What is lignin?
- Lignin is a group of complex aromatic polymers resulting from the oxidative combination of 4-hydroxyphenylpropanoids.
- Lignin is a phenolic polymer that is amorphous, irregular three-dimensional, and high branches.
- The process of biosynthesis of lignin results from the radical coupling reactions of monomers to form cross-linked phenol polymer.
- Lignin is an essential component of the lignocellulosic biomass where it accounts for about 15-30% of the total weight.
- It is the most abundant aromatic biopolymer that consists of about 30% of the organic carbon on Earth.
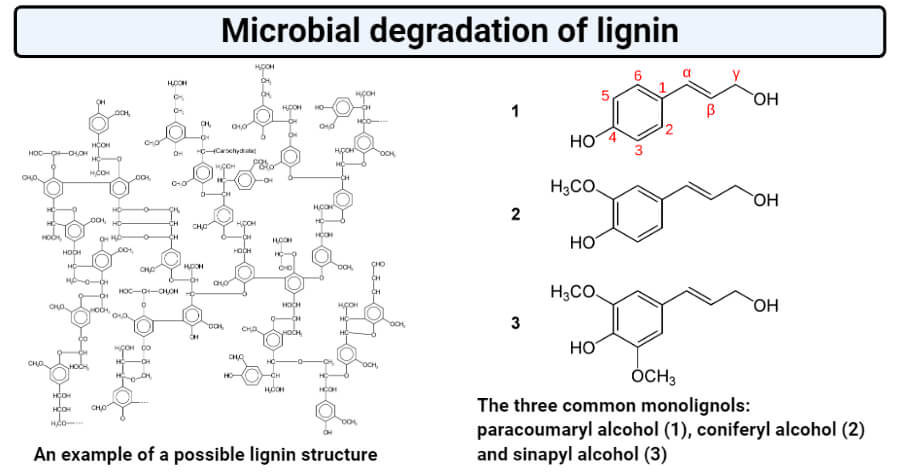
- The chemical species like hydroxycinnamyl alcohols (or monolignols) coniferyl alcohol and sinapyl alcohol, with typically minor amounts of p-coumaryl alcohol, are considered the primary building blocks of lignin.
- However, the chemical composition might vary from species to species.
- The name lignin is derived from the Latin word ‘lignum’ which was first used to derive the relationship between lignin and coniferyl alcohol units.
- Lignin is an important component of the plant cell wall where it provides structural support, helps in water and nutrients transport, and protects from chemical and biological attacks.
- The polysaccharides component of plants is highly hydrophobic, which allows for the transport of water through cell wall components like lignin.
- It has many applications including, energy production, production of aromatic compounds, and production of industrially important compounds.
Structure of lignin
Figure: Structure of lignin. Image Source: Karol Głąb and Yikrazuul.
- Lignin is composed of phenylpropane units linked together by the chemical linkages of alkyl-alkyl, alkyl-aryl, and aryl-aryl groups.
- The precursors of lignin synthesis in nature include p-coumaryl alcohol, coniferyl alcohol, and sinapyl alcohol.
- Different plant species might contain different ratios of the precursors, as the lignin in softwood mainly consists of G-unit, whereas that in hardwood and herbaceous plants consists of G-S units and G-S-H units respectively.
- Natural lignin is not a simple connection of the monomers, but it is formed by the irregular coupling or addition of the precursors.
- There might be significant differences in lignin content in different tissues or different plant species.
- Ecological factors, like plant growth, nutrition, climate, and illumination, also affect the chemical structure of lignin.
- The chemical structure of lignin is one of the most challenging problems left in the field of natural polymers.
- Lignin molecules and their degradation products have many asymmetric centers, but there is no optical activity, so it is not constructed as cellulose or protein by a single key-type linkage.
- Many carbon-carbon bonds might exist between the structural units, and the molecular structure of lignin cannot be determined by the general decomposition method.
- Therefore, lignin is a class of complex molecular structure of polymers that cannot be described by a structural formula, and the properties of lignin are defined only on the basis of the elements, functional groups, and their combination in each unit.
- Natural lignin consists of three important elements: carbon, hydrogen, and oxygen, of which the carbon content is much higher than in homogenous carbohydrates.
- Besides, important structural characteristics of lignin include the functional groups, including alcohol hydroxyl group, carbonyl group, carboxyl group, phenolic hydroxyl group, methoxyl, and sulfonic acid.
