The pentose phosphate pathway is a metabolic pathway parallel to glycolysis which generates NADPH and pentoses (5-carbon sugars) as well as ribose 5-phosphate.
The pentose phosphate pathway is also called the phosphogluconate pathway or hexose monophosphate shunt. While it involves glucose oxidation, its primary role is anabolic rather than catabolic. It is an important pathway that generates precursors for nucleotide synthesis and is especially important in red blood cells (erythrocytes).
Pentose Phosphate Pathway participates in the breakdown of glucose and is crucial in the production of reducing equivalents and significant intermediates required for cellular biosynthesis. The PPP is necessary to support nucleotide synthesis, preserve cellular redox balance, and guard against oxidative stress. In this essay, we shall examine the reactions, results, phases, and importance of the pentose phosphate pathway, focusing on its function in erythrocytes.
Location of the Pentose Phosphate Pathway
Pentose Phosphate Pathway occurs in the cytosol of liver cells, adrenal cortex, and lactating mammary glands. Cytosol is the fluid part of the cytoplasm. In plants, most steps take place in plastids.
Reactions of the Pentose Phosphate Pathway
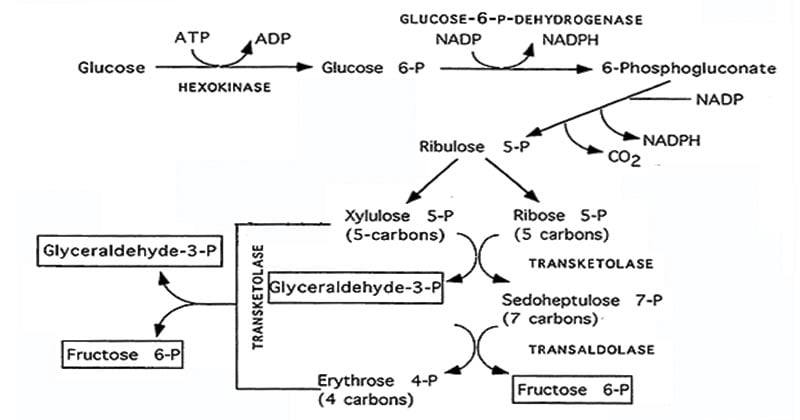
The PPP consists of two distinct phases: the oxidative and non-oxidative phases. These phases work in coordination to generate reducing power in the form of NADPH and produce ribose 5-phosphate (R5P), which serves as a precursor for nucleotide synthesis.
1. Oxidative Phase of Pentose Phosphate Pathway
- The oxidative phase is the first phase of the Pentose Phosphate Pathway (PPP) and is primarily responsible for the generation of NADPH.
- It starts with the conversion of glucose-6-phosphate (G6P) to 6-phosphogluconolactone, catalyzed by the enzyme glucose-6-phosphate dehydrogenase (G6PDH).
- This reaction generates the first molecule of NADPH by transferring electrons from G6P to NADP+.
- Next, 6-phosphogluconolactone is hydrolyzed into 6-phosphogluconate by the enzyme 6-phosphogluconolactonase.
- Finally, 6-phosphogluconate dehydrogenase converts 6-phosphogluconate to ribulose-5-phosphate (Ru5P) while generating the second molecule of NADPH.
- This series of reactions not only generates NADPH but also results in the production of Ru5P, which serves as an important precursor for nucleotide synthesis.
The oxidative phase of the PPP primarily occurs in the cytoplasm of most cells and is regulated by the enzyme G6PDH. The activity of G6PDH is tightly regulated by factors such as the availability of NADP+ and the levels of reactive oxygen species (ROS). G6PDH activity is essential for maintaining the balance between NADPH production and the cellular demand for reducing equivalents.
2. Non-Oxidative Phase of Pentose Phosphate Pathway
- In the non-oxidative phase of the PPP, different sugar phosphates are converted into one another, which results in the synthesis of significant metabolites and the creation of ribose-5-phosphate (R5P).
- Depending on cellular requirements, this step gives the pathway flexibility by permitting the use of intermediates for glycolysis or other metabolic pathways.
- The ribose-5-phosphate isomerase enzyme converts ribose-5-phosphate (R5P) from ribose-5-phosphate (Ru5P), which initiates the non-oxidative phase.
- R5P is a crucial precursor for nucleotide synthesis and is essential for cellular functions like the synthesis of DNA and RNA as well as ATP and GTP.
- Interconversion of sugar phosphates occurs through a series of reactions catalyzed by the enzymes transketolase and transaldolase.
