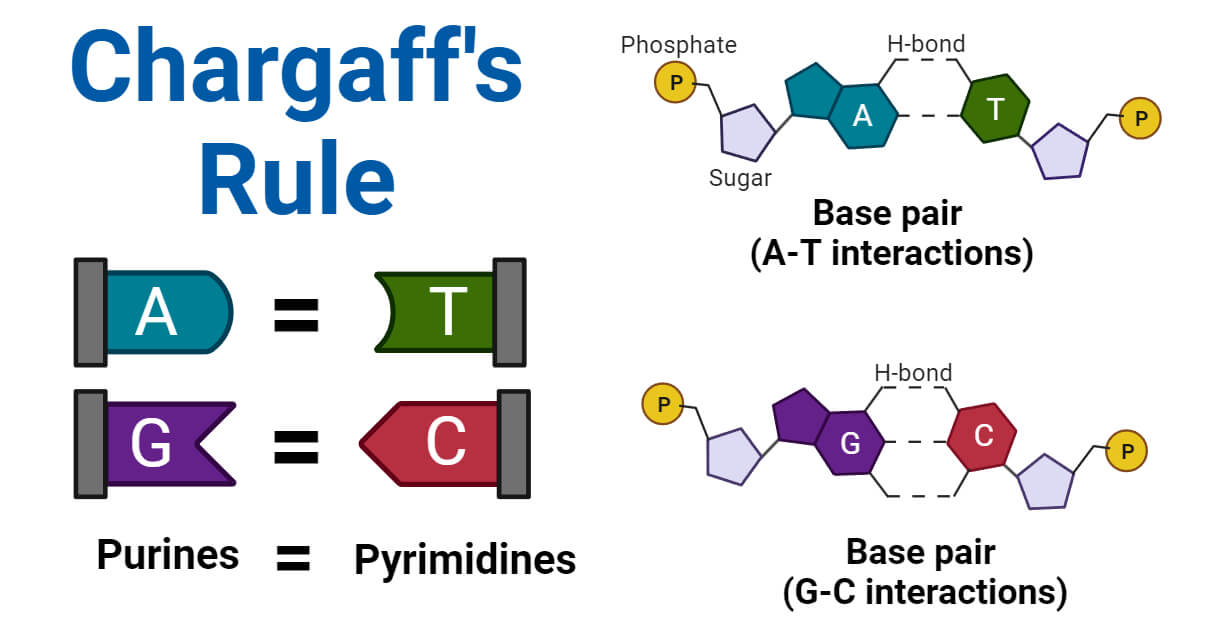
• Chargaff's Rules elucidate the fundamental principles of DNA base composition and pairing. • DNA is composed of four nucleotide bases: adenine (A), thymine (T), cytosine (C), and guanine (G). • Chargaff's First Rule underscores complementary base pairing: A pairs with T, and C pairs with G. • This base pairing is critical for DNA's double-stranded structure and the stability of the DNA molecule. • Chargaff's Second Rule reveals consistent ratios of A to T and C to G in DNA strands. • Equal ratios of complementary base pairs contribute to the stability and structure of the DNA double helix.
What is Chargaff’s Rules?
Erwin Chargaff was an Austrian-American biochemist born in 1905 and known for pioneering molecular genetics research. He made significant contributions to our understanding of DNA and RNA composition. Chargaff’s meticulous work on DNA base composition laid the foundation for Chargaff’s Rules.
The significance of Chargaff’s Rules in genetics and DNA research
Chargaff’s Rules are fundamental principles that provide insights into the structure and behavior of DNA. They outline the specific pairing rules between the four nucleotide bases in DNA, forming the basis of DNA’s double-stranded structure.
These rules are crucial in understanding DNA replication and genetic inheritance and have been instrumental in the discovery of the DNA double helix, fundamentally shaping the field of molecular genetics and modern biology.
Base Composition in DNA
DNA, or deoxyribonucleic acid, is the hereditary material in most living organisms. It consists of nucleotide units, each comprising a sugar molecule (deoxyribose), a phosphate group, and one of four nitrogenous bases.
These bases are crucial for the genetic code and include adenine (A), thymine (T), cytosine (C), and guanine (G). The unique arrangement of these bases along the DNA molecule forms the genetic instructions necessary for an organism’s growth, development, and functioning.
Overview of the Four Nucleotide Bases
Adenine (A): Adenine is one of the four nucleotide bases in DNA and pairs with thymine (T) through two hydrogen bonds.
Thymine (T): Thymine is a pyrimidine base that pairs with adenine (A) in DNA through the hydrogen above bonds.
Cytosine (C): Cytosine, another pyrimidine base, forms a pairing with guanine (G) through three hydrogen bonds.
Guanine (G): Guanine, a purine base, pairs with cytosine (C) through the mentioned hydrogen bonds.
Chargaff’s First Rule
Chargaff’s First Rule, observed by biochemist Erwin Chargaff, states that in a DNA molecule, the amount of adenine (A) equals the amount of thymine (T), and the amount of cytosine (C) equals the amount of guanine (G).
This implies a fundamental symmetry and consistency in the ratios of these base pairs within a DNA molecule.