- Lignin is a macromolecule that is linked by benzene propane units to form a three-dimensional lignin structure via various chemical bonds.
- Nature and relative proportions of the bonds between benzene propane units are the main determinants of the chemical structure of lignin.
Microorganisms involved in lignin degradation
- The biological degradation of lignin is one of the most important steps in the biospheric carbon and oxygen cycle.
- Much of the lignin biosynthesized by plants is mineralized and returned to the atmosphere as CO2, yet the microbiology of lignin degradation is not well understood.
- The range of microorganisms now known to degrade lignin includes a wide variety of both fungi and bacteria.
- Besides, microorganisms like cyanobacteria and actinomycetes are also known for the degradation of lignin; however, the level of degradation varies with microorganisms.
- The degradation and transformation of lignocellulosic wastes are attributed to the metabolism of indigenous microorganisms.
- Different microbial population dominates at various stages and has distinct roles in the degradation of organic matter.
1. Lignin-degrading bacteria
- Bacterial degradation of wood generally occurs slowly, and on wood surfaces with high moisture content.
- Because of the lack of penetrating ability, bacteria usually invade wood cells simultaneously with fungi.
- The occurrence of lignin-degrading enzymes has been observed in Mycobacterium tuberculosum, M. avium, Pseudomonas syringae, P. aeruginosa, P. putida, Bordetella pertussis, Xanthomonas campestris, Escherichia coli, Caulobacter crescentus Rhodobacter capsulatus, Yersinia pestis, Campylobacter jejuni, and Aquifex aeolicus.
- Bacteria like Paenibacillus, Aneurinibacillus aneurinilyticus, and Bacillus sp. are found to degrade the kraft lignin.
- Rumen bacteria like Fibrobacter succinogenes, Ruminococcus albus, and Flaavefaciens are major degraders of plant fiber cell walls and have an emerging role for bacteria in lignin degradation and bio-product formation.
2. Lignin-degrading Actinomycetes
- Traditionally, it has been suggested that actinomycetes play a role in lignocellulose breakdown, but details of the scale and methods used for such breakdown are less known.
- Streptomyces and other actinomycetes have been identified as lignin-degrading species and can be isolated from a wide variety of sources, including a range of soils, high-temperature environments, and termite guts.
- Lignin-degrading enzymes have been observed in five different species of Streptomyces; Streptomyces antibioticus, S. griseus, S. coelicolor, S. cyaneus, and S. lavendulae.
- Peroxidase and polyphenol oxidase activity has also been observed in the thermophilic Streptomyces isolates and mesophilic Streptomyces.
3. Lignin-degrading Fungi
- Among different wood-decaying fungi, only the white rots have the potential to degrade all three major components of wood entirely.
- These fungi mainly belong to the Ascomycetes, Deuteromycetes, or Basidiomycetes group.
- Typical examples of white-rot fungi are Ganoderma applanatum and Heterobasidion annosum that preferentially remove lignin without a substantial loss of cellulose and cause white-pocket or white-mottled type of rot.
- Other Ascomycetes like Rhizoctonia solani, Aspergillus nidulans, Podospora anserina, Neurospora crassa, Gaeumannomyces graminis var. tritici and Trichoderma reesei have also been described to produce laccase and other lignocellulolytic enzymes.
- Overlapping habits of lignin degradation have been found in the three main ecological groups as wood-decaying, mycorrhiza-forming, and litter-decomposing fungi.
- Wood-decay fungi such as Hypholoma sp. (Nematoloma sp.), Pleurotus sp., and Armillaria are also capable of colonizing soil in contact with wood debris and causing lignin degradation.
- Litter-decomposing fungi (e.g., Stropharia rugosoannulata) are able to grow on the straw that is usually favored only by wood-decay fungi.
- Agaricus bisporus is also a litter-degrading fungus which secretes laccases and manganese peroxidases, and its ability to break down lignin and cellulose enables it to function as a typical white-rot fungus.
Enzymes involved in the degradation of lignin
- Lignin does not contain hydrolyzable linkages, which means that the enzymes must be oxidative in nature to degrade it.
- Lignin is a stereo-irregular compound, which indicates that the enzymes attack the substrate in a more nonspecific way compared to other natural polymers.