- These enzymes facilitate the transfer of carbon units between different sugar phosphates, such as R5P, xylose-5-phosphate, and sedoheptulose-7-phosphate.
- This interconversion allows the cell to balance the production of R5P for nucleotide synthesis with the utilization of intermediates for other metabolic pathways.
- The non-oxidative phase of the PPP plays a crucial role in connecting carbohydrate metabolism with nucleotide biosynthesis.
- It enables cells to adapt their metabolic fluxes based on the cellular demand for nucleotides and other metabolites.
Therefore, NADPH is produced during the oxidative phase of the PPP, supplying reducing equivalents for a number of cellular functions. The interconversion of sugar phosphates during the non-oxidative phase results in the creation of R5P, an essential precursor for the synthesis of nucleotides. Together, these phases support biosynthesis, uphold cellular redox equilibrium, and fulfill the requirements of nucleotide synthesis.
The overall reaction of the pentose phosphate pathway
3 Glucose-6-P + 6 NADP+→ 3 ribulose-5-P + 3 CO2 + 6 NADPH
3 Ribulose-5-P → 2 xylulose-5-P + Ribose-5-P
2 Xylulose-5-P + Ribose-5-P → 2 fructose-6-P + Glyceraldehyde-3-P
Outcomes of the Pentose Phosphate Pathway
The Pentose Phosphate Pathway (PPP) produces two major outcomes: the generation of NADPH and the production of ribose-5-phosphate (R5P). These outcomes play essential roles in cellular metabolism and are critical for various cellular processes.
1. Generation of NADPH
- NADPH is a vital product of the oxidative phase of the PPP. It serves as a key reducing equivalent in numerous cellular reactions.
- NADPH is required for maintaining the cellular redox balance, as it acts as a reducing agent in various biosynthetic processes. It is involved in the synthesis of fatty acids, cholesterol, and other important metabolites.
- One of the significant roles of NADPH is its involvement in protecting cells against oxidative stress.
- Reactive oxygen species (ROS) are generated as byproducts of cellular metabolism and can cause oxidative damage to cells if not properly controlled.
- NADPH plays a crucial role in the regeneration of reduced glutathione, an important antioxidant molecule that neutralizes ROS.
- This redox cycle between glutathione and NADPH helps maintain cellular redox homeostasis and protects cells from oxidative damage.
- In erythrocytes, the PPP is particularly important for NADPH generation. Erythrocytes lack mitochondria and rely on the PPP as their primary source of NADPH.
- NADPH in erythrocytes is utilized to maintain the reduced state of glutathione, which protects these cells from oxidative stress and ensures their survival.
2. Production of Ribose-5-Phosphate
- The PPP also plays a crucial role in the production of R5P, a five-carbon sugar phosphate. R5P is an essential precursor for nucleotide synthesis.
- Nucleotides are the building blocks of DNA and RNA, and they are also involved in energy transfer (ATP and GTP) and cellular signaling processes.
- The non-oxidative phase of the PPP is responsible for the production of R5P.
- Through a series of enzymatic reactions catalyzed by transketolase and transaldolase, different sugar phosphates are interconverted, ultimately leading to the synthesis of R5P.
- This ensures a steady supply of R5P for nucleotide biosynthesis, supporting cellular growth, DNA replication, and RNA synthesis.
- The production of R5P through the PPP is particularly important in rapidly dividing cells, such as cells in the bone marrow responsible for hematopoiesis, where a high demand for nucleotides exists.
- Additionally, in situations of increased cellular growth or proliferation, such as during tissue regeneration or in cancer cells, the PPP becomes crucial for providing an adequate supply of R5P for nucleotide synthesis.
Therefore, the Pentose Phosphate Pathway generates NADPH, which is vital for maintaining the cellular redox balance, protecting against oxidative stress, and supporting biosynthetic processes. The production of R5P through the PPP ensures a steady supply of this precursor for nucleotide synthesis, supporting cellular growth, DNA replication, and RNA synthesis. These outcomes of the PPP play critical roles in cellular metabolism and have implications for various physiological processes, including the protection of erythrocytes from oxidative damage and the support of cellular proliferation and growth.
Significance of the Pentose Phosphate Pathway
The Pentose Phosphate Pathway (PPP) holds significant importance in cellular metabolism due to its crucial roles in maintaining redox balance, protecting against oxidative stress, and supporting biosynthetic processes. The pathway’s significance is particularly evident in various cell types, including erythrocytes.
1. Redox Balance and Protection Against Oxidative Stress
- One of the primary roles of the PPP is to generate NADPH, which serves as a key-reducing equivalent in numerous cellular reactions.
