In the treatment and control of infectious diseases, especially when caused by pathogens that are often drug-resistant, susceptibility (sensitivity) testing is used to select effective antimicrobial drugs. Susceptibility testing is not usually indicated when the susceptibility reactions of a pathogen can be predicted, for example:
– Proteus species are generally resistant to nitrofurantoin and tetracyclines,
– S. pyogenes is usually susceptible to penicillin,
– K. pneumoniae is generally ampicillin-resistant,
– Anaerobes are susceptible to metronidazole.
Susceptibility tests must never be performed on commensal organisms or contaminants because this would mislead the clinician and could result in the patient receiving ineffective and unnecessary antimicrobial therapy, causing possible side effects and resistance to other potentially pathogenic organisms.
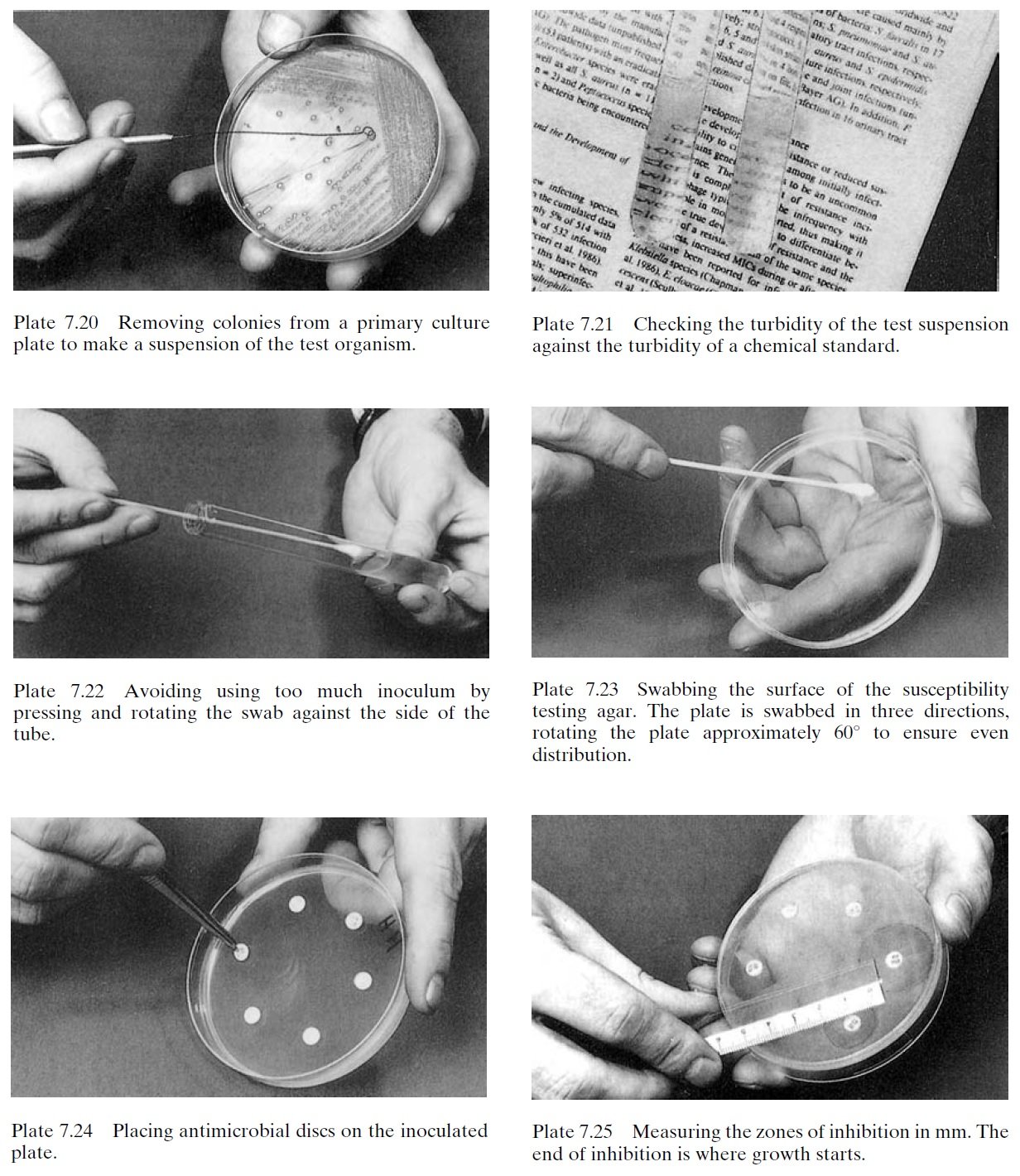
Figure: Antimicrobial Susceptibility Testing (AST) Procedure; Source: Basic laboratory procedures in clinical bacteriology, WHO, 2nd edition, 2003
Susceptibility testing techniques
Laboratory antimicrobial susceptibility testing can be performed using:
– A dilution technique
– A disc diffusion technique.
Dilution susceptibility tests
Manual or semi-automated dilution susceptibility tests are performed in Microbiology Reference Laboratories for epidemiological purposes or when a patient does not respond to treatment thought to be adequate, relapses while being treated, or when there is immunosuppression. Dilution techniques measure the minimum inhibitory concentration (MIC). They can also be used to measure the minimum bactericidal concentration (MBC) which is the lowest concentration of antimicrobial required to kill bacteria. A dilution test is carried out by adding dilutions of an antimicrobial to a broth or agar medium. A standardized inoculum of the test organism is then added. After overnight incubation, the MIC is reported as the lowest concentration of antimicrobial required to prevent visible growth. By comparing the MIC value with known concentrations of the drug obtainable in serum or other body fluids, the likely clinical response can be assessed.
