A centrifuge is a device used to separate components of a mixture on the basis of their size, density, the viscosity of the medium, and the rotor speed.
- The centrifuge is commonly used in laboratories for the separation of biological molecules from a crude extract.
- In a centrifuge, the sample is kept in a rotor that is rotated about a fixed point (axis), resulting in strong force perpendicular to the axis.
- There are different types of centrifuge used for the separation of different molecules, but they all work on the principle of sedimentation.
Relative Centrifugal Force (RCF)
- Relative centrifugal force is the measure of the strength of rotors of different types and sizes.
- This is the force exerted on the contents of the rotor as a result of the rotation.
- RCF is the perpendicular force acting on the sample that is always relative to the gravity of the earth.
- The RCF of the different centrifuge can be used for the comparison of rotors, allowing the selection of the best centrifuge for a particular function.
The formula to calculate the relative centrifugal force (RCF) can be written as:
RCF (g Force)= 1.118 × 10-5 × r × (RPM)2
where r is the radius of the rotor (in centimeters), and RPM is the speed of the rotor in rotation per minute.
Centrifuge Rotors
Rotors in centrifuges are the motor devices that house the tubes with the samples. Centrifuge rotors are designed to generate rotation speed that can bring about the separation of components in a sample. There are three main types of rotors used in a centrifuge, which are:
1. Fixed angle rotors

Figure: Fixed angle rotors. Image Source: Beckman Coulter, Inc.
- These rotors hold the sample tubes at an angle of 45° in relation to the axis of the rotor.
- In this type of rotor, the particles strike the opposite side of the tube where the particles finally slide down and are collected at the bottom.
- These are faster than other types of rotors as the pathlength of the tubes increases.
- However, as the direction of the force is different from the position of the tube, some particles might remain at the sides of the tubes.
2. Swinging bucket rotors/ Horizontal rotors

Figure: Swinging bucket rotors/ Horizontal rotors. Image Source: Beckman Coulter, Inc.
- Swinging bucket rotors hold the tubes at an angle of 90° as the rotor swings as the process is started.
- In this rotor, the tubes are suspended in the racks that allow the tubes to be moved enough to acquire the horizontal position.
- In this type of rotors, the particles are present along the direction or the path of the force that allows the particles to be moved away from the rotor towards the bottom of the tubes.
- Because the tubes remain horizontal, the supernatant remains as a flat surface allowing the deposited particles to be separated from the supernatant.
3. Vertical rotors

Figure: Vertical rotors. Image Source: Beckman Coulter, Inc.
- Vertical rotors provide the shortest pathlength, fastest run time, and the highest resolution of all the rotors.
- In vertical rotors, the tubes are vertical during the operation of the centrifuge.
- The yield of the rotor is not as ideal as the position of the tube doesn’t align with the direction of the centrifugal force.
- As a result, instead of settling down, particles tend o spread towards the outer wall of the tubes.
- These are commonly used in isopycnic and density gradient centrifugation.
Types of Centrifuge

