Amphotericin B belongs to the polyene class of antifungal agents. Polyenes are broad-spectrum antifungal, biochemically characterized by having macrolide (made of a lactone ring with conjugated double bonds that is attached to one or more sugar moieties) along with a hydroxyl group, making it amphiphilic.
- This amphiphilic nature helps them to target, bind, and disrupt the fungal cell membrane (made of ergosterol) by forming pores.
- This compound is derived from the actinobacteria Streptomyces spp.
- Other than amphotericin B, polyenes consist of nystatin and natamycin.
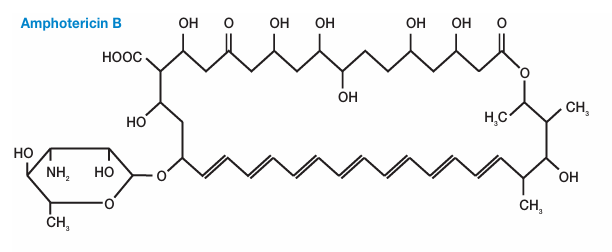
Structure of Amphotericin B
Amphotericin B was discovered from the fermentation broth of Streptomyces nodosus culture M4575, which was isolated from a soil sample of the Orinoco basin in Venezuela.
Source: https://pubchem.ncbi.nlm.nih.gov/compound/Ergosterol
- AmB structure is characterized as having a macrocyclic lactone ring with a series of conjugated double bonds and is referred to as a hydrophobic “tail” or polyene tail. Polyhydroxyl chain runs parallel on the opposite side, consisting of mycosamine group and hydroxyl group, and is referred to as the hydrophilic “head”.
- The amphipathic nature makes it poorly soluble in water. For oral administration, AmB was combined with sodium deoxycholate, which formed a micelle in 5% glucose solvent. This solution didn’t destroy the constitution of AmB, and could be given intravenously.
- The preparation with sodium deoxycholate was named Fungizone, which is also commonly referred to as AmB deoxycholate.
- AmB deoxycholate is used to treat systemic mycoses like disseminated histoplasmosis and mucormycosis, and is the first-line treatment of cryptococcal meningitis in combination with flucytosine.
- Even though the antifungal agent has a high toxic effect on the host, it is still relevantly used as a fungicide (more than 70 years after its discovery) and has the lowest incidence of resistance.
Source: https://pubchem.ncbi.nlm.nih.gov/compound/Ergosterol
Mechanism of Action of Amphotericin B
Pore formation
- Amphotericin B specifically targets the ergosterol (lipid found in fungal cell membrane), altering the permeability of the membrane, forming trans-membrane pores through which ions (K+ and Mg²+), nutrients, and cellular contents seep out and consequently cause the cellular death of fungi.
- AmB specifically targets the ergosterol because the structure of ergosterol enables better binding of AmB’s hydrophobic tail than that of cholesterol in mammalian cell membranes.
- After the polyene tail binds to the ergosterol, the hydrophilic polyol of AmB forms aqueous channels, which are stabilized by intermolecular H-bonding between the amino and carboxyl groups of hydrophilic heads of adjacent polyene molecules.
- In the presence of ergosterol, AmB forms a wide (about 0.46nm) full pore (that consists of 2 ring complexes of polyene).
- Ergosterol has a high cellular significance due to its ability to regulate membrane proteins, cell division, cell signaling, membrane fluidity, and endocytosis.
The interaction of Amphotericin B with ergosterol involves three different interactive forces:
- Van der Waal’s force: When both the ergosterol and amphotericin B are oriented in parallel or co-planar fashion, such interactions take place.
- H-bond: Interaction takes place between the 3β-OH group of ergosterol and the mycosamine group of Amphotericin B.
- π-π electronic bonding: Interaction occurs between the ergosterol side chain and the polyene tail of Amphotericin B.
Surface adsorption model
- This model suggests that the high affinity of AmB towards ergosterol leads to its binding with it, adsorbing it and altering the membrane organization, but in the process, it does not form pores.
