The complex network of biochemical processes known as metabolism allows organisms to synthesize vital chemicals for growth and survival and extract energy from foods. The phosphoketolase pathway, present in some microbes, stands out as an intriguing and adaptable mechanism within this enormous landscape of metabolic pathways. This route greatly aids their metabolic diversity and adaptation to specific environmental situations.
Scientists may decipher the fundamental principles that govern microbial metabolism and exploit its potential for various applications by comprehending the complexities of the phosphoketolase pathway.
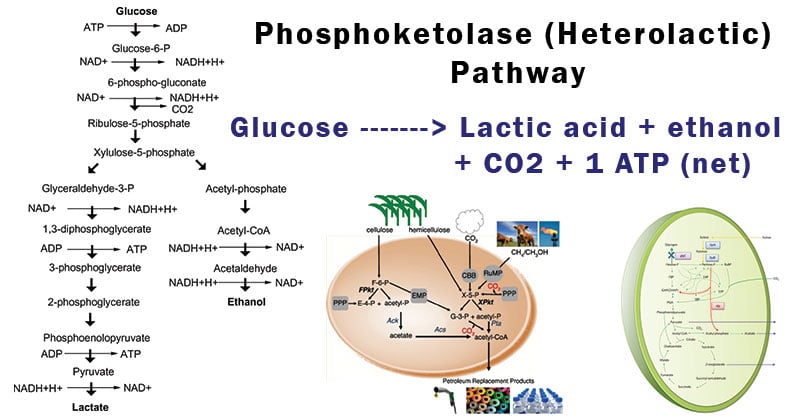
The phosphoketolase pathway, also known as the heterolactic pathway, involves the breakdown of carbohydrates, particularly pentose sugars, into important metabolic intermediates.
- This pathway is characterized by a series of enzymatic reactions that convert xylulose-5-phosphate, a key intermediate in pentose sugar metabolism, into acetyl-CoA, a fundamental building block for many cellular processes.
- The pathway is named after the enzyme phosphoketolase, which catalyzes the initial step in the pathway.
- Understanding the phosphoketolase pathway’s stages might help one better understand how microorganisms undergo metabolic changes.
- The mechanism starts with the transformation of xylulose-5-phosphate into glyceraldehyde-3-phosphate and acetyl-phosphate and then uses different pathways to transform glyceraldehyde-3-phosphate into either pyruvate or erythrose-4-phosphate.
- These responses demonstrate how easily bacteria may change their metabolic processes in response to varying food availability and environmental factors.
Furthermore, the phosphoketolase pathway has drawn a lot of interest because of the possible applications it may have. This route presents prospects for sustainable bio-based production, human health, and environmental remediation, from the generation of biofuels and bioplastics to its function in gut health and carbon flux management. Understanding the phosphoketolase pathway broadens our knowledge of microbial metabolism and provides new opportunities.
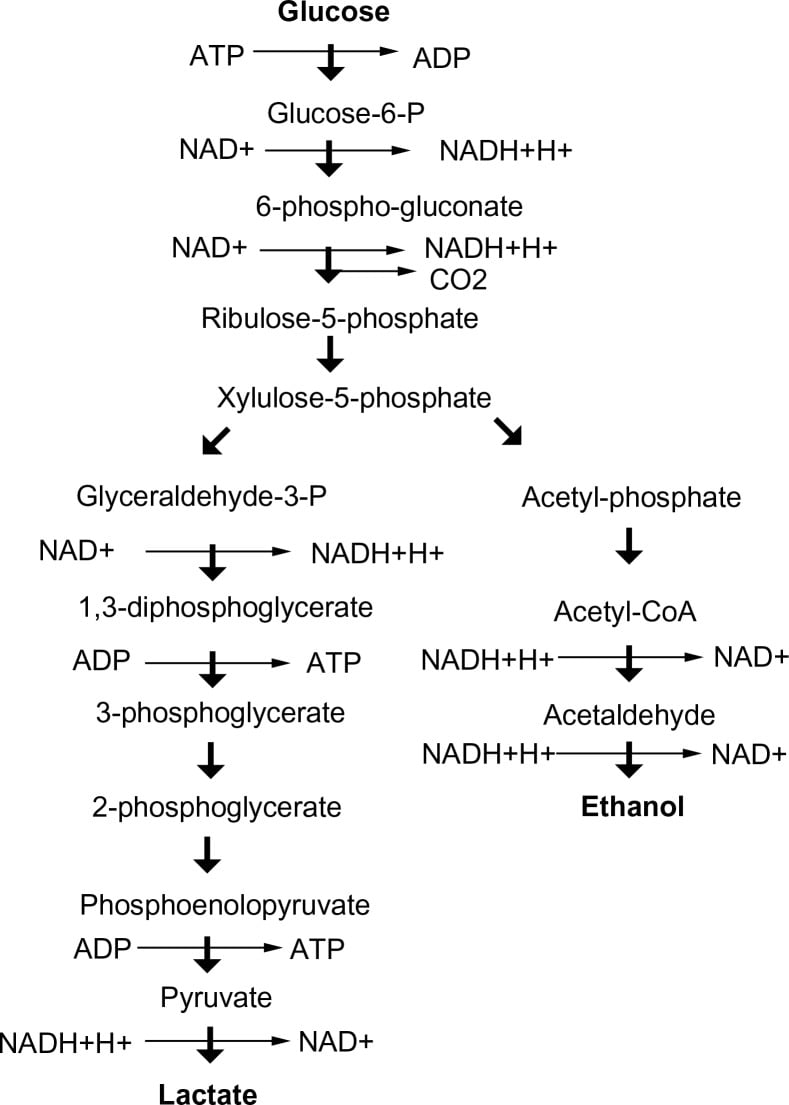
Steps in Phosphoketolase Pathway
The phosphoketolase pathway is a metabolic pathway that occurs in various bacteria, yeast, and archaea. It involves the breakdown of carbohydrates, particularly pentoses, into different metabolic intermediates, such as acetyl-phosphate, acetyl-CoA, and erythrose-4-phosphate.