- Primarily, three different enzymes are involved in lignin degradation; manganese peroxidase (MnP), lignin peroxidase (LiP), and laccases. However, many other enzymes are participating in lignin degradation.
1. Lignin peroxidases (LiP)
- Lignin peroxidase is an extracellular heme-containing peroxidase that is dependent on H2O2 and degrades a variety of lignin-related compounds.
- These enzymes have an unusually high redox potential and low optimum pH, typically showing little specificity toward substrates.
- Lignin peroxidase is well known as part of the ligninolytic system both of aphyllophoralic and agaricalic fungi.
- These peroxidases preferably oxidize methoxylated aromatic ring without a free phenolic group.
- Methoxylated benzenes and benzyl alcohols are the simplest aromatic substrates for lignin peroxidase.
- The role of lignin peroxidase in ligninolysis could be the further transformation of lignin fragments which are initially released by manganese peroxidase.
- LiP is used commercially to mineralize a variety of recalcitrant aromatic compounds, like three- and four-ring polyaromatic hydrocarbons, polychlorinated biphenyls, and natural dyes.
2. Manganese peroxidases (MnP)
- Manganese peroxidase is an extracellular heme-containing peroxidase with a requirement for Mn2+ as its reducing substrate that has lignin-reducing properties.
- Manganese peroxidase is one of the most common lignin-degrading peroxidases produced by the majority of wood-decaying fungi and many litter-decomposing fungi.
- Structurally, these enzymes are glycosylated proteins with an iron protoporphyrin IX (heme) prosthetic group.
- The enzyme oxidizes Mn2+ to Mn3+, which in turn oxidizes phenolic structures to phenoxyl radicals. Mn3+ formed is highly reactive and complexes with chelating organic acids such as oxalate or malate.
- As the redox potential of the MnP-Mn complex is lower than that of lignin peroxidase, it preferably oxidizes phenolic substrates.
- The phenoxyl radicals formed might further react, resulting in the eventual release of CO2.
- The phenoxyl radicals formed subsequently cleave Cα -Cα or alkyl-phenyl bonds causing depolymerization to smaller intermediates including quinones and hydroxyl quinines.
3. Laccases
- Laccases are a group of lignin-degrading enzymes consisting of N-glycosylated extracellular blue oxidases and four copper atoms in the active site that are distributed among different binding sites.
- Laccases catalyze the oxidation of several aromatic hydrogen donors with subsequent reduction of oxygen to water.
- Moreover, laccases oxidize not only the phenolic and methoxyphenolic acids but also decarboxylate them and attack their methoxy side chains or groups.
- Several fungal laccases have been considered for the oxidation of compounds like 1-(3,4-dimethoxyphenyl)-2-(2-methoxyphenoxy) propan-1,3-diol (I), and phenolic lignin model compounds like phenol red in the presence of the redox mediators.
- Laccases have been reported to oxidize many recalcitrant substances, such as chlorophenols polycyclic aromatic hydrocarbons (PAHs), lignin-related structures, and organophosphorus compounds.
Factors affecting lignin degradation
The process of lignin degradation can be affected by a number of factors like;
1. Moisture content
- Degradation of lignin occurs most rapidly when the source is completely saturated, and free water is present.
- Increasing the amount of water has little effect on the degradation process until aeration becomes impaired.
2. Added Nitrogen
- The growth and biosynthesis of lignin-degrading enzymes require a particular concentration of nitrogen.
- It is observed that the rate of lignin degradation increases with the increase in nitrogen concentration but only to a certain degree.
- Besides, the addition of a very high concentration of nitrogen tends to have a detritus effect on the overall carbohydrate degradation, along with lignin degradation.
3. Added glucose
- The process of lignin degradation slows down with the addition of glucose as an excess of readily available energy source like glucose causes decreased consumption of lignin.
- When all the available energy sources are omitted, the microbial action on the lignin-rich compounds increases.
4. Aeration
- The rate of lignin and carbohydrate metabolism increases with the increase in oxygen content in the environment.
- This is due to the fact that most of the microorganisms involved in lignin degradation are aerobic microorganisms that thrive in a high oxygen atmosphere.
- Besides, efficient lignin degradation is also seen under a CO2 concentration of 30% and an O2 concentration of 10%.
- However, a pure O2 atmosphere might be toxic if any other form of energy like glucose or cellulose is present as it suppresses lignin degradation.
