Introduction
A normal bacterial cell is known as a vegetative form of a bacterial cell. It can multiply freely when provided with conducive environmental conditions like the provision of nutrients, temperature, water, and oxygen. But some groups of bacteria, when exposed to unfavorable conditions such us, lack of nutrients, an insufficient supply of oxygen or CO2, lack of water and moisture, they have the ability to form a protective covering to protect themselves when they are exposed to harsh environmental conditions adapting comfortably to these conditions. This special structure is known as an endospore.
An endospore is a non-vegetative structure produced by a group of bacteria belonging to the Firmicute family. They have special characteristics that stabilize them to survive in adverse conditions for long periods of time.
These include; a phase of dormancy, a tough outer covering and they are non-reproductive forms called spores, which offer great resistance to high temperature, radiations, and chemicals like disinfectants and acids. This enables them to survive thus they are difficult to stain with basic dyes and hence a special stain is applied that uses a special dye, along with heat-steam. This staining technique is known as the Endospore stain, also known as the spore stain. It is used majorly to detect and identify the presence of a bacterial endospore and bacterial vegetative forms in a cell.
Examples of these endospore-forming bacteria include Clostridium spp and Bacillus spp. These bacteria naturally grow in soil, but they have great clinical implications by causing human bacterial infections eg Clostridium tetani causes tetanus, Clostridium botulinum produces botulin toxin which causes paralysis, Bacillus cereus causing food poisoning and Bacillus anthracis causes anthrax in cattle and humans.
Endospore staining techniques are classified based on the types of reagents used;
- Schaeffer Fulton Stain- used Malachite Green dye and safranin
- Dorner method of endospore staining –uses Carbolfuchsin stain, acid alcohol, and Nigrosin solution)
Image Source: Microbiology Info
Objectives of endospore staining
- To detect for the presence of an endospore.
- To identify endospore producing bacteria
- To differentiate between the vegetative forms and the endospore
Principle
Endospore staining is a differential stain that aims at detecting, identifying and differentiating an endospore from the vegetative cell (an underdeveloped endospore). The principle of the role is to detect the presence or absence of the endospore, but some procedures have modified the technique by increasing the concentrations of the dyes, increasing the duration of heat fixing, application of ultraviolet radiation.
With the improved technology in microscopy, some use phase-contrast microscopy which is fast and it produces more detailed morphologies of the bacterial endospore.
1. Schaeffer Fulton Stain
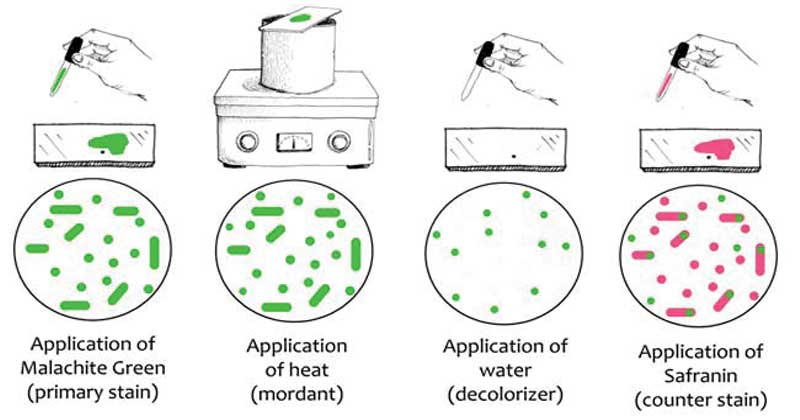
In basic Laboratories, the simplest endospore staining technique is the Schaeffer-Fulton technique because of its easy and it is rapid to identify the bacteria. It applies the use of Malachite green dye (alkaline solution with a pH of 11-11.2 and its a water-soluble dye) along with the use of steamed-heat which softens the endospore covering allowing penetration of the dye into the spore. The malachite green dye binds to the spore mildly and if washed with water, without fixing, it easily washes away, and that’s why the application of steamed heat is important to allow the dye to penetrate the endospore. Water is used as a decolorizing agent, to was away from the malachite dye from vegetative forms. Lastly, the use of a counterstain, Safranin reagent, also knows as the secondary stain, is to stain the vegetative forms of the underdeveloped Furmigates vegetative forms after the malachite dye has been washed away by the decolorizing agent (Water).
Malachite Green dye and Safranin work well in bacteria because of the alkaline nature of the Malachite Green reagents which are charged positively while the cytoplasm of the bacterial cell is basophilic hence there is an attraction between the malachite green dye with the bacterial cell, making it easier to absorb the dye. Visualization of the cells under the microscope will show the appearance of pink-red stain for the vegetative cell forms, which take up the counterstain while the endospores will appear as green dotted particles (ellipses), having taken up the Malachite green dye.
Reagents
- Malachite green dye
- Water (Decoloriser)
- Safranin
Procedure
Equipment: Glass slide, Inoculation loop, Bunsen burner
Preparation of microscope slide (adapted for all other endospore staining techniques)
- Clean the glass slide (with visible circles), with alcohol to remove any stains.
- Using a sterile inoculation loop, put two small drops of water in each circle.
- Aseptically, open the tube with a bacteria culture and flame it at the top and collect a loopful of the bacterial culture from the tube, Flame the tube again and close.
- Smear the bacterial culture in the drop of water on the slide.