- NADPH plays a vital role in maintaining the cellular redox balance, ensuring that the ratio of reduced to oxidized species remains optimal for cellular function.
- NADPH is particularly important in protecting cells against oxidative stress. Reactive oxygen species (ROS), such as superoxide radicals and hydrogen peroxide, are natural byproducts of cellular metabolism and can cause damage to cells if not properly neutralized.
- NADPH plays a critical role in the regeneration of reduced glutathione, a potent antioxidant that scavenges ROS and prevents cellular damage.
- Erythrocytes heavily rely on the PPP for NADPH production and redox balance maintenance.
- These cells lack mitochondria and cannot generate ATP via oxidative phosphorylation.
- Therefore, the PPP becomes their primary source of NADPH. NADPH in erythrocytes is utilized to maintain the reduced state of glutathione, protecting these cells from oxidative stress and ensuring their survival.
2. Nucleotide Synthesis
In order to promote biosynthetic processes, such as the production of fatty acids, cholesterol, and other significant metabolites, the PPP is crucial. These biosynthetic processes require reducing power, which is provided by NADPH, which is produced during the oxidative phase of the PPP.
A crucial step needed for the synthesis of nucleotides, ribose-5-phosphate (R5P), is also directly produced by the PPP. For the production of DNA and RNA, as well as for cellular functions involving energy transfer (ATP and GTP), and signaling, nucleotides are necessary building blocks.
- The production of R5P through the non-oxidative phase of the PPP ensures a steady supply of this precursor for nucleotide biosynthesis.
- In rapidly dividing cells, such as those in the bone marrow responsible for hematopoiesis, the PPP becomes crucial for providing an adequate supply of R5P to support the high demand for nucleotides.
- Furthermore, in situations of increased cellular growth or proliferation, such as during tissue regeneration or in cancer cells, the PPP plays a vital role in supplying R5P for nucleotide synthesis.
Understanding the significance of the PPP provides valuable insights into cellular metabolism and homeostasis. It highlights the pathway’s roles in maintaining redox balance, protecting against oxidative stress, and supporting biosynthetic processes, with particular relevance to the vital functions of erythrocytes and the production of nucleotides necessary for cellular growth and proliferation.
Significance of the PPP in Erythrocytes
- Erythrocytes, or red blood cells, heavily rely on the PPP to maintain their structure and function.
- These cells lack mitochondria and cannot generate ATP via oxidative phosphorylation. As a result, the PPP serves as their primary source of NADPH.
- NADPH is necessary to maintain the reduced state of glutathione, which protects erythrocytes from oxidative damage caused by reactive oxygen species.
- Without sufficient NADPH production, erythrocytes become vulnerable to hemolysis and premature destruction.
- The PPP in erythrocytes also plays a crucial role in regulating the levels of reactive oxygen species, contributing to their overall oxidative stress resistance.
- Moreover, erythrocytes require a constant supply of ribose-5-phosphate for nucleotide synthesis. Nucleotides are essential for DNA and RNA synthesis, as well as for maintaining the pool of ATP and GTP in erythrocytes.
- The PPP ensures a steady supply of ribose-5-phosphate, enabling efficient nucleotide synthesis and supporting the high turnover rate of erythrocytes.
- This is particularly important in situations such as increased erythropoiesis or in conditions of increased oxidative stress.
Regulation of Pentose Phosphate Pathway
The pentose phosphate pathway (PPP) is controlled by numerous ways. Feedback inhibition is one such method, in which the pathway’s end products stop specific enzymes to modulate its rate. The enzymes glucose-6-phosphate dehydrogenase (G6PDH) and 6-phosphogluconate dehydrogenase (6PGDH) are particularly crucial in this control.
- NADPH, a PPP product, acts as a feedback inhibitor of G6PDH, whereas NADPH and ATP inhibit 6PGDH.
- The ratio of NADP+ to NADPH in the cell also affects PPP regulation. High NADPH levels inhibit G6PDH, lowering route activity, whereas low NADPH levels reverse this inhibition and promote the PPP.
- Hormones, particularly insulin and glucagon, play a role in PPP regulation. Insulin stimulates glucose absorption into cells, increasing the availability of substrate for the PPP.
- It also stimulates G6PDH, the pathway’s major regulatory enzyme. Glucagon, on the other hand, inhibits the PPP by reducing insulin levels.
- Another element of PPP regulation is transcriptional regulation, which controls the expression of enzymes participating in the process at the genetic level.