Disc diffusion susceptibility tests
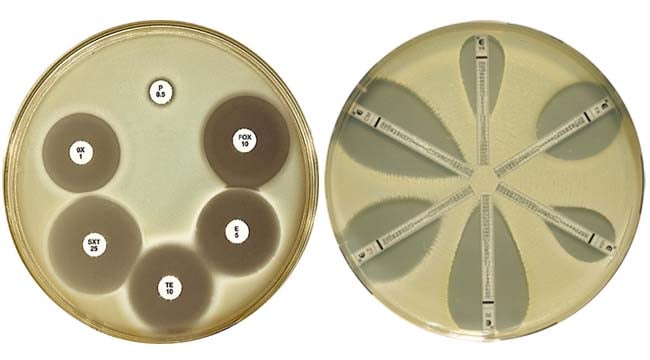
Disc diffusion techniques are used by most laboratories to test routinely for antimicrobial susceptibility. A disc of blotting paper is impregnated with a known volume and appropriate concentration of an antimicrobial, and this is placed on a plate of susceptibility testing agar uniformly inoculated with the test organism. The antimicrobial diffuses from the disc into the medium and the growth of the test organism is inhibited at a distance from the disc that is related (among other factors) to the susceptibility of the organism. Strains susceptible to the antimicrobial are inhibited at a distance from the disc whereas resistant strains have smaller zones of inhibition or grow up to edge of the disc. For clinical and surveillance purposes and to promote reproducibility and comparability of results between laboratories, WHO recommends the (NCCLS) modified Kirby-Bauer disc diffusion technique.
Kirby-Bauer NCCLS modified disc diffusion technique
The validity of this carefully standardized technique depends on, for each defined species, using discs of correct antimicrobial content, an inoculum which gives confluent growth, and a reliable Mueller Hinton agar. The test method must be followed exactly in every detail. After incubation at 35° C for 16–18 hours, zone sizes are measured and interpreted using NCCLS standards. These are derived from the correlation which exists between zone sizes and MICs. The NCCLS Kirby-Bauer technique should only be used for well-evaluated bacterial species. It is not suitable for bacteria that are slow-growing, need special nutrients, or require CO2 or anaerobic incubation.
Stokes disc diffusion technique
In this disc technique, both the test and control organisms are inoculated on the same plate. The zone sizes of the test organism are compared directly with that of the control. This method is not as highly standardized as the Kirby-Bauer technique and is used in laboratories particularly when the exact amount of antimicrobial in a disc cannot be guaranteed due to difficulties in obtaining discs and storing them correctly or when the other conditions required for the Kirby-Bauer technique cannot be met. One way laboratories performing the Stokes technique could change to a technique comparable to the WHO recommended Kirby-Bauer technique is to use highly stable Rosco Diagnostica antibiotic tablets (Neo-Sensitabs) instead of less stable paper discs.
Limitations of antimicrobial susceptibility tests
Susceptibility tests measure antimicrobial activity against bacteria under laboratory conditions (in vitro activity), not in the patient (in vivo activity). It cannot be assumed therefore, that an antimicrobial that kills or prevents an organism from growing in vitro will be a successful treatment.
Selecting appropriate antimicrobial treatment also involves considering the patient’s clinical condition, any underlying condition (e.g. liver or kidney disease), the type and site of the infection, any history of drug hypersensitivity, age of the patient and whether a patient is pregnant. It is also necessary to know the activity of the different drugs including their rates of absorption, diffusion in the tissues, metabolism, excretion, and also possible toxicity and effects on the patient’s normal microbial flora. The cost and availability of a drug will also need to be considered.

Your notes are really nice, simplified and understandable. Thank you so much 🙏🙏🙏🙏
Estima Sagar Aryal, se podria utilizar los pocillos hechos en la placa de agar para colocar alli el antimicrobiano (extracto de planta) y de esa manera realizar el ensayo de difusion en agar, medir el halo de inhibicion? lo encontre este metodo como parte del metodo de difusion ya que tendria el mismo principio del disco difusion.
Yes, you can make a hole in the agar plate, place an antimicrobial (plant extract) there and thus perform the agar diffusion assay.
very useful informetion
Very helpful information thanks
Hello sir,
I am a medical laboratory student. I found it very important. Carry on!!!
Thank you so much
Hy Sir ! I am your great fan. I am microbiology student and i read your every article which contain alots of new , interesting and fabolus information related to microbiology. We pray for your good health. Carry on
Very good
Very useful information for the Microbiology students and technical people who are working in microbiology laboratories in healthcare. If some information about Fungus which are medically important for human in your blog, I will love to read because I have reported lots of fungus like Aspergillus spp., Alternaria species, Mucor spp in critical care patients who are immunocompromised.
Very useful information for the Microbiology students and technical people who are working in microbiology laboratories in healthcare. If some information about Fungus which are medically important for human in your blog, I will love to read because I have reported lots of fungus like Aspergillus spp., Alternaria species, Mucor spp in critical care patients who are immunocompromised.