- The concept of confocal microscopy was initially developed by Marvin Minsky in the 1950s, at Harvard University with an aim of viewing the neural network without staining the tissues but it did not bear fruit due to lack of enough light source and a computerized system to store the large data.
- The work was later adapted by David Egger M. and Mojmir Petran, forming a multiple-beam confocal microscope in the late 1960s. they used a spinning disk known as Nipkow which they used to examine brain tissues and ganglion cells that were unstained. The technique was later modified and published by Egger forming a mechanical scanned confocal laser microscope, that was able to visualize images of cells.
- Later developments in science including the development of computers and laser technology and digital manipulations of images using algorithms, grew the advances in confocal microscopy, forming practically usable confocal microscopes by a series of scientists including G. Fred Brakenhoff (1979), Colin Sheppard, Tony Wilson, Brad Amos, and John White (1980s).
- The first commercial confocal microscope was developed in 1987 with improved optics and electronics, powerful lasers with high scanning efficiency.
- The modern confocal Microscope has all the possible integration of technology and mechanical components including optical components, which perform the primary function of the configuration by use of electronic detectors, a computer, and laser systems.
- The functioning of the confocal microscope is a collective role of all its components to produce an electronic image. To date, these microscopes have been used to investigate molecules, microbial cells, and tissues.
The price of a Confocal Microscope is from USD$500,000 to USD$1,000,000.

Principle of the Confocal Microscope
- Normally a conventional (wide-field) Microscope uses different wavelengths from a light source, to visualize and illuminate a large area of a specimen, forming fuzzy, murky, and crowded images, because cell sample images are captured from all directions, without a focal point.
- To avoid these issues, a Confocal Microscope is used. In wide-field or Fluorescent microscopes, the whole specimen receives light, receiving complete excitement and emitting light which is detected by a photodetector on the microscope. However, with the confocal microscope, point illumination is the principle working mechanism.
- A specimen is stained with fluorochrome is examined. When a beam of light is focused at a particular point of the fluoro-chromatic specimen, it produces an illumination that is focused by the objective lens to a plane above the objectives. The objective has an aperture on the focal plane located above it, which primarily functions to block any stray light from reaching the specimen.
- A measure of the illumination point is about 0.25 to 0.8 um in diameter, determined by the objective numerical aperture and 0.5 to 1.5 um deep, with the brightest intensity.
- The specimen normally lies between the camera lens and the perfect point of focus, known as the plane of focus. Using the laser from the microscope, the laser scans over a plane on the specimen (beam scanning0 or by moving the stage (stage scanning). A detector then will measure the illumination producing an image of the optical section. scanning several optical sections, they are collected in a computerized system as data, forming a 3D image. The image can be measured and quantified.
