Epidermophyton floccosum is a filamentous dermatophytic fungus known to cause skin and nail infections in humans. It is an anthropophilic dermatophyte hence it is transmissible from one individual to another. Common infections caused by dermatophytes include tinea pedis (athlete’s foot), tinea crusis, tinea corporis, and onychomycosis. It is the third most common cause of Tinea pedis (athletes foot).
Habitat of Epidermophyton floccosum
- It is an ascomycete and therefore it has a worldwide distribution.
- It commonly occurs in tropical and subtropical regions.
- It does not live in the soil.
- It is common in North America and Asian countries.
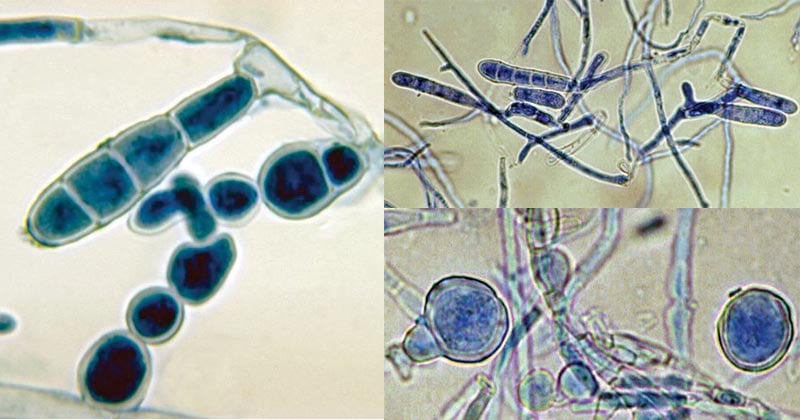
Figure: Epidermophyton floccosum. Image Source: Wikipedia and The University of Adelaide.
Morphological and Cultural Characteristics of Epidermophyton floccosum
- Growth in basic mycological medium, Epidermophyton floccosum grows slowly, producing greenish-brown or khaki-colored colonies, with a suede-like surface.
- The colonies have a centralized occurrence that is raised and folded, a flat periphery, and submerged fringe growth.
- Mature cultures produce white pleomorphic tufts of mycelium with a deep yellowish-brown reverse pigment.
- Microscopic observation shows smooth, thin-walled macroconidia occurring in clusters on the hyphal threads.
- They are also observed to be filamentous fungus, with septate and hyaline hyphae.
- The hyphae contain smooth, whin-walled, clavate, club-shaped, clusters macroconidia.
- Chlamydospores are also formed in mature/old cultures.
- They do not form microconidia.
- The distinction of Epidermophytes floccosum from the other dermatophytes (Microsporum and Trichophyton)is that they do not form microconidia and they form macroconidia which are shorter, wide, and smooth.
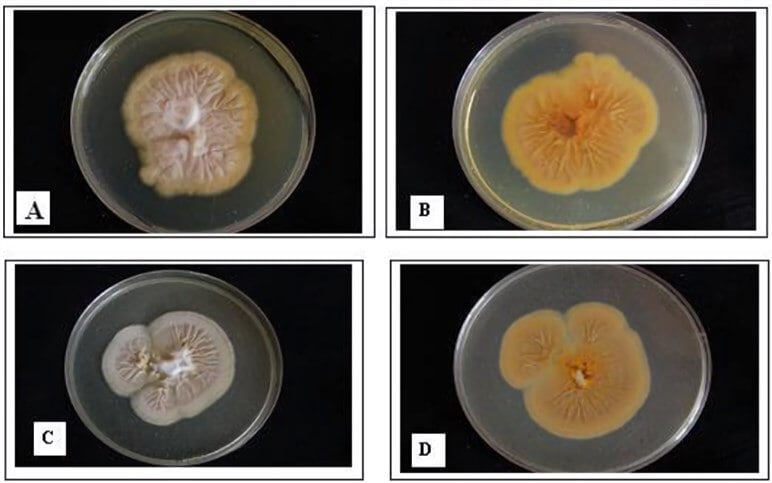
Figure: Pleomorphism of E. floccosum. A: Overview of the Wild-Type surface; B: Revise side of wild-type; C: Overview of pleomorphic E. floccosum; D: Revise side of pleomorphic E. floccosum. (Al-Janabi, 2009) Image Source: Creative Biolabs.