The Principle of Complementary Base Pairing
Chargaff’s observation led to the understanding of complementary base pairing. Adenine (A) always pairs with thymine (T), and cytosine (C) pairs with guanine (G).
This pairing is based on hydrogen bonding: A-T pairs are held together by two hydrogen bonds, while three hydrogen bonds hold C-G pairs together. In contrast, the complementary base pairing is vital for the stability and structure of the DNA double helix.
Adenine (A) Pairing with Thymine (T), and Cytosine (C) Pairing with Guanine (G)
Adenine (A) and thymine (T) form a complementary base pair, and their pairing is represented as A-T.
Cytosine (C) and guanine (G) form another complementary base pair, and their pairing is represented as C-G.
These pairings adhere to the principle of complementary base pairing, playing a central role in DNA’s structure and its ability to replicate accurately during cell division.
Chargaff’s Second Rule
Chargaff’s Second Rule stipulates that in any DNA molecule, the ratio of adenine (A) to thymine (T) and cytosine (C) to guanine (G) is approximately equal.
This holds in the DNA of various species, regardless of their origin or complexity.
The significance lies in the fact that this equal proportion supports the principle of complementary base pairing, which forms the fundamental structure of DNA.
Equal Proportions of Complementary Base Pairs in a DNA Molecule
The equal proportions of A to T and C to G in a DNA molecule contribute to the stability and structural integrity of the DNA double helix.
The base pairs adenine-thymine and cytosine-guanine form complementary pairs, which means they have specific hydrogen bond interactions.
A Pairs with T via two hydrogen bonds, and C pairs with G via three hydrogen bonds, ensuring a consistent structure that assists in genetic replication and maintenance.
Implications of This Rule for DNA Structure and Replication
This uniformity in base pair ratios is central to the helical structure of DNA. The A-T and C-G base pairs fit together neatly within the double helix, maintaining a uniform width.
During DNA replication, each strand acts as a template for the creation of a new complementary strand precisely because of this base pairing. It ensures that the genetic information is accurately preserved and transmitted during cellular division and reproduction.
Significance and Applications of Chargaff’s Rules
Role in Understanding DNA Structure and Function
Chargaff’s Rules significantly contributed to our understanding of DNA’s structure and functioning. The equal proportions of complementary base pairs laid the foundation for comprehending the stable structure of the DNA double helix.
At the same time, the mechanism of base pairing is critical for genetic information storage and retrieval.
Contributions to the Discovery of the DNA Double Helix by Watson and Crick
Chargaff’s Rules played a pivotal role in the discovery of the DNA double helix by Watson and Crick.
The complementary base pair ratios provided crucial clues that guided Watson and Crick in constructing their accurate model of the DNA molecule. It subsequently earned them the Nobel Prize in Physiology or Medicine in 1962.
Importance in DNA Sequencing and Genetic Research
Chargaff’s Rules are foundational in DNA sequencing and genetic research. Understanding base pair ratios and the rules of complementary base pairing is essential for decoding the genetic code.
In contrast, identifying genetic variations and conducting molecular studies is crucial for advancements in medicine, genetics, forensics, and various scientific disciplines.
Modern Understanding and Extensions of Chargaff’s Rules
Modern research has affirmed and refined Chargaff’s observations, providing deeper insights into the nuances of base composition and its relation to genetic processes. Ongoing studies continue to uncover subtleties in DNA structure, replication, and gene regulation, enhancing our understanding of genetics and molecular biology.
How Chargaff’s Observations Are Incorporated into Modern Molecular Biology?
Chargaff’s Rules remain fundamental in modern molecular biology. They are incorporated into various methodologies and technologies used for DNA analysis, sequencing, and manipulation.
Understanding base pairing principles is crucial in the design and interpretation of experiments, leading to a more comprehensive understanding of genetic mechanisms and molecular processes in living organisms.
Conclusion
Chargaff’s Rules have been a cornerstone in understanding the fundamental structure of DNA and its role in genetics. The equal proportions of adenine-thymine and cytosine-guanine underscore the stability of the DNA double helix and are pivotal in genetic replication.
With enduring relevance and applications in modern molecular biology, Chargaff’s Rules remain an integral foundation for genetic research and advancements in various scientific domains.
Reference
- https://www.ck12.org/c/biology/base-pairing/lesson/chargaffs-base-pairing-rules-advanced-bio-adv/
- http://epgp.inflibnet.ac.in/epgpdata/uploads/epgp_content/S001174BS/P001199/M010849/ET/1479280871P4M14eTextSept22.pdf
- https://groups.molbiosci.northwestern.edu/holmgren/Glossary/Definitions/Def-C/Chargaff%27s_rule.html
- https://bio.libretexts.org/Courses/Sacramento_City_College/BIOL_440%3A_General_Microbiology_(Hughes)/02%3A_Week_2/03%3A_Biochemistry_of_the_Genome_(Part_A)/3.01%3A_Structure_and_Function_of_DNA
- http://henge.bio.miami.edu/mallery/lec/150/gene/chargaff.htm
- https://www.mun.ca/biology/scarr/Chargaffs_Rules.html
- https://www.jbc.org/article/S0021-9258(20)61522-8/fulltext