- Its outcome is also favored by the aperture found above the objective which blocks stray light.
- Images produced by the confocal microscope have a very good contract and resolution capacity despite the thickness of the specimen. Images are stored in the high-resolution 3D image of the cell complexes including their structures.
- The main characteristic of the Confocal Microscope is that it only detects what is focused and anything outside the focus point, appears black.
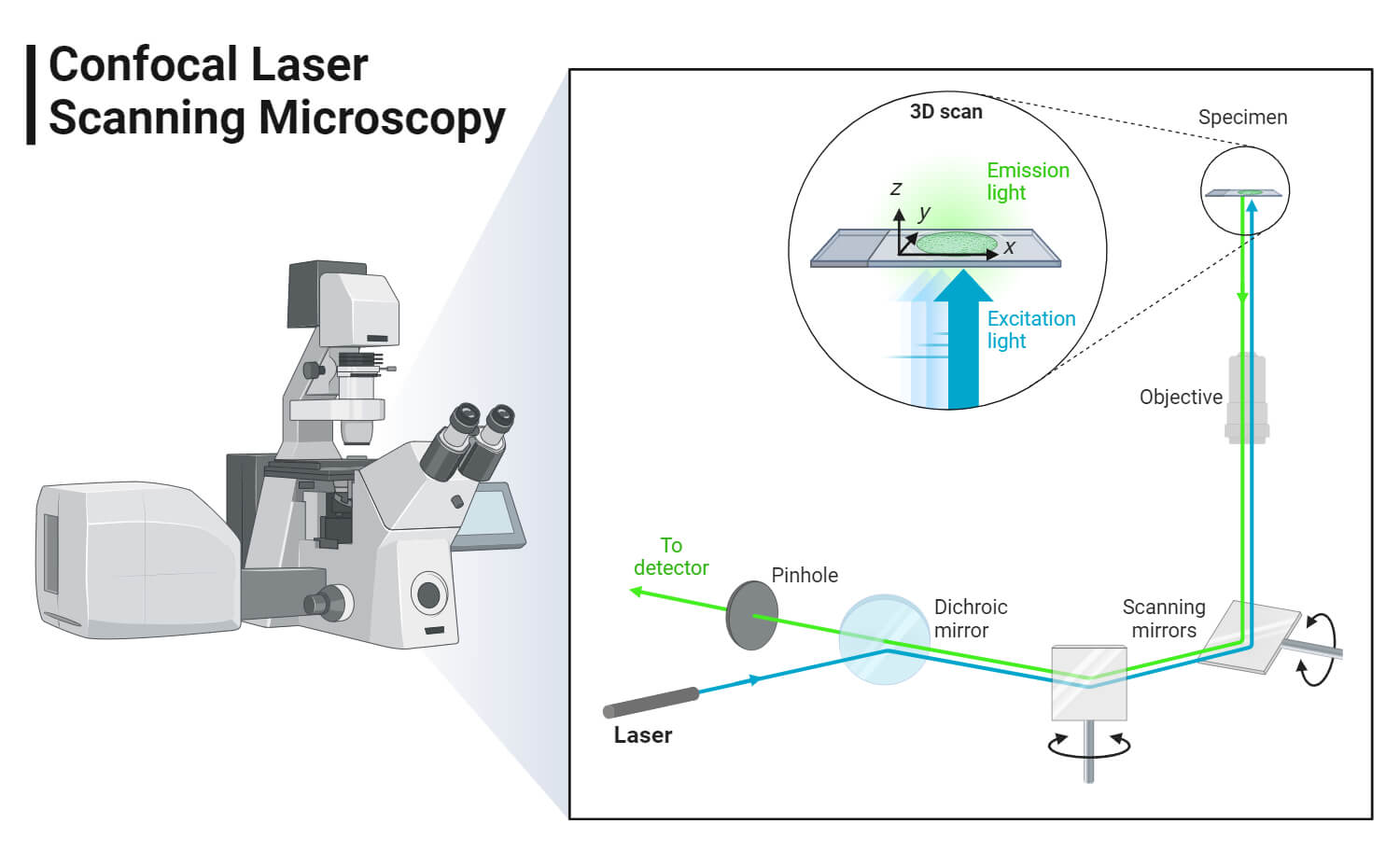
The image of the specimen is formed when the microscope scanner, scans the focused beam across a selected area with the control of two high-speed oscillating mirrors. Their movement is facilitated by galvanometer motors. One mirror moves the beam from left to right on the lateral X-axis while the second mirror translates the beam along the Y-axis. After a scan on the X-axis, the beam moves rapidly back to the starting point to start a new scan, a process known as flyback. No information is collected during the flyback process, therefore the point of focus, which is the area of interest is what is illuminated by the laser scanner.
Parts of the Confocal Microscope
The Confocal Laser Scanning Microscope is made up of a few components:
- Objective lens
- Out-of-focus plane
- In-focus plane
- Beam splitters
- Detector
- Confocal pinhole (aperture)
- Laser
- Oscillator Mirrors
Types of Confocal Microscope
- Confocal laser scanning Microscope – It uses several mirrors that scan along the X and Y axes on the specimen, by scanning and descanning, and the image passes through a pinhole into the detector.
- Spinning disk, also known as the Nipkow disk, is a type of confocal microscope that uses several movable apertures (pinholes) on a disc to scan for spots of light in a parallel manner over a specified plane, over a long period. The longer the time the less the excitation energy required for illumination, as compared to the Confocal laser scanning microscope. Lessened excitation energy reduces phototoxicity and photobleaching, hence it’s mainly used to image live cells.
- Dual spinning Disk or Microlens enhanced confocal Microscope -, it was invented by Yokogawa electric; it works similarly to the spinning disk, the only difference is, it has a second spinning disk with micro-lenses that is found before the spinning disk that contains the pinholes. The micro-lenses capture broadband of light focusing it into each pinhole, thus increasing the amount of light that is directed into each pinhole, reducing the amount of light that is blocked by the spinning disk. These Confocal Microscopes with enhanced Microlenses are much more sensitive than the spinning disks.
- Programmable array Microscope (PAM) – this type of confocal microscope uses a spatial light modulator (SLM – an object that imposes some form of spatially-varying modulation on a beam of light). The SLM has a set of movable apertures (pinholes), with arrays of pixels of opacity, reflectivity, or optical rotation. The SLM also has microelectrochemical mirrors that capture the image by a charge-coupled device (CCD) camera.
