Column chromatography is a method that separates and purifies the components of a mixture on a vertical column or tube based on their interactions with the stationary and mobile phases.
It is a simple method that is used for applications like separating complex mixtures, isolating individual components, purifying chemical compounds, and monitoring chemical reactions. This method involves a vertical column that is filled with stationary phase and the mobile phase flows through thepacked column to separate the components of the mixture.
Principle of Column Chromatography
Column chromatography separates components of a mixture based on their different interactions with the stationary and the mobile phase. This method uses a vertical column packed with the stationary phase. The sample is loaded and then carried throughout the column by the mobile phase either by gravity or air pressure. Compounds that bind more strongly to the stationary phase move slowly while those with weaker interactions move more quickly. This difference in movement causes the components to separate as they pass through the column. The separated components are collected and analyzed.
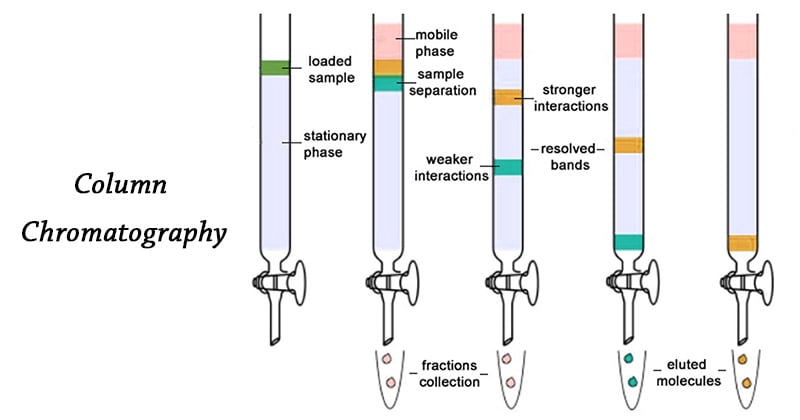
Components of Column Chromatography
- Sample/Analyte: This is the mixture that needs to be separated and analyzed. It is usually dissolved in solvent before loading into the column for better separation and smooth loading.
- Column: This is a vertical tube that provides the space for separation. It holds the stationary phase. The bottom of the column contains a cotton or glass wool plug to keep the stationary phase in place.
- Stationary Phase: This is the solid material packed inside the column. They act as adsorbent and interact with compounds in the mixture differently leading to separation. It can be packed as particles or coated as a thin layer inside the column. Commonly used materials for stationary phase are silica gel, alumina, ion-exchange resins, polymer-based resins, and cellulose. Among these, silica and alumina are the most widely used.
- Mobile Phase: The mobile phase is the solvent used in chromatography. It flows through the column and carries the sample with it. It flows either by gravity or applied pressure. In column chromatography, the mobile phase is a liquid or a gas. Some of the solvents used in column chromatography are hexane, methanol, water, and chloroform.
- Pump: It is used to control the flow rate of the solvent through the column. Pumps are not required in gravity column chromatography as the mobile phase flows by gravity alone. However, methods like flash chromatography and HPLC need pumps to supply higher flow rates and pressure.
- Injectors: They are used to introduce the sample mixture into the top of the column in a controlled way.
- Detector: The detector monitors the separation of compounds from the column. It helps to identify and quantify the separated compounds.
- Fraction collector: This is used to collect the separated compounds into separate tubes for further analysis or use.
Types of Column Chromatography
Column chromatography can be divided into different types based on the flow of solvent and the separation method.
Based on the flow of solvent through the column:
- Gravity chromatography: In this method, the solvent flows down the column naturally using gravity. This method is simple, cost-effective, and does not need complex pressurized systems which is suitable for small-scale laboratory use. However, it has low resolution and is not suitable for separating complex mixtures.
- Flash chromatography: This method uses pressurized air or pumps to push the solvent quickly through the column. Flash columns do not require clamps at the bottom to control the flow like in gravity columns. Compared to traditional gravity chromatography, this method is faster and provides better resolution. However, it can separate only small amounts of sample.
