In order to maintain glucose homeostasis and guarantee a consistent source of energy, living creatures, including humans, engage in the biological process of glycogenesis. It entails the creation of glycogen, a glucose storage form, from glucose molecules.
Glycogenesis is the process of glycogen synthesis, in which glucose molecules are added to chains of glycogen for storage.
The phases of glycogenesis are examined in this article, along with the mechanisms for control and regulation that underpin it. Additionally, it explores epinephrine (adrenaline) and calcium ions as two important variables that affect glycogenesis. To understand the complex control of glycogen metabolism, it is essential to know these features.
Location of Glycogenesis
Glycogenesis takes place in the cytoplasm of cells in the muscle, liver, and adipose tissue.
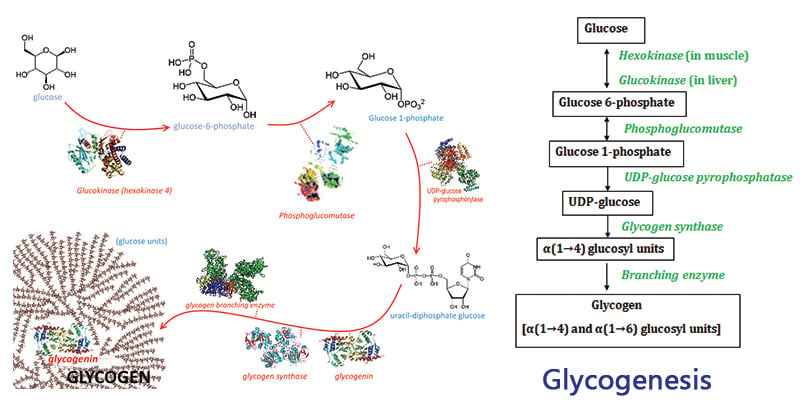
Steps of Glycogenesis
Glycogenesis is a multistep process that takes place primarily in the liver and muscle cells, although other tissues also contribute to glycogen synthesis. The following steps outline the process of glycogenesis:
1. Glucose Uptake
- In this step, glucose molecules from the bloodstream are transported into the liver or muscle cells through glucose transporters, such as GLUT2 in the liver and GLUT4 in muscle cells.
- This facilitated diffusion process ensures an adequate supply of glucose for glycogen synthesis within the cells.
- Reaction: Glucose + ATP → Glucose-6-Phosphate + ADP(Catalyzed by hexokinase)
2. Conversion to Glucose-6-Phosphate (G6P)
- Once inside the cell, glucose is converted to glucose-6-phosphate (G6P) through a phosphorylation reaction.
- The enzyme hexokinase or glucokinase catalyzes this reaction, using ATP as a phosphate donor.
- The conversion of glucose to G6P traps glucose within the cell and makes it more reactive for further metabolic processes.
- Reactions: Glucose + ATP → Glucose-6-Phosphate + ADP (Hexokinase or Glucokinase)
3. Isomerization to Glucose-1-Phosphate (G1P)
- In this step, the G6P isomerizes to glucose-1-phosphate (G1P).
- The enzyme phosphoglucomutase catalyzes this reaction by transferring the phosphate group from the sixth carbon to the first carbon of the glucose molecule.
- This isomerization is an essential step for the subsequent incorporation of glucose into the growing glycogen chain.
- Reactions: Glucose-6-Phosphate ⇌ Glucose-1-Phosphate (Catalyzed by phosphoglucomutase)
4. Activation of Glucose for Glycogen Synthesis
- To activate glucose for glycogen synthesis, G1P undergoes activation by the enzyme UDP-glucose pyrophosphorylase.
- The reaction involves the transfer of UTP (uridine triphosphate) to G1P, resulting in the formation of UDP-glucose and inorganic pyrophosphate (PPi).
- This step ensures that glucose is energetically favorable for incorporation into glycogen.
- Reactions: Glucose-1-Phosphate + UTP ⇌ UDP-Glucose + PPi (Catalyzed by UDP-glucose pyrophosphorylase)
5. Glycogen Elongation
- Glycogen synthase, the key regulatory enzyme of glycogenesis, catalyzes the elongation of the glycogen chain.
- It adds glucose units to the growing glycogen molecule by utilizing UDP-glucose as the substrate.
- The enzyme forms an α-1,4-glycosidic bond between the glucose molecules, extending the glycogen chain.
- Reactions: UDP-Glucose + Glycogen (n residues) → Glycogen (n+1 residues) + UDP (Catalyzed by glycogen synthase)
6. Branching of Glycogen:
- When the glycogen chain reaches a certain length, branching occurs to ensure efficient synthesis and degradation of glycogen.
- The branching enzyme, α-1,6-glucosyltransferase, catalyzes the transfer of a segment of the glycogen chain and attaches it via an α-1,6-glycosidic bond.
- This branching creates branches within the glycogen structure, allowing for easier access by enzymes involved in glycogen synthesis and breakdown.
- Reaction: Glycogen (n residues) → Glycogen (n-1 residues) + Glycogen (m residues) (Catalyzed by branching enzyme, α-1,6-glucosyltransferase)
Control and Regulation of Glycogenesis
The process of glycogenesis is strictly regulated and controlled by various important elements in order to maintain glucose homeostasis. These systems make sure that glycogen synthesis happens when the body needs energy storage and glucose levels are high.