The pathway consists of three key steps:
1. Conversion of Xylulose-5-Phosphate
- The initial step in the phosphoketolase pathway involves the conversion of xylulose-5-phosphate, a pentose phosphate, into glyceraldehyde-3-phosphate (GAP) and acetyl-phosphate.
- This reaction is catalyzed by the enzyme phosphoketolase, which cleaves xylulose-5-phosphate into these two products.
- The acetyl-phosphate can further be converted into acetyl-CoA, a versatile molecule involved in various metabolic processes.
- Reaction: Xylulose-5-phosphate + Pi → Glyceraldehyde-3-phosphate + Acetyl-phosphate
2. Glyceraldehyde-3-Phosphate Conversion
- The produced glyceraldehyde-3-phosphate from the first step can follow two alternative routes.
- In the first route, glyceraldehyde-3-phosphate is directly converted into pyruvate through the action of the enzyme glyceraldehyde-3-phosphate dehydrogenase.
- This step generates ATP through substrate-level phosphorylation, contributing to energy production in the cell.
- In the second route, glyceraldehyde-3-phosphate is converted into erythrose-4-phosphate, which can be utilized for the synthesis of nucleotides and aromatic amino acids.
- Glyceraldehyde-3-phosphate (GAP) can follow two alternative routes.
a) Conversion to Pyruvate
- GAP is directly converted into pyruvate by the enzyme glyceraldehyde-3-phosphate dehydrogenase (GAPDH).
- This step generates ATP through substrate-level phosphorylation.
- Reaction: Glyceraldehyde-3-phosphate + NAD+ + Pi → Pyruvate + NADH + H+ + ATP
b) Conversion to Erythrose-4-Phosphate
- GAP can be converted into erythrose-4-phosphate (E4P) by the enzyme transaldolase.
- E4P is an important intermediate for nucleotide and aromatic amino acid synthesis.
- Reaction: Glyceraldehyde-3-phosphate + Fructose-6-phosphate → Erythrose-4-phosphate + Xylulose-5-phosphate
3. Formation of Acetyl-CoA
- In the final step of the pathway, the generated acetyl-phosphate can be converted into acetyl-CoA through the action of acetate kinase and phosphate acetyltransferase enzymes.
- Acetyl-CoA is a central metabolite involved in various biochemical pathways, including the tricarboxylic acid (TCA) cycle, fatty acid synthesis, and amino acid metabolism.
- Thus, the phosphoketolase pathway contributes to the production of acetyl-CoA, which serves as a crucial building block for cellular processes.
- Reaction: Acetyl-phosphate + ADP → Acetate + ATP
- Acetate + CoA → Acetyl-CoA + CoA
Phosphoketolase Pathway Overall Reaction
The overall reaction that characterizes the phosphoketolase pathway is as follows:
- Xylulose-5-phosphate + 2ADP + 2Pi → Acetyl-CoA + erythrose-4-phosphate + 2ATP + 2H2O
- The transformation of xylulose-5-phosphate into acetyl-CoA, erythrose-4-phosphate, ATP, and water is highlighted in this process.
- It demonstrates the metabolic pathway’s flexibility by producing both compounds high in energy and biosynthesis intermediates.