- PPP gene expression is regulated by transcription factors such as NRF2.
- NRF2 can be triggered in response to cellular stress, such as oxidative stress, resulting in increased expression of PPP enzymes to fulfill the demand for NADPH and antioxidant defense.
- The availability of glucose-6-phosphate, the PPP’s beginning substrate, also influences its control.
- Higher quantities of glucose-6-phosphate can stimulate the process, whilst lower levels can shift glucose-6-phosphate to glycolysis.
It should be noted that the regulation of the pentose phosphate pathway varies depending on tissue and physiological factors. The complex interplay between signaling pathways, metabolic demands, and the cellular redox state result in intricate control.
Conclusion
In conclusion, the Pentose Phosphate Pathway (PPP) is a highly significant metabolic pathway with diverse roles in cellular metabolism. The pathway’s two distinct phases, oxidative and non-oxidative, work together to generate NADPH and produce ribose-5-phosphate (R5P), yielding crucial outcomes that impact various cellular processes. The PPP plays a central role in maintaining cellular redox balance. By generating NADPH, the pathway provides reducing equivalents necessary for numerous biosynthetic reactions and facilitates the regeneration of reduced glutathione, an essential antioxidant that protects cells from oxidative damage. The production of NADPH is particularly vital in cells that lack mitochondria, such as erythrocytes, which heavily rely on the PPP to maintain their redox equilibrium and safeguard against oxidative stress.
Furthermore, the PPP contributes to biosynthetic processes by supplying NADPH as a reducing agent. This is crucial for the synthesis of fatty acids, cholesterol, and other important metabolites. Additionally, the production of R5P through the non-oxidative phase of the PPP supports nucleotide synthesis, which is essential for DNA and RNA formation, energy transfer, and cellular signaling. The pathway’s ability to provide a steady supply of R5P ensures the availability of nucleotides, supporting cellular growth, proliferation, and the high turnover rate of erythrocytes.
The significance of the PPP extends beyond its metabolic contributions. Understanding the pathway’s roles and regulation provides valuable insights into cellular homeostasis and the maintenance of vital functions in specific cell types, such as erythrocytes. By protecting against oxidative stress, the PPP contributes to overall cellular health and longevity. Its involvement in nucleotide synthesis makes it crucial in processes requiring rapid cell division, tissue regeneration, and conditions associated with increased cellular growth.
Hence, the Pentose Phosphate Pathway is a fundamental metabolic pathway with profound significance in cellular metabolism. Its roles in maintaining redox balance, protecting against oxidative stress, and supporting biosynthetic processes, particularly nucleotide synthesis, highlight its critical contributions to cellular homeostasis and various physiological processes. Further research on the PPP is essential for a comprehensive understanding of cellular metabolism and its implications for human health and disease.
References
- Smith, C. M., Marks, A. D., Lieberman, M. A., Marks, D. B., & Marks, D. B. (2005). Marks’ basic medical biochemistry: A clinical approach. Philadelphia: Lippincott Williams & Wilkins.
- Rodwell, V. W., Botham, K. M., Kennelly, P. J., Weil, P. A., & Bender, D. A. (2015). Harper’s illustrated biochemistry (30th ed.). New York, N.Y.: McGraw-Hill Education LLC.
- Lehninger, A. L., Nelson, D. L., & Cox, M. M. (2000). Lehninger principles of biochemistry. New York: Worth Publishers.
- Madigan, M. T., Martinko, J. M., Bender, K. S., Buckley, D. H., & Stahl, D. A. (2015). Brock biology of microorganisms (Fourteenth edition.). Boston: Pearson.
- Stincone, Anna, et al. “The return of metabolism: biochemistry and physiology of the pentose phosphate pathway.” Biological Reviews 90.3 (2015): 927-963.
- Ge, Tongxin, et al. “The role of the pentose phosphate pathway in diabetes and cancer.” Frontiers in Endocrinology 11 (2020): 365.
- Pentose phosphate pathway – https://www.khanacademy.org/test-prep/mcat/biomolecules/carbohydrate-metabolism/a/pentose-phosphate-pathway
- Pentose Phosphate Pathway – https://microbenotes.com/pentose-phosphate-pathway/
- PENTOSE PHOSPHATE PATHWAY: FUNCTION, PRODUCTS, AND REGULATION – https://www.tuscany-diet.net/2018/09/01/pentose-phosphate-pathway/

very useful!
Lehninger’s got it?
Thanks for the information.
Although, very informative section, thanks
The explanation was so lucid and very easy to follow, would help students.