- Insulin and glucagon are the two main hormones that regulate glycogenesis.
- The hormone insulin, which is secreted by the pancreas, is crucial in fostering glycogenesis.
- When blood glucose levels rise, insulin is produced to aid in glucose absorption by cells and promote the creation of glycogen.
- Glycogen synthase is made more active by insulin, which also encourages the conversion of G1P to glycogen.
- Contrarily, when fasting or low blood sugar, glucagon counteracts the actions of insulin and controls the metabolism of glycogen.
- Although the pancreas also secretes it, it works against insulin.
- Glucagon stimulates gluconeogenesis and glycogenolysis, the process by which glycogen is broken down.
- This is accomplished by blocking glycogen synthase and turning on glycogen phosphorylase, the enzyme that breaks down glycogen.
Toggle Control and Regulation Subsection
An extra degree of regulation exists in addition to the hormonal control provided by glucagon and insulin in the form of covalent alteration of glycogen synthase and glycogen phosphorylase.
- Toggle control is the technique of reversibly phosphorylating certain enzymes to either activate or deactivate them. Protein kinase A (PKA) phosphorylates glycogen synthase to decrease its activity and stop glycogen production.
- Glycogen synthase is dephosphorylated by protein phosphatase 1 (PP1) to become active and encourage glycogen synthesis while PKA is inactive.
- Phosphorylation controls the activity of glycogen phosphorylase as well.
- Dephosphorylated glycogen phosphorylase is inert, whereas phosphorylated glycogen phosphorylase catalyzes the breakdown of glycogen.
Epinephrine (Adrenaline)
The adrenal glands release the hormone epinephrine, sometimes known as adrenaline, during times of stress or physical activity. To provide a quick supply of glucose for energy, epinephrine triggers the breakdown of glycogen. Glycogen phosphorylase is phosphorylated and activated by PKA, which also inhibits glycogen synthase. This is accomplished through a signaling mechanism.
Calcium Ions
- Additionally, calcium ions are crucial for controlling glycogen metabolism.
- Glycogen phosphorylase is activated and glycogen breakdown is promoted by elevated intracellular calcium concentrations, which are frequently brought on by muscular tension.
- This is accomplished by calcium ions binding to calmodulin, which activates the CaMK protein kinase.
- Phosphorylase kinase is then phosphorylated and activated by CaMK, which in turn phosphorylates and activates glycogen phosphorylase.
Conclusion
Glycogenesis is a tightly controlled process that enables the storage of extra glucose as glycogen. Glycogen is created by a complex process that transforms glucose into a store of energy. Insulin and glucagon, as well as covalent modification via toggle control, play a role in the control and regulation of glycogenesis.
The metabolism of glycogen is also influenced by epinephrine and calcium ions, which largely promote glycogen breakdown. The complexity of glucose homeostasis and energy management in the body must be understood in order to fully appreciate these systems.
References
- Glycogenesis – https://byjus.com/chemistry/glycogenesis/
- Biochemistry, Glycogenesis – https://www.ncbi.nlm.nih.gov/books/NBK549820/
- Glycogenesis – https://www.britannica.com/science/glycogenesis
- Glycogenesis Definition – https://biologydictionary.net/glycogenesis/
- Glycogenesis – https://www.sciencedirect.com/topics/neuroscience/glycogenesis
- Glycogenesis – https://bio.libretexts.org/Bookshelves/Biochemistry/Fundamentals_of_Biochemistry_(Jakubowski_and_Flatt)/02%3A_Unit_II-_Bioenergetics_and_Metabolism/15%3A_Glucose_Glycogen_and_Their_Metabolic_Regulation/15.02%3A_Glycogenesis
- David Hames and Nigel Hooper (2005). Biochemistry. Third ed. Taylor & Francis Group: New York.
- Lehninger, A. L., Nelson, D. L., & Cox, M. M. (2000). Lehninger principles of biochemistry. New York: Worth Publishers.
- https://www.checkdiabetes.org/glycogenesis/

Thank you for your effort i have read and got it clear, continue embracing it till we also get there.
the text is more because I have caught many concepts within the few minutes.
kongole to the author.
you wrote “Glucagon suppresses gluconeogenesis” this is wrong Glucagon stimulates gluconeogenesis, plz fix it
Hi Talha,
Thanks for the correction.
We have updated the page with, “Glucagon stimulates gluconeogenesis and glycogenolysis, the process by which glycogen is broken down.”
Thank you,
Sagar
I really appreciate with this concrete idea of yours may allah let us know more ways to pass this sciences idea or experience to others or in general in in a food web.
Thank u for ur cooperation
I am A BSc Medical Laboratory Technology. It helps me making notes.
I am BSC Nursing.I like how the pioneers embrace the knowledge of Biochemistry.More thanks for helping me have an easier revision for Biochemistry
I’m a BS Psychology student. The categorization of details and info, helped me a lot in my biochemistry lecture class.
I’m BSND student, it helped me alot. Thank you!
Very imformative content, it helped me alot during my last minute revision