1. Benchtop centrifuge

Figure: Thermo Scientific™ Sorvall™ ST 8 Small Benchtop Centrifuge. Image Source: Thermo Scientific.
- Benchtop centrifuge is a compact centrifuge that is commonly used in clinical and research laboratories.
- It is driven by an electric motor where the tubes are rotated about a fixed axis, resulting in force perpendicular to the tubes.
- Because these are very compact, they are useful in smaller laboratories with smaller spaces.
- Different variations of benchtop centrifuges are available in the market for various purposes.
- A benchtop centrifuge has a rotor with racks for the sample tubes and a lid that closes the working unit of the centrifuge.
2. Continuous flow centrifuge
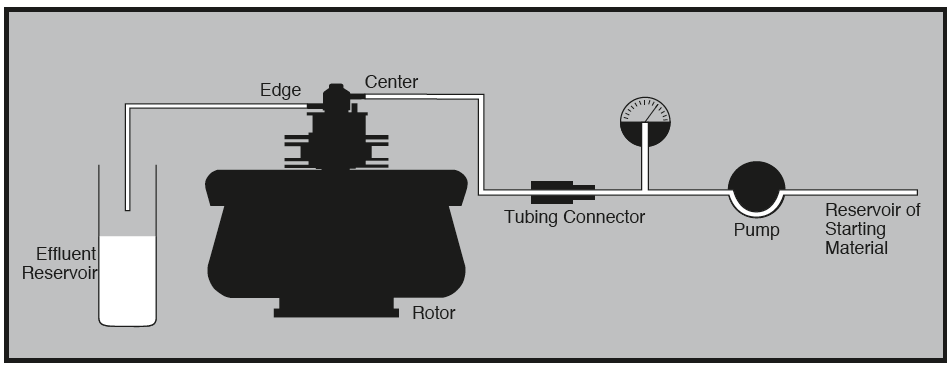
Figure: Equipment arrangement during continuous flow centrifugation. Image Source: Beckman Coulter, Inc.
- Continuous flow centrifuge is a rapid centrifuge that allows the centrifugation of large volumes of samples without affecting the sedimentation rates.
- This type of centrifuge allows the separation of a large volume of samples at high centrifugal force, thus removing the tedious part of emptying and filling the tubes with each cycle.
- They have a shorter pathlength which facilitates the process of pelleting out the solid part out of the supernatant, thus maintaining the speed of the process.
- They also have larger capacities which saves time as the sample doesn’t have to be load and unloaded over and over again like in traditional centrifuges.
- Up to 1 liter of samples can be centrifuged by this centrifuge at a time period of 4 hours or less.
3. Gas centrifuge
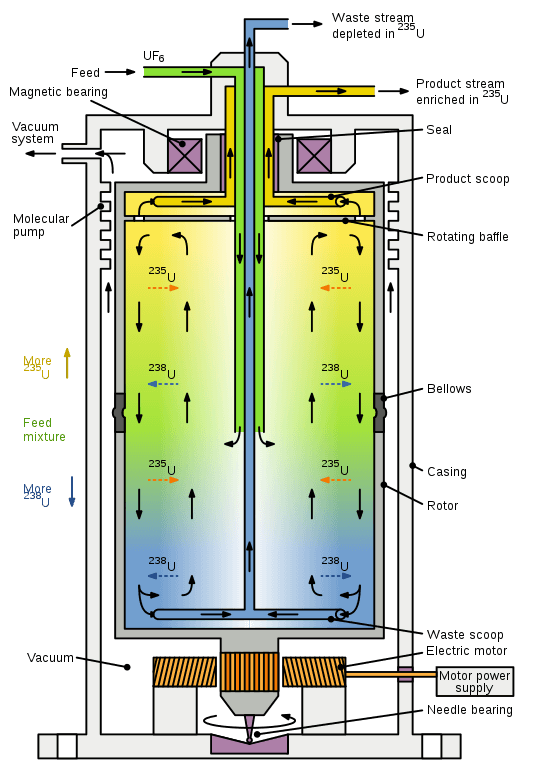
Figure: Diagram of a gas centrifuge with countercurrent flow, used for separating isotopes of uranium. Image Source: Wikipedia (Inductiveload).
- A gas centrifuge is a centrifuge explicitly used for the separation of gases based on heir isotopes.
- This centrifuge is based on the same principle of centrifugal force as all other centrifuges where the molecules are separated on the basis of their masses.
- This centrifuge is used mainly for the extraction and separation of uranium -235 and uranium-238.
- The gas centrifuge works on eh design of the continuous flow of gas in and out of the centrifuge, unlike other centrifuge working on batch processing.
- These centrifuges are arranged in cascades so that the gases are separated into two units based on their isotopes and then are passed onto the next centrifuge for further processing.
- Gas centrifuges have replaced other gaseous diffusion methods as they provide a yield of higher concentration of the gases than the previous techniques.
4. Hematocrit centrifuge

Figure: AHN myLab® Hematocrit Centrifuge. Image Source: AHN Biotechnologie GmbH.
- Hematocrit centrifuges are specialized centrifuges used for the determination of volume fraction of erythrocytes (RBCs) in a given blood sample.
- This centrifuge provides hematocrit values that can be used for testing in biochemistry, immunity, blood test, and other general clinical tests.
- Hematocrit centrifuges may be used to help diagnose blood loss, polycythemia (an elevation of the erythrocyte count to above-normal levels), anemia, bone marrow failure, leukemia, and multiple myeloma.
- The microhematocrit centrifuge quickly attains speeds of 11,000 rpm and RCFs of up to 15,000 g to spin tube samples.
- The components of a hematocrit centrifuge are similar to that of the benchtop centrifuge, but this centrifuge is specialized for the use of blood samples.
5. High-speed centrifuge

Figure: Avanti JXN-30 Series High-speed centrifuge. Image Source: Beckman Coulter, Inc.
- High-speed centrifuge, as the name suggests, is the centrifuge that can be operated at somewhat larger speeds.
- The speed of the high-speed centrifuge can range from 15,000 to 30,000 rpm.
- The high-speed centrifuge is commonly used in more sophisticated laboratories with the biochemical application and requires a high speed of operations.
- High-speed centrifuges are provided with a system for controlling the speed and temperature of the process, which is necessary for the analysis of sensitive biological molecules.
- The high-speed centrifuges come with different adapters to accommodate the sample tubes of various sizes and volumes.
- All three types of rotors can be used for the centrifugation process in these centrifuges.
6. Low-speed centrifuge