Process (Simple Steps) of lignin degradation
The overall process of lignin degradation can be explained in two simple steps;
1. Depolymerization
- The first step of lignin degradation is the depolymerization of aryl and biaryl compounds such as β-aryl ethers.
- This step is a non-specific step that occurs extracellularly by different bacterial and fungal enzymes.
- Depolymerization of lignin occurs as a result of cleavage of β-O-4 ether bond that represents about 50% or more of the total linkages in lignin.
- This step decreases the length of the lignin polymer so as to obtain dimeric or oligomeric units, which can then further be degraded into smaller molecules.
- Depolymerization in microorganisms is catalyzed by different lignin-degrading enzymes found in many microorganisms like peroxidases and phenol oxidases.
- These enzymes attack the lignin randomly, then convert the phenolic group to free-radicals, and these radicals lead to lignin depolymerization.
2. Solubilization and Mineralization
- The smaller molecules of lignin formed after depolymerization are now taken by different microorganisms that catalyze a series of conversion by various in vivo enzymes.
- Most of the linkages within the lignin molecules have their specific metabolic pathways to cleave these specific linkages.
- The mineralization and solubilization of oligomers and monomers result in the formation of CO2 and other essential molecules that can be utilized by the organisms.
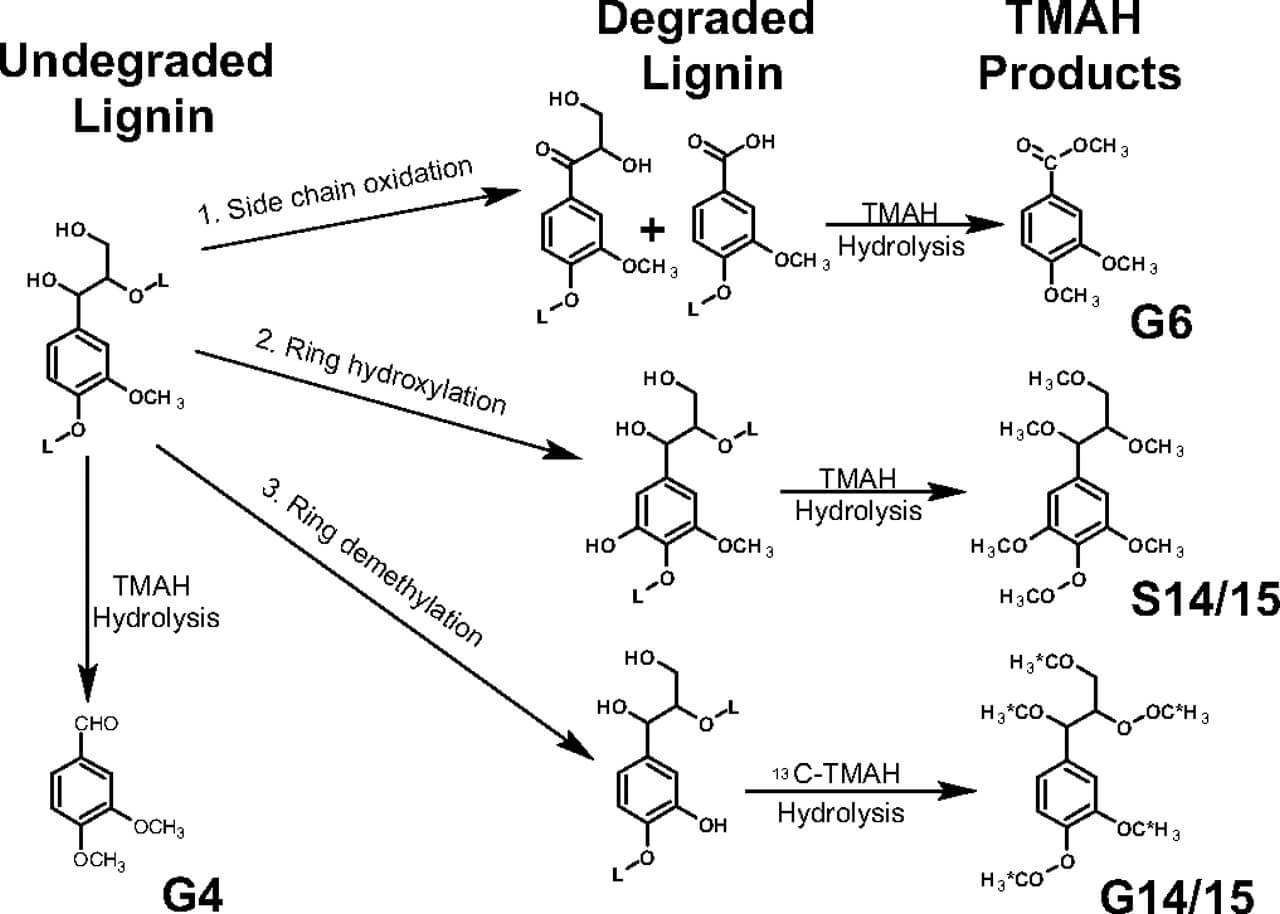
Figure: Lignin biodegradation reactions. Reactions include propyl side-chain oxidation/cleavage (reaction 1), ring hydroxylation (reaction 2), and demethylation (reaction 3). Reaction 1 is characteristic of the white-rot fungus, whereas reactions 2 and 3 are characteristic of brown-rot fungus. Application of TMAH thermochemolysis to each of the degradative reactions is pictured following each reaction, showing chemical structures of compounds G4, G6, G14/15, and S14/15 that result from this analysis. Asterisks represent 13C-labeled carbons resulting from 13C-TMAH analysis. Image Source: https://doi.org/10.1073/pnas.0805257105.
Mechanisms of microbial degradation of lignin
- The mechanisms of microbial degradation of lignin might be different with different types of enzymes involved.
- Three particularly important enzymes; laccases and lignin peroxidases are involved in the in vitro depolymerization of larger lignin molecules.
- Other mechanisms are involved in the solubilization of lignin into even smaller usable molecules within the microorganisms.