Sterol Sponge model
- Anderson et al. described this mode of action, which involves AmB forming parallel aggregates at the membrane that act as a sterol sponge and sequester the ergosterol.
Reactive Oxidation
- Many studies have provided evidence of AmB inducing oxidative stress, which ultimately leads to protein carbonylation, lipid peroxidation, DNA damage, and subsequent death of fungal cells (apoptosis).
- Various testimonies of evidence, like that of C. albicans producing catalase and superoxide dismutase (SOD) during administration of AmB, leading to K+ leakage from cells. Another observation involves the production of spermidine, spermine, and putrescine by C. albicans to counteract and reduce the effect of Reactive Oxygen Species (ROS) whenever AmB is given.
- Some suggest the origin of oxidative damage by AmB may come from auto-oxidation of AmB itself.
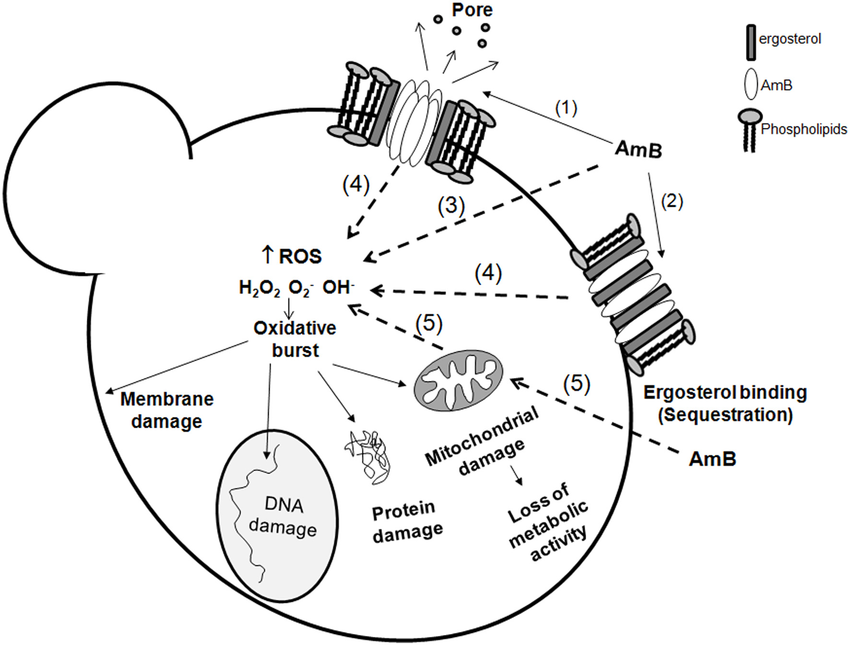
Source: https://doi.org/10.3389/fmicb.2012.00286
Drug Resistance of Amphotericin B
Sterol alteration
- The recurrent mechanism of resistance is the alteration of sterol composition via mutation of ERG (responsible for the biosynthesis pathway of ergosterol).
- In fungi like C. albicans, loss of function of ERG11& ERG3 leads to other sterol byproducts like lanosterol, 4,14-dimethyl-zymosterol, or eburicol. Substitution in ERG11 and reduced function of ERG5 have also yielded similar byproducts in AmB-resistant C. albicans.
- The AmB resistance observed in C. neofromans was attributed to a mutation in EGR2 that led to its inactivation. A sterol-independent mechanism was also observed in AmB-resistant C. neoformans.
Catalase production
- The AmB triggers stress-related mechanisms in fungal cells via a surge of a strong oxidative burst.
- Exponential increase in catalase production by AmB-resistant Aspergillus terreus has equipped them to better tolerate the oxidative stress caused by AmB and thus prevent cell death.
Cell wall alteration
- An increased level of 1,3-α-glucan fraction in the cell wall has been described as one of the factors for the increased resistance mechanism in A. flavus by Seo et. al., 1999.