- High-pressure liquid chromatography (HPLC): It is also called high-performance liquid chromatography. This method uses very high pressures up to 5000 psi or more. It provides high resolution and can work using small particle-sized stationary phases. It is suitable for analyzing small sample volumes and detecting very low concentrations of compounds.
Based on the separation mechanism:
- Adsorption Chromatography separates compounds based on their adsorption properties onto a solid stationary phase. Compounds with stronger adsorption move slowly through the column and those with weaker adsorption move faster.
- Partition Chromatography separates components based on their partitioning between the stationary and mobile phases. Both phases are liquid in this method. It is useful for separating polar compounds like organic acids and amines.
- Gel Filtration or Size Exclusion Chromatography uses a porous stationary phase to separate molecules based on their size. Smaller molecules are trapped in the stationary phase and elute later than larger molecules. It is commonly used for purification of proteins, enzymes, and polymers.
- Ion Exchange Chromatography is used for separation of charged biomolecules like proteins, peptides, and nucleotides. In this method, the stationary phase contains charged groups which allow the separation of components based on their net charge.
- Affinity Chromatography: In this method, separation is based on highly specific interactions between a ligand attached to the stationary phase and the target molecule in the mixture. It is used for isolating biomolecules like antibodies, enzymes, or receptors.
Sample preparation
Sample preparation is an important step in chromatography as it directly affects the accuracy of the separation process. Proper sample preparation helps remove impurities that might damage or interfere with the separation process. It involves steps like dissolving the sample in the right solvent, filtering, or concentrating the sample.
In column chromatography, the sample should be dissolved in a suitable solvent before loading. The solution must be filtered or centrifuged to remove any particles that could clog the column or disrupt the flow. Sample preparation may also include advanced methods like solid-phase extraction or derivatization depending on the type of sample.
Procedure or Steps of Column Chromatography
1. Column Preparation
- At first, a suitable column is selected based on the separation method.
- A small plug of glass wool or cotton wool is placed at the bottom of the column to prevent the adsorbent from leaking while allowing solvent flow.
- A layer of sand is added above the cotton to create a flat base to prevent uneven packing.
- The column is also partially filled with solvent to help in uniform packing and remove air pockets.
- This column is vertically attached to a ring stand. A pinch clamp is also fixed to the bottom outlet to control solvent flow. The clamp is closed to prevent solvent or adsorbent from draining.
2. Column Packing
- Before packing, a suitable stationary phase material is selected. Silica and alumina are the most commonly used packing materials.
- There are two methods of packing the stationary phase in the column:
- Dry packing: In dry packing, the dry adsorbent is added directly into the column and the mobile phase is allowed to flow through it. This method is easier and quicker but can form bubbles inside the column.
- Wet packing: In wet or slurry packing, the adsorbent is first mixed with the solvent to form a slurry and then transferred into the column by pouring or pipetting. Wet packing is usually preferred as it results in more uniform packing and eliminates air bubbles.
- After filling the column, another thin layer of sand is added on top. This helps with even sample application and protects the stationary layer from disruption when adding the sample or mobile phase.
3. Sample Loading
- The sample is dissolved in a small amount of solvent and loaded to the top of the packed column.
- The sample container is rinsed with small amount of solvent and transferred to the column to transfer the entire sample.
- The clamp is briefly opened to allow the sample to enter the adsorbent bed.
4. Separation/Elution
- The solvent is slowly added on top of the column to start the separation process.
- The column must never run dry to prevent cracking so the solvent must be continuously added to maintain it above the top of the adsorbent.
- The compounds in the sample will separate as they move through the stationary phase at different rates.
- The solvent can be changed to a more polar one to elute strongly retained compounds.
5. Fraction Collection and Analysis
- The separated compounds or fractions are collected in labeled test tubes or vials as they exit the column.
- The collected fractions are analyzed to determine the composition of each fraction.
