Complement refers to a set of serum proteins that cooperates with both the innate and the adaptive immune systems to eliminate blood and tissue pathogens.
Complement components can be classified into seven functional categories:
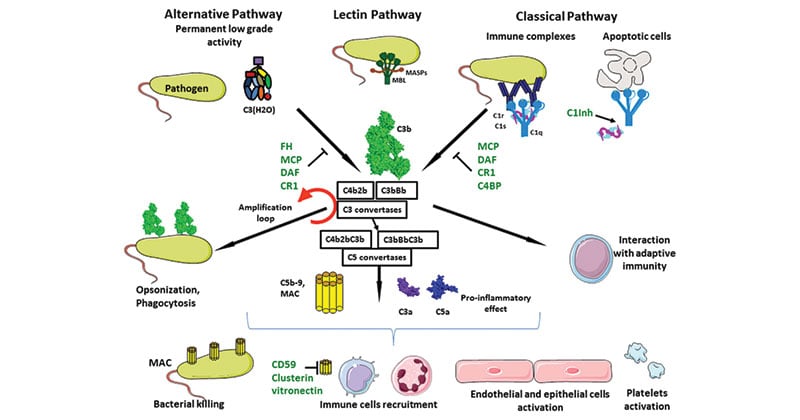
- Initiator complement components: These proteins initiate their respective complement cascades by binding to particular soluble or membrane-bound molecules. Once bound to their activating ligand, they undergo conformational alterations resulting in changes in their biological activity. The C1q complex, Mannose-Binding Lectin (MBL) etc. are examples of initiator complement components.
- Enzymatic mediators: Several complement components, (e.g., C1r, C1s, MASP2, and factor B) are proteolytic enzymes that cleave and activate other members of the complement cascade. Some of these proteases are activated by binding to other macromolecules and undergoing a conformational change. Others are inactive until cleaved by another protease enzyme and are thus termed zymogens: proteins that are activated by proteolytic cleavage. The two enzyme complexes that cleave complement components C3 and C5, respectively, are called the C3 and C5 convertases and are of central importance in the complement cascades.
- Membrane-binding components or opsonins: Upon activation of the complement cascade, several proteins are cleaved into two fragments, each of which then takes on a particular role. For C3 and C4, the larger fragments, C3b and C4b, serve as opsonins, enhancing phagocytosis by binding to microbial cells and serving as binding targets for phagocytic cells bearing receptors for C3b or C4b.
- Inflammatory mediators: Some small complement fragments act as inflammatory mediators. These fragments enhance the blood supply to the area in which they are released, by binding to receptors on endothelial cells lining the small blood vessels and inducing an increase in capillary diameter. They also attract other cells to the site of tissue damage. Because such effects can be harmful in excess, these fragments are called anaphylatoxins, meaning substances that cause anaphylaxis (“against protection”). Examples include C3a, C5a, and C4a.
- Membrane attack proteins: The proteins of the membrane attack complex (MAC) insert into the cell membranes of invading microorganisms and punch holes that result in lysis of the pathogen. The complement components of the MAC are C5b, C6, C7, C8, and multiple copies of C9. Membrane attack proteins are most important in lysis of foreign bodies once complement cascade is activated. MAC is the cytolytic end product of the complement cascade; it forms a transmembrane channel, which causes osmotic lysis of the target cell.
- Complement receptor proteins: A complement receptor is a receptor of the complement system, a part of the mediated innate immune system. Complement receptors are responsible for detecting pathogens by mechanisms not mediated by antibodies. Receptor molecules on cell surfaces bind complement proteins and signal-specific cell functions. For example, some complement receptors such as CR1 bind to complement components such as C3b on the surface of pathogens, triggering phagocytosis of the C3-bound pathogen. Binding of the complement component C5a to C5aR receptors on neutrophils stimulates neutrophil degranulation and inflammation. Complement receptors are named with “R,” such as CR1, CR2, and C5aR.
- Regulatory complement components: Host cells are protected from unintended complement-mediated lysis by the presence of membrane-bound as well as soluble regulatory proteins. These regulatory proteins include factor I, which degrades C3b, and Protectin, which inhibits the formation of the MAC on host cells.
