- Photosynthesis is the biological process that converts sunlight into chemical energy. It is employed by plants, algae and certain bacteria.
- It provides the basis for life on Earth and is the ultimate source of all fossil fuels and of the oxygen we breathe.
- There are a few key methods to calculate the rate of photosynthesis. These include:
1) Measuring the uptake of CO2
2) Measuring the production of O2
3) Measuring the production of carbohydrates
4) Measuring the increase in dry mass
- Among these, measuring the production of O2 is the simplest and commonly used method to determine photosynthetic activity.
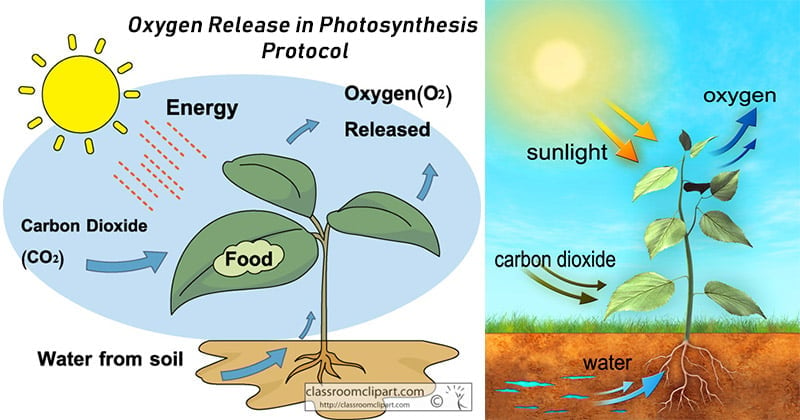
Principle of Oxygen Release in Photosynthesis
During oxygenic photosynthesis, light energy transfers electrons from water (H2O) to carbon dioxide (CO2), to produce carbohydrates. In this transfer, the CO2 is “reduced,” or receives electrons, and the water becomes “oxidized,” or loses electrons. Ultimately, oxygen is produced along with carbohydrates.
Oxygenic photosynthesis is written as follows:
6CO2 + 12H2O + Light Energy → C6H12O6 + 6O2 + 6H2O
Here, six molecules of carbon dioxide (CO2) combine with 12 molecules of water (H2O) using light energy. The end result is the formation of a single carbohydrate molecule (C6H12O6, or glucose) along with six molecules each of breathable oxygen and water.
Oxygenic photosynthesis functions as a counterbalance to respiration by taking in the carbon dioxide produced by all breathing organisms and reintroducing oxygen to the atmosphere.
In the lab, the evolution of oxygen can be detected by using a plant (most commonly aquatic) which is provided with water, carbon-dioxide and exposed to an appropriate light source such that it can carry out photosynthesis. A small amount of sodium bicarbonate added to the water solution serves as the carbon source for photosynthesis that is, the bicarbonate serves as an alternate dissolved source of carbon dioxide. On exposure to light, photosynthesis proceeds and oxygen is released. This release of oxygen can be seen in the form of gas bubbles which can be counted to determine the rate of the process or collected, analyzed and measured later.
Materials Required of Oxygen Release in Photosynthesis
- Elodea plant stem (aquatic plant stem)
- Test tube
- Distilled water
- Sodium bicarbonate
- Scissors
- Light source
- Time keeper (watch)
Procedure of Oxygen Release in Photosynthesis
PART A. Setting up the Experiment
- Obtain a stem of Elodea and remove several leaves from bottom of the stem.
- Cut a portion of the stem at an angle and lightly crush the cut end of the stem.
- Place a small pinch of sodium bicarbonate into the test tube.
- Place the plant into the test tube, bottom end up.
- Fill the test with distilled water so that the stem is completely submersed.
PART B. Running the Experiment
- Notice the bubbles evolved and count them in fixed time intervals when exposed to light.
- Alternatively, the gas can be collected in a pipette or microsyringe and its amount measured.
- The effects of light intensity on photosynthesis can also be investigated by placing the light source at different distances as:
- Place a source of light about 5 cm from the plant.
- Wait for one minute
- After one minute, count the amount of bubbles of oxygen rising from the cut end of the stem for 3 minutes. If bubbles fail to appear after 2 minutes repeat part A.
- Run a second minute trial at 10 cm from the light sources. Record your results.
- Run a third trial 3 minute trial with at a 15 cm distance from a light and record your results.
Expected Results of Oxygen Release in Photosynthesis
- Small gas bubbles are released from the stem. The rate of bubbling can serve as an indication of the rate of photosynthesis.
- When placed closer to a light source, the rate of bubbling will speed up, and as the plant is taken further away, the bubbles will slow down again.
References
- Principles of photosynthesis | Request PDF. Available from: https://www.researchgate.net/publication/229062430_Principles_of_photosynthesis [accessed Nov 03 2018].
- http://www.saps.org.uk/secondary/teaching-resources/157-measuring-the-rate-of-photosynthesis
- https://studylib.net/doc/7103900/elodea-and-photosynthesis-lab
- https://docs.google.com/document/d/1f0VR74VyV1OsY1LDuM7wToWxje-7 ukZuAtaceS8qK8/edit?hl=en_US
- https://www.livescience.com/51720-photosynthesis.html