- Thin Layer Chromatography (TLC) can be used to monitor the progress of separation and compare the fractions.
- The solvent is evaporated to isolate the purified product.
Factors Affecting Column Chromatography
- Column dimensions including the length and internal diameter of the column can affect separation. Longer columns improve resolution but take more time and pressure while shorter ones allow faster analysis. Similarly, wider columns can hold more sample but narrower columns use less solvent and improve resolution.
- The particle size of the stationary phase is also important for separation. Smaller particles provide more surface area for interactions which improves the separation but very fine particle can slow the flow.
- Mobile phase properties like solvent type, polarity, and pH can directly affect how the compounds move through the column.
- Another factor is temperature. Higher temperature can reduce the viscosity of the mobile phase and result in faster and more efficient separations.
Common Products and Manufacturers of Column Chromatography
| Common Products | Manufacturers |
| Hypersil GOLD, Acclaim, Dionex columns | Thermo Fisher |
| InfinityLab, ZORBAX, Polaris | Agilent Technologies |
| XBridge, ACQUITY, SunFire, BioResolve | Waters Corporation |
| Bio-Scale, Econo-Pac, Econo-Column, Aminex | Bio-Rad Laboratories |
| Vantage, Quikscale, IsoPak | Sigma-Aldrich |
| CHIRALPAK, CHIRALCEL | Daicel Corporation |
| Kinetex, Luna Omega, Lux, Claricep | Phenomenex |
| RediSep columns, CombiFlash systems | Teledyne |
Applications of Column Chromatography
- Column chromatography is used to isolate and purify components from complex mixtures.
- It can be used to study different components of a sample.
- It is used in the pharmaceutical industry to purify active drugs ingredients.
- In food industry, it can be used to check additives or harmful contaminants in food products.
- It is also used in environmental testing or monitoring to identify pollutants in environmental samples.
- It is used to study biological macromolecules like proteins.
- It can also be used to clean up samples before analytical methods like mass spectrometry.
Advantages of Column Chromatography
- Column chromatography helps to separate complex mixtures into their individual components which is easier to study and analyze.
- It has a simple setup that requires basic equipment and is easy to set up in a lab.
- It can work with different kinds of compounds so it is useful for a wide range of applications.
- It is also simple and cost-effective for routine use in many labs.
- Different types of solvents can be used in this process.
- After separation, the compounds can be collected and used again for further analysis.
Limitations of Column Chromatography
- The process can be time-consuming.
- It is less effective in separating very small amounts of sample.
- This method can be expensive compared to simpler methods like thin layer chromatography.
- It needs large amounts of solvents which can be costly.
- The process has to be continuously monitored to avoid problems like column drying out or uneven flow.
Troubleshooting and Safety Considerations
- Poor separation can occur due to incorrect solvent polarity, unsuitable stationary phase, sample overloading, or poor column packing. This can be solved by adjusting solvent polarity, using suitable stationary phase, and ensuring proper column packing.
- If the sample is not fully dissolved in the solvent or loaded too quickly and unevenly, smearing of bands may occur.
- Any blockage at the bottom of the column must be cleaned regularly to avoid clogged columns.
- The adsorbent can crack if it is left to dry so the column must be continuously wet with solvent to avoid cracks.
- Fractions collected after separation should be clearly labeled for analysis.
- Safety procedures should be adopted like wearing personal protective equipment (PPE), working in well-ventilated fume hood, proper waste disposal, and handling glassware safely.
Recent Advances and Innovations
- Development of novel particle designs like sub-2-micron particles, superficially porous particles (SPPs) or core-shell particles, and monolithic columns have improved the speed and efficiency of separation.
- New stationary phase materials like polymeric stationary phases and mixed-mode phases have been developed.
- Precision-engineered column packing methods like droplet microfluidics and centrifugal parallel packing help pack the particles more evenly and provides better control over the particle size, shape, and chemical properties.
- Use of