9
- Hawman, D. W., & Feldmann, H. (2023). Crimean–Congo haemorrhagic fever virus. Nature Reviews Microbiology, 21(7), 463–477. https://doi.org/10.1038/s41579-023-00871-9
- Kortekaas, J., Vloet, R. P. M., McAuley, A. J., Shen, X., Bosch, B. J., De Vries, L., Moormann, R. J. M., & Bente, D. A. (2015). Crimean-Congo Hemorrhagic Fever Virus Subunit Vaccines Induce High Levels of Neutralizing Antibodies But No Protection in STAT1 Knockout Mice. Vector-Borne and Zoonotic Diseases, 15(12), 759–764. https://doi.org/10.1089/vbz.2015.1855
- Muzammil, K., Rayyani, S., Sahib, A. A., Gholizadeh, O., Sameer, H. N., Kazem, T. J., Mohammed, H. B., Kalajahi, H. G., Zainul, R., & Yasamineh, S. (2024). Recent Advances in Crimean-Congo Hemorrhagic Fever Virus Detection, Treatment, and Vaccination: Overview of Current Status and Challenges. Biological Procedures Online, 26. https://doi.org/10.1186/s12575-024-00244-3
- Ozdarendeli, A. (2023). Crimean–Congo Hemorrhagic Fever Virus: Progress in Vaccine Development. Diagnostics, 13. https://doi.org/10.3390/diagnostics13162708
- Rodriguez, S., Hawman, D., Sorvillo, T., O’Neal, T., Bird, B., Rodriguez, L., Bergeron, É., Nichol, S., Montgomery, J., Spiropoulou, C., & Spengler, J. (2022). Immunobiology of Crimean-Congo hemorrhagic fever. Antiviral Research, 105244. https://doi.org/10.1016/j.antiviral.2022.105244
- Serretiello, E., Astorri, R., Chianese, A., Stelitano, D., Zannella, C., Folliero, V., Santella, B., Galdiero, M., Franci, G., & Galdiero, M. (2020). The emerging tick-borne Crimean-Congo Haemorrhagic Fever Virus: A narrative review. Travel Medicine and Infectious Disease, 101871. https://doi.org/10.1016/j.tmaid.2020.101871
- Tlenchiyeva, T., Myrzakhmetova, B., Zhapparova, G., Bisenbayeva, K., Toitanova, A., & Kutumbetov, L. (2023). CRIMEAN-CONGO HEMORRHAGIC FEVER: REVIEW OF THE EPIDEMIOLOGICAL SITUATION, DISTRIBUTION AREA, VIRUS CHARACTERISTICS, DIAGNOSIS, AND DISEASE PREVENTION. Biosafety and Biotechnology. https://doi.org/10.58318/957-5702-2024-17-6-22
- Türk, Y. K., & Kacı, F. (2024). Crimean-Congo Haemorrhagic Fever Virus: From Genomic Insights to Control Strategies. Journal of the Institute of Science and Technology. https://doi.org/10.21597/jist.1412021

its really very helpfull i hope you will be successfull in your life