- An enlarged cell wall in C. tropicalis, which was due to an excessive level of 1,3-β-glucan, can inhibit the penetration of AmB, leading to resistance.
Chaperone in creating or maintaining the AmB resistance
- Hsp90 is a central stress response factor because it aids proteins that help the cell adapt to stress. The possible ways it could lead to a resistance mechanism are:
-Its disruption may lead to the expression of new genetic variation, and chances of some being highly tolerant to the antifungal agent will be very likely.
-Hsp90 can stabilize the unstable mutated protein (some of which can provide resistance) and conserve this functionality.
- Elevated expression of the Hsp70 gene in A. flavus has been a significant contribution to its resistance. Inhibition of Hsp70 consequently made it susceptible.
Toxicity of Amphotericin B
Even though AmB is considered to be highly selective towards ergosterol, administration of high dosage can result in its interaction with the cholesterol in mammalian cell membranes, which leads to non-selective cleavage.
Two types of toxicity are prominent:
Acute toxicity: The microbial origin of amphotericin B is recognized by TLR2 and CD14, and the immune system triggers a proinflammatory cytokine production. This results in acute infusion-related adverse effects like fevers, nausea, vomiting, hypertension, etc.
Chronic toxicity: Intense exposure of AmB on renal cells via the Low-Density Lipoprotein (LDL) receptor mediates endocytosis of the cell.
It also causes vasoconstriction in afferent renal arterioles.
Conclusion
Despite exhibiting host toxicity, Amphotericin B is very prominent as a first-line drug for clinical treatment of systemic mycoses. The lipid conjugate of AmB, like AmB lipid complex (ABLC) has shown less nephrotoxicity than AmB deoxycholate; similarly, liposomal AmB (L-AmB) has shown less infusion-related reaction. The amide conjugate of AmB exhibits higher antifungal activity and low acute toxicity in vivo. These derivatives of AmB have a promising future in clinical implementation, along with the persistent advantage of its lower incidence of resistance.
References
- Anderson, T.M., Clay, M.C., Cioffi, A.G., Diaz, K.A., Hisao, G.S., Tuttle, M.D., Nieuwkoop, A.J., Comellas, G., Maryum, N. & Wang, S. (2014). Amphotericin forms an extramembranous and fungicidal sterol sponge. Nat. Chem. Biol., 10, 400.
- Cao, Y., Zhu, Z., Chen, X., Yao, X., Zhao, L., Wang, H., Yan, L., Wu, H. Chai, Y. & Jiang, Y. (2013). Effect of amphotericin B on metabolic profiles pf C. albicans. J proteome Res. 12, 2921-2932.
- Carolus, H., Pierson, S. Lagrou, K. & Van Dijck, P. (2020) Amphotericin B and Other Polyenes-Discovery, Clinical Use, Mode of Action and Drug Resistance. J. Fungi, 6, 321.
- Greenwood, D., Barer, M., Slack, R. & Irving, W. (2012). Medical microbiology (18th edition). Churchill Livingstone Elsevier.
- Ngece, K., Ntondini, T. L., Khwaza, V., Paca, A.M. & Aderibigbe, B.A. (2024). Polyene-Based Derivatives with Antifungal Activities. Pharmaceutics, 16, 1065. doi: 10.3390/pharmaceutics16081065
- Seo, K., Akiyoshi, H. & Ohnishi, Y. (1999). Alteration of cell wall composition leads to amphotericin B resistance in Aspergillus flavus. Microbiol. Immunol., 43, 1017-1025.
- Skol-Anderson, M.L., Brajtburg, K., Medoff, G. (1986). Amphotericin B-induced oxidative damage and killing of C. albicans. J. Infect. Dis., 154, 76-83.
- Willey, J.M., Sherwood, L.M. Woolverton, C.J. (2008). Prescott, Harley and Klein’s microbiology (7th edition). McGraw-Hill.