- The overall process of lignin degradation can be explained as in vitro degradation and in vivo degradation.
A. In vitro degradation
1. Biodegradation of lignin with laccase
- The biodegradation of lignin by the laccase enzyme system is an oxidative process consisting of enzyme-catalyzed and mediator-facilitated radical reactions.
- This process degrades both phenolic and non-phenolic aromatic structures in lignin. However, the initial attack is on the phenolic moiety, followed by the degradation of non-phenolic benzylic structures.
- As a result of the initial attack, phenol residues are released with oxidized side chains. This enables the enzyme to penetrate the bulk lignin polymer and act as natural mediators to oxidize non-phenolic regions.
- The mechanism of lignin degradation proceeds by a Cα-Cβ cleavage of non-phenolic sites in lignin with solubilization of lignin fragments by the formation of hydrophilic lignin-mediator complexes.
- This occurs by one of the three oxidation mechanisms; electron transfer, radical hydrogen atom transfer, and ionic mechanism.
2. Biodegradation of lignin by lignin peroxidase
- The catalytic cycle of lignin peroxidase-like manganese peroxidase is initiated by the binding of H2O2 or an organic peroxide to a native ferric enzyme and formation of an iron-peroxide complex.
- The complex thus formed is cleaved, resulting in the oxidation of Mn2+ into Mn3+.
- Chelates of Mn3+ with carboxylic acids (e.g. oxalate, malonate, malate) cause one-electron oxidations of various substrates.
- Phenolic and amino-aromatic compounds are oxidized by the removal of hydrogens to form phenoxyl and amino radicals, respectively.
- These radicals are the sources of peroxides that are generated via auto-catalytical reactions and can be used by MnP in the absence of external H2O2.
- The underlying mechanism of decomposition of lignin is based on initial one-electron oxidation of the substrate by enzyme-generated Mn3+, which produces a phenoxyl radical intermediate.
- This radical is further oxidized by Mn3+ to form a carbon-centered cation. The subsequent loss of proton yields the ketone dimer.
- An attack by water on the cation, followed by the cleavage of the arylglycerol-aryl structure, produces the other depicted products.
B. In vivo degradation
1. β-O-4 ether degradation
- The cleavage of the β-O-4 ether bond is considered an important step for lignin depolymerization as it represents about 50% of all linkage sin lignin.
- The pathway and mechanism of β-O-4 degradation start with LigD, a Cα-dehydrogenase.