- The initial substrate, xylulose-5-phosphate (X5P), is broken down by phosphoketolase into two products: The transformation of X5P into acetyl-phosphate and the subsequent transformation of acetyl-phosphate into acetyl-CoA results in the formation of two molecules of acetyl-CoA.
- Erythrose-4-phosphate (E4P): As mentioned in step 2, E4P is created through the alternate method of glyceraldehyde-3-phosphate conversion.
- The process results in the exchange of phosphate groups in addition to the production of acetyl-CoA and E4P:
- a) Two molecules of ATP are produced: one during the conversion of acetyl-phosphate to acetyl-CoA, and the other during the conversion of glyceraldehyde-3-phosphate to pyruvate (assuming that route is used).
- b) Both the conversion of ATP to ADP and the conversion of X5P to GAP need two molecules of Pi (inorganic phosphate).
- Two ADP molecules are also converted into ATP as part of the process, which symbolizes the resynthesis of ATP for cellular energy.
Applications of Phosphoketolase Pathway
The phosphoketolase pathway has several important applications in various fields:
1. Industrial Production
- The route is used to produce important chemicals in biotechnological operations.
- For example, it has been used to make platform chemicals like acetyl-CoA, a molecule that may be used to make high-value compounds like biofuels and plastics.
- The phosphoketolase pathway offers a different and sustainable approach to producing these substances by converting pentose sugars, such as xylulose-5-phosphate, into acetyl-CoA.
2. Probiotics and Gut Health
- Some probiotic bacteria that live in the human gut have the phosphoketolase pathway.
- Through the phosphoketolase route, these bacteria may use dietary pentose carbohydrates, which are inefficiently metabolized by other gut microorganisms.
- They can fill ecological niches and improve the general health of the gut microbiota because of this capacity.
- Additionally, the process produces short-chain fatty acids like acetic acid, which might provide the host with energy and improve intestinal health.
3. Carbon Flux Regulation
- The control of carbon flow in microbial metabolism is greatly influenced by the phosphoketolase pathway.
- It offers a different pathway for the digestion of carbohydrates, especially pentose sugars that may be present in the environment.
- Because of their adaptability, microbes can efficiently use various carbon sources and adjust to shifting nutritional circumstances.
- Understanding how the phosphoketolase pathway is regulated and modulated might help with metabolic engineering techniques that maximize the synthesis of desired chemicals by microbes.
4. Bioremediation
- Due to their diverse metabolic capabilities, several bacteria with the phosphoketolase pathway may break down complex chemical molecules, including aromatic compounds.
- This characteristic has potential uses in bioremediation, where these bacteria may be used to remove toxins and pollutants from the environment.
Conclusion
A remarkable metabolic route that enables organisms to adapt and survive in many settings is the phosphoketolase pathway. Its capacity to transform pentose sugars into acetyl-CoA and other crucial intermediates emphasizes the role it plays in biosynthesis and energy generation.
The pathway’s potential for a variety of biotechnological and environmental applications is further shown by its uses in industrial production, gut health, carbon flux management, and bioremediation.
The phosphoketolase pathway is a perfect example of how incredible and complicated metabolic pathways are in microbial metabolism, to sum up.
Its research not only clarifies the variety of metabolic processes but also offers chances for ground-breaking biotechnological and environmental science applications.
References
- Phosphoketolase – https://www.sciencedirect.com/topics/agricultural-and-biological-sciences/phosphoketolase
- Heterolactic (Phosphoketolase) Pathway – http://www.biosciencenotes.com/heterolactic-phosphoketolase-pathway/
- Phosphoketolase (Heterolactic) Pathway – https://microbenotes.com/phosphoketolase-heterolactic-pathway/
- Özcan, Emrah, et al. “A genome-scale metabolic network of the aroma bacterium Leuconostoc mesenteroides subsp. cremoris.” Applied Microbiology and Biotechnology 103 (2019): 3153-3165.
- Årsköld, Emma, et al. “Phosphoketolase pathway dominates in Lactobacillus reuteri ATCC 55730 containing dual pathways for glycolysis.” Journal of bacteriology 190.1 (2008): 206-212.
- Difference Between Homolactic and Heterolactic Fermentation – https://www.differencebetween.com/difference-between-homolactic-and-heterolactic-fermentation/
- Burgé, Grégoire, et al. “Relationships between the use of Embden Meyerhof pathway (EMP) or Phosphoketolase pathway (PKP) and lactate production capabilities of diverse Lactobacillus reuteri strains.” Journal of Microbiology 53.10 (2015): 702-710.

Thank you Sagar.
Can you say some examples of specific lactic acid bacteria
Common lactic acid bacteria are Lactobacillus, Leuconostoc, Pediococcus, Lactococcus, and Streptococcus.
What is the net gain of ATP from this pathway?