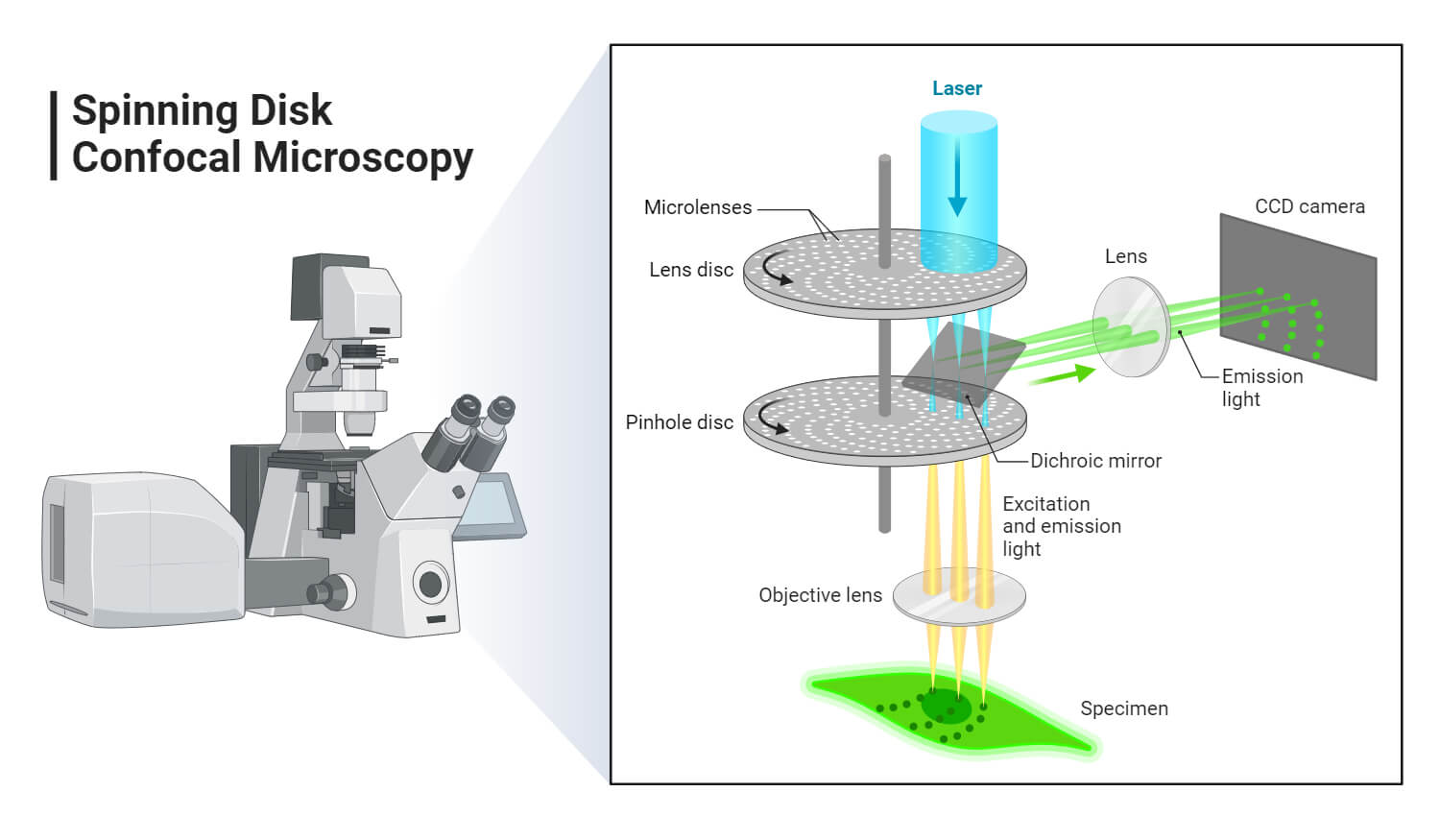
Each of the confocal microscopes has its advantages and disadvantages, but they all capture the images by recording the images, and sometimes they can be programmed to get high-density images, especially the Programmed array Microscope and the Spinning disk confocal Microscope.
Applications of Confocal Microscope
The Confocal Microscope is used in a wide range of fields including Biomedical sciences, Cells Biology, genetics, Microbiology, Developmental Biology, Spectroscopy, Nanoscience (nanoimaging), and Quantum Optics.
- In Biomedical sciences, it is used in the analysis of eye corneal infections, by quantifying and qualitatively analyzing the endothelial cells of the cornea.
- Used to identify the presence of fungal elements in the corneal stroma, during keratomycosis infection, or rapid diagnosis and quick therapeutic response.
- It is used in pharmaceutical industries, to ensure the maintenance of thin-film pharmaceuticals, allowing control of the quality and uniformity of drug distribution.
- It is used to retrieve data from some 3D optical storage systems. This has helped in quantifying the age of Magdalen’s papyrus.
Advantages of Confocal Microscope
- The advantage of the Confocal microscope is that it improves the outcome of the image because it analyses the image from one optical point to another, therefore there is no interference with scattered light from other parts of the specimen.
- They have better resolution and each point of interest is visualized and captured.
- It can be used to study live and fixed cells
- It can be used to collect serial optical sections.
- It illuminates uniformly across the focus points.
- They adjust their magnification electronically, without changing the objectives, by a factor known as the zoom factor.
- It generates 3D sets of images.
Limitations of Confocal Microscope
- They have a limited number of excitation wavelengths, with very narrow bands.
- They are expensive to produce the ultraviolet rays used by the Confocal Microscopes
- They are also expensive to manufacture and purchase.
References
- https://www.olympus-lifescience.com/en/microscope-resource/primer/techniques/confocal/confocalintro/
- https://www.britannica.com/technology/microscope/Confocal-microscopes
- https://ibidi.com/content/216-confocal-microscopy
- https://www.olympus-lifescience.com/en/microscope-resource/primer/techniques/confocal/
- https://en.wikipedia.org/wiki/Spatial_light_modulator
- https://en.wikipedia.org/wiki/Confocal_microscopy

good
more notes on confocal microscope