- LigD oxidizes the hydroxyl group at Cα position, then the β-etherase, LigE or LigF, cleave the β-O-4 ether bond and generate vanillin and αglutathionyl-β-hydroxypropiovanillone (GS-HPV) as intermediate.
- Finally, the GS-HPV is oxidized by LigG, glutathione-S-transferase.
- The glutathione is cleaved, and the remaining β−hydroxypropiovanillone can be further oxidized to vanillin.
2. Bi-phenyl degradation pathway
- The biphenyl linkage represents 10% of the total linkages in softwood lignin.
- When the 5, 5’-dehydrodivanillate (DDVA) enters the organism, LigX, a DDVA O-demethylase, demethylates one of the methoxy group and convert it to the hydroxyl group.
- The product of LigX is the substrate for oxidative meta-cleavage by LigZ, OH-DDVA dioxygenase.
- The product from LigZ is further hydrolyzed by LigY, a hydrolase for the meta-cleavage compound of OH-DDVA.
- Finally, the end products of the cleavage of LigY are 4-carboxy-2-hydroxypentadienoic acid and 5-carboxyvanillic acid (5CVA), and the 5CVA is further metabolized into one of the central products, vanillate.
Example of lignin degradation
Lignin degradation by Pseudomonas
- Pseudomonas metabolize various low molecular weight lipid found in lignocellulose biomass.
- It produces a benzaldehyde lyase which cleaves the acyloin linkage of the 1,2-diarylethene compounds and benzoin.
- The enzyme is highly specific, cleaving only anison (4,4’-dimethoxybenzoin) and benzoin.
- Some species of Pseudomonas might even produce protocatechuate-4,5-dioxygenase which catalyzed the ring-fission of protocatechuate and 3-methyl-gallic acid.
References
- Kuhad R.C., Kuhar S., Sharma K.K., Shrivastava B. (2013) Microorganisms and Enzymes Involved in Lignin Degradation Vis-à-vis Production of Nutritionally Rich Animal Feed: An Overview. In: Kuhad R., Singh A. (eds) Biotechnology for Environmental Management and Resource Recovery. Springer, India. https://doi.org/10.1007/978-81-322-0876-1_1
- Vanholme, Ruben et al. “Lignin biosynthesis and structure.” Plant physiology 153,3 (2010): 895-905. doi:10.1104/pp.110.155119
- Annunziata, Maria Grazia. “What Is Lignin Made of? New Components Discovered!.” Plant physiology 180,3 (2019): 1255. doi:10.1104/pp.19.00561
- Vinardell, Maria Pilar, and Montserrat Mitjans. “Lignins and Their Derivatives with Beneficial Effects on Human Health.” International journal of molecular sciences 18,6 1219. 7 Jun. 2017, doi:10.3390/ijms18061219
- Leisola, M.S.A., Ulmer, D.C. & Fiechter, A. Factors affecting lignin degradation in lignocellulose by Phanerochaete chrysosporium. Arch Microbiol 137, 171–175 (1984). https://doi.org/10.1007/BF00414462
- Yang, H.H., Effland, M.J. and Kirk, T.K. (1980), Factors influencing fungal degradation of lignin in a representative lignocellulosic, thermomechanical pulp. Biotechnol. Bioeng., 22: 65-77. https://doi.org/10.1002/bit.260220106
- Zimmermann, Wolfgang. (1990). Degradation of lignin by bacteria. Journal of Biotechnology. 13. 119-130. 10.1016/0168-1656(90)90098-V.
- Hofrichter, Martin. (2002). Review: Lignin conversion by manganese peroxidase (MnP). Enzyme and Microbial Technology. 30. 454-466. 10.1016/S0141-0229(01)00528-2.
- Jin, L., Nicholas, D.D. & Kirk, T.K. Mineralization of the methoxyl carbon of isolated lignin by brown-rot fungi under solid substrate conditions. Wood Sci.Technol.24, 263–276 (1990). https://doi.org/10.1007/BF01153559
- Grzegorz Janusz, Anna Pawlik, Justyna Sulej, Urszula Świderska-Burek, Anna Jarosz-Wilkołazka, Andrzej Paszczyński, Lignin degradation: microorganisms, enzymes involved, genomes analysis and evolution, FEMS Microbiology Reviews, Volume 41, Issue 6, November 2017, Pages 941–962, https://doi.org/10.1093/femsre/fux049
- Chonlong Chio, Mohini Sain, Wensheng Qin. Lignin utilization: A review of lignin depolymerization from various aspects.Renewable and Sustainable Energy Reviews. Volume 107. 2019. Pages 232-249. https://doi.org/10.1016/j.rser.2019.03.008.