Figure: Low-speed centrifuges – ScanSpeed 406 and ScanSpeed 416. Image Source: LaboGene.
- Low-speed centrifuges are the traditional centrifuges that are commonly used in laboratories for the routine separation of particles.
- These centrifuges operate at the maximum speed of 4000-5000 rpm.
- These are usually operated under room temperature as they are not provided with a system for controlling the speed or temperature of the operation.
- Swinging bucket and fixed angle type of rotors can be used in these centrifuges.
- These are easy and compact centrifuges that are ideal for the analysis of blood samples and other biological samples.
- The low-speed centrifuge works on the same principle as all other centrifuges, but the application is limited to the separation of simpler solutions.
7. Microcentrifuge

Figure: Microfuge 16 and Microfuge 20. Image Source: Beckman Coulter, Inc.
- Microcentrifuges are the centrifuges used for the separation of samples with smaller volumes ranging from 0.5 to 2 µl.
- Microcentrifuges are usually operated at a speed of about 12,000-13,000 rpm.
- This is used for the molecular separation of cell organelles like nuclei and DNA and phenol extraction.
- Microcentrifuges, also termed, microfuge, use sample tubes that are smaller in size when compared to the standard test tubes used in larger centrifuges.
- Some microcentrifuges come with adapters that facilitate the use of larger tubes along with the smaller ones.
- Microcentrifuges with temperature controls are available for the operation of temperature-sensitive samples.
8. Refrigerated centrifuges

Figure: Allegra 64R Refrigerated Benchtop Centrifuge. Image Source: Beckman Coulter, Inc.
- Refrigerated centrifuges are the centrifuges that are provided with temperature control ranging from -20°C to -30°C.
- A different variation of centrifuges is available that has the system of temperature control which is essential for various processes requiring lower temperatures.
- Refrigerated centrifuges have a temperature control unit in addition to the rotors and racks for the sample tubes.
- These centrifuges provide the RCF of up to 60,000 xg that is ideal for the separation of various biological molecules.
- These are typically used for collecting substances that separate rapidly like yeast cells, chloroplasts, and erythrocytes.
- The chamber of refrigerated centrifuge is sealed off from the outside to meet the conditions of the operations.
9. Ultracentrifuges

Figure: Ultracentrifuges. Image Source: Beckman Coulter, Inc.
- Ultracentrifuges are the centrifuges that operate at extremely high speeds that allow the separation of much smaller molecules like ribosomes, proteins, and viruses.
- It is the most sophisticated type of centrifuge that allows the separation of molecules that cannot be separated with other centrifuges.
- Refrigeration systems are present in such centrifuges that help to balance the heat produced due to the intense spinning.
- The speed of these centrifuges can reach as high as 150,000 rpm.
- It can be used for both preparative and analytical works.
- Ultracentrifuges can separate molecules in large batches and in a continuous flow system.
- In addition to separation, ultracentrifuges can also be used for the determination of properties of macromolecules like the size, shape, and density.
10. Vacuum centrifuge/ Concentrators

Figure: Savant™ SpeedVac™ SPD120 Vacuum Concentrator and Kits. Image Source: Thermo Scientific.
- Vacuum centrifuge utilizes the centrifugal force, vacuum and heat to speed up the laboratory evaporation of samples.
- These centrifuges are capable of processing a large number of samples (up to 148 samples at a time).
- This type of centrifuge is used in chemical and biological laboratories for the effective evaporation of solvents present in samples, thus concentrating the samples.
- These are commonly used in high throughput laboratories for samples that might have a large number of solvents.
- A rotary evaporator is used to remove the unnecessary solvents and eliminate solvent bumping.
- The centrifuge works by lowering the pressure of the chamber, which also decreases the boiling point of the samples.
- This causes the solvents to be evaporated, concentrating the particles to be separated.
Reference
- Wilson, K., Walker, J. (2018). Principles and Techniques of Biochemistry and Molecular Biology. Eighth edition. Cambridge University Press: New York.
- Serwer, BACTERIOPHAGES: SEPARATION OF, Editor(s): Ian D. Wilson, Encyclopedia of Separation Science, Academic Press, 2000, Pages 2102-2109,
- ISBN 9780122267703, https://doi.org/1016/B0-12-226770-2/07381-6. (http://www.sciencedirect.com/science/article/pii/B0122267702073816)
- https://www.beckman.com/resources/fundamentals/principles-of-centrifugation/rotor-types
- https://www.beckman.com/centrifuges/rotors/vertical-angle
- https://www.beckman.com/resources/reading-material/application-notes/principles-of-continuous-flow-centrifugation
- http://origin.who.int/medical_devices/innovation/hospt_equip_8.pdf
- https://www.news-medical.net/Life-Science-and-Laboratory/Microcentrifuges
- https://www.mybiosource.com/learn/testing-procedures/rate-zonal-centrifugation/

amazing well explained
This is very helpful 👍
I love it, easy understanding.
This is going to get me full marks in my activity 😄
I am interested in using this principle in separating salt from sea water.
thank you very much
so much more useful
I love it,it help
super duper nice