Pathogenesis and clinical manifestations of Epidermophyton floccosum
- Epidermophyton floccosum is transmitted from person-to-person via skin contact, causing cutaneous and subcutaneous infections on the skin, and nails.
- As a dermatophyte, it causes dermatophytosis affecting the keratinized regions in the body including hair, skin, and nails.
- Symptoms vary depending on the site of the body affected, such as:
- Hair infections are Tinea capitis, Tinea barbae characterized by invasion of hair follicles (without perforation), hair loss (ectothrix), and/or hair breakage (endothrix).
- On the skin, the formation of lesions is commonly characterized by circular or annular ringworm infections (tinea corporis).
- As an anthropophilic fungus, it does not cause inflammations or hypersensitivity.
- They have also been associated with causing keratitis of the eye.
- Infection of the nails is commonly characterized by discoloration, dystrophy, hyperkeratosis, and onycholysis.
- Manifestations occur several weeks after contracting the fungus.
- Disseminated and invasive disease infections are rare.
- However, invasive Epidermophyton floccosum infection has been documented in persons with Behcet’s syndrome.
Laboratory Diagnosis of Epidermophyton floccosum
Specimen: Skin scrapings, pus/lesion biopsies, hair scarpings
Microscopic Examination
10-20% KOH Wet mount for observation of smooth, thin-walled, wide macroconidia.
Cultural Examination
- Growth in Sabouraud Dextrose Agar with cycloheximide and chloramphenicol to suppress mold and bacterial growth and observation of growth in 3 weeks. The colonies formed are greenish-brown or khaki-colored with a suede-like surface.
- The colonies also appear raised and folded at the center with a flattened periphery.
- Extended growth produces white colonies showing pleomorphism in mycelial growth, producing deep yellow to brown mycelium on reverse pigmentation.
Molecular characterization and differentiation
- Use of PCR-Restriction Fragment Length Plormophism (PCR-RFLP) is used to distinguish the 12 species that cause dermatophytosis infections.
- Real-time PCR is also used to identify Epidermophyton floccosum after fungal lysis
Treatment of Epidermophyton floccosum infections
- Tinea capitis is tretaed with griseofulvin or oral azoles (ketoconazole, itraconazole, and voriconazole).
- Tinea corporis, tinea crisis, tinea pedis, and tinea manuun are treated tropically using naftifine, terbinafine, butenafine, clotrimazole, econazole, ketoconazole, miconazole, oxiconazole, sulconazole, cyclopyrox, and tolnaftate.
- Nail infections, topical therapy is normally unsuccessful, and therefore, the use of systemic oral therapy for a prolonged period is advised.
References
- Medical Microbiology by Jawertz, 23rd Edition
- Microbiology by Prescott, 5th Edition
- Drfungus/epidermophytonfloccosum
- https://www.creative-biolabs.com/drug-discovery/therapeutics/epidermophyton-floccosum.htm
- https://www.sciencedirect.com/book/9780323401814/principles-and-practice-of-pediatric-infectious-diseases
Sources
- 4% – https://mycology.adelaide.edu.au/descriptions/dermatophytes/epidermophyton/
- 2% – https://en.m.wikipedia.org/wiki/Epidermophyton_floccosum
- 1% – https://www.msdsonline.com/resources/sds-resources/free-safety-data-sheet-index/epidermophyton-floccosum/
- 1% – https://reference.medscape.com/features/slideshow/nail-diseases
- 1% – https://mospace.umsystem.edu/xmlui/bitstream/handle/10355/3582/WhatWayTreatTineaCruris.pdf
- 1% – https://link.springer.com/article/10.1007/s10453-016-9462-2
- 1% – https://link.springer.com/article/10.1007%2Fs00404-015-3914-8
- 1% – https://cdn.intechopen.com/pdfs/35104/InTech-Restriction_fragment_length_polymorphism_analysis_of_pcr_amplified_fragments_pcr_rflp_and_gel_electrophoresis_valuable_tool_for_genotyping_and_genetic_fingerprinting.pdf