- Christopher Lew Paul, Yao Bin, Ji Yun. Lignin Biodegradation with Laccase-Mediator Systems. Frontiers in Energy Research. DOI=10.3389/fenrg.2014.00012
- Chapter 2 – Structure and Characteristics of Lignin. Lignin Chemistry and Applications. Elsevier. 2019. Pages 25-50. https://doi.org/10.1016/B978-0-12-813941-7.00002-3.
- Don L. Crawford, Ronald L. Crawford. Microbial degradation of lignin. Enzyme and Microbial Technology. Volume 2, Issue 1. 1980. Pages 11-22. https://doi.org/10.1016/0141-0229(80)90003-4.
Sources
- 4% – https://www.sciencedirect.com/science/article/pii/B9780128139417000023
- 2% – https://www.sciencedirect.com/science/article/pii/S136403211930142X
- 2% – https://www.sciencedirect.com/science/article/pii/0141022980900034
- 2% – https://www.researchgate.net/publication/223374143_Large_scale_production_of_manganese-peroxidase_using_agaric_white-rot_fungi
- 2% – https://www.researchgate.net/publication/222915661_Review_Lignin_conversion_by_manganese_peroxidase_MnP
- 1% – https://www.sciencedirect.com/science/article/pii/S0734975003001344
- 1% – https://www.sciencedirect.com/science/article/pii/S0141022901005282
- 1% – https://www.sciencedirect.com/science/article/pii/S0065216405580043
- 1% – https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3825317/
- 1% – http://www.plantphysiol.org/content/153/3/895
- <1% – https://www.sciencedirect.com/science/article/pii/S0960852413005865
- <1% – https://www.sciencedirect.com/science/article/pii/S0038071712002829
- <1% – https://www.sciencedirect.com/science/article/abs/pii/S0958166909001268
- <1% – https://www.researchgate.net/publication/49647829_Oxidation_of_polycyclic_aromatic_hydrocarbons_by_the_bacterial_laccase_CueO_from_E_coli
- <1% – https://www.researchgate.net/publication/35947053_Degradation_of_recalcitrant_biopolymers_and_polycyclic_aromatic_hydrocarbons_by_litter-decomposing_basidiomycetous_fungi
- <1% – https://www.researchgate.net/publication/329625877_Advances_in_microbial_lignin_degradation_and_its_applications
- <1% – https://www.researchgate.net/publication/251232174_Wood_Decaying_Fungi
- <1% – https://www.researchgate.net/publication/223053539_Degradation_of_lignin_by_bacteria
- <1% – https://www.researchgate.net/publication/11833869_Fungal_Laccase_Properties_and_Activity_on_Lignin
- <1% – https://www.pnas.org/content/105/35/12932
- <1% – https://www.jstage.jst.go.jp/article/pjab/80/5/80_5_204/_pdf/-char/en
- <1% – https://www.irjet.net/archives/V7/i10/IRJET-V7I10332.pdf
- <1% – https://www.chemguide.co.uk/mechanisms/freerad/alkenehbr.html
- <1% – https://royalsocietypublishing.org/doi/10.1098/rsos.191202
- <1% – https://pubs.acs.org/doi/10.1021/bi047318e
- <1% – https://onlinelibrary.wiley.com/doi/full/10.1111/j.1574-6968.2001.tb10911.x
- <1% – https://healthyeating.sfgate.com/three-elements-carbohydrates-including-sugar-8845.html
- <1% – https://gardenerdy.com/plant-growth-factors/
- <1% – https://en.wikipedia.org/wiki/Lignin_biosynthesis
- <1% – https://en.wikipedia.org/wiki/Lignin
- <1% – https://biotechnologyforbiofuels.biomedcentral.com/articles/10.1186/s13068-017-0735-y
- <1% – https://aem.asm.org/content/68/3/1305
- <1% – http://web.nchu.edu.tw/pweb/users/taiwanfir/lesson/10476.pdf

Concise and comprehensive !
Thank you very much. Excellent work
Thank you for this information. I am trying to understand lignin formation and degradation in relation to some chronic conditions in the human body.