- Air dry till its completely dry.
- Heat-fix the slide with smear facing up, by running it over the blue flame 3-4 times NB: Do not flame the side with the bacteria
- Leave to cool and then start to stain.
Staining procedure:
- Cover the smears with a piece of absorbent paper.
- Place the slide over a staining rack, that has a beaker/water bath of steaming water.
- Flood the absorbent paper with malachite green and let it steam for 3-5 minutes.
- Remove the stained absorbent paper carefully and discard and allow it to cool for 1-2 minutes.
- Gently rinse the slide with tap water by tilting the slide to allow the water to flow over the smeared stain. This is to remove the extra dye present on the slide on both sides and to also remove extra dye staining any vegetative forms in the heat-fixed smear.
- Add the counterstain, safranin for 1 minute.
- Rinse the slide with water, on both sides to remove the safranin reagent.
- Ensure the bottom of the slide is dry before placing it on the stage of the microscope to view with the oil immersion lens, at 1000x for maximum magnification.
Result
The vegetative forms will take up the pink/red stain from safranin while the endospores will stain green, from the malachite green dye.
Interpretation of results
The vegetative forms stain pink/red because they take up the counterstain (Safranin) while the endospores take up the green from the Malachite green.
This is because, during smearing and heat fixing, the malachite green penetrates into the endospore with the help of the heat from the steam, and during the water-rinse, the dye is not easily washed away.
And for the vegetative forms, the dye is easily washed away because of their fragile outer covering, hence they take up the last stain which is the counterstain, hence they appear pink-red.
2. Dorner method for staining
Reagents:
Carbolfuchsin stain,
Decolorizing solvent (acid-alcohol)
Counterstain (Nigrosin solution)
Procedure
(Preparation of microscopic slide is adapted from the above procedure)
Staining procedure
- cover the smear with an absorbent paper.
- Saturate it with carbol-fuschin and heat fix by steaming over a boiling water bath or beaker for 5-10 minutes while adding more dye to the smear.
- Remove the absorbent paper and decolorize it with acid-alcohol for 1 minute; rinse with tap water and tap dry.
- Add a thin film of nigrosin reagent as a counterstain.
- Visualize the slide under the oil immersion lens (1,000X) for the presence of endospores.
Result
Vegetative cells appear colorless, while endospores are red.
Applications of the Endospore stain
- For detection of Firmicute groups of bacteria i.e Clostridium spp and Bacillus spp
- For identification of endospore producing bacteria in samples
- for differentiation of spore-producing bacterial from vegetative forms of bacteria
Advantages of endospore staining
- Being a Differential stain, allows you to identify specific bacteria that have an endospore
- It also allows the detection and presence of vegetative forms in bacterial culture, besides identifying the presence of endospore producing bacteria.
Disadvantages
- It can only specifically identify the presence of endospore-forming bacteria
NOTE
- Another endospore staining technique that is not commonly used is known as the Klein method of endospore staining.
- The difference between these Shcauffer Fulton and Klein staining techniques is the application of dyes i.e in Schahuffer Fulton stain, Malachite Green dye is used while in the Klein methodology, Methylene blue solution is used.
References
- Oktari A., Supriatin Y., Kamal M., Syafrullah H., The bacterial Endospore Stain on Schaufferton using methylene Blue Solution; Journal of Physics,2017.
- Marisse A. H., Anna Z., Endospore Stain Protocol; American Society of Microbiology. 2012.
Internet Sources
- https://laboratoryinfo.com/endospore-staining/
- 1% – https://quizlet.com/93824768/biology-flash-cards/
- 1% – https://quizlet.com/72357585/lab-midterm-flash-cards/
- 1% – https://quizlet.com/121171377/bacillus-flash-cards/
- 1% – https://quizlet.com/11226939/microbiology-lab-practical-flash-cards/
- 1% – https://en.wikipedia.org/wiki/Endospore
- <1% – https://www.wikihow.com/Remove-an-Iron-on-Transfer-From-Clothes
- <1% – https://www.ctahr.hawaii.edu/bnf/Downloads/Training/Rhizobium%20technology/Exercise%201.doc
- <1% – https://www.cartercenter.org/resources/pdfs/health/ephti/library/lecture_notes/env_occupational_health_students/MedicalBacteriology.pdf
- <1% – https://quizlet.com/85871953/stains-flash-cards/
- <1% – https://quizlet.com/192529190/microbiology-practical-review-flash-cards/
- <1% – https://en.wikipedia.org/wiki/Gram_stain
- <1% – https://en.m.wikipedia.org/wiki/Malachite_green
- <1% – https://ca.answers.yahoo.com/question/index?qid=20140530083604AAhVsS2
- <1% – https://brainly.com/question/10509484
- <1% – https://answers.yahoo.com/question/index?qid=20100704202926AA7Q6zs
- <1% – http://www.ruf.rice.edu/~bioslabs/bios318/staining.htm
- <1% – http://www.austincc.edu/microbugz/endospore_stain.php
- <1% – http://vlab.amrita.edu/?sub=3&brch=73&sim=208&cnt=2
- <1% – http://spot.pcc.edu/~jvolpe/b/bi234/lab/differentialTests/endospore_stain.htm
- <1% – http://shs-manual.ucsc.edu/policy/fecal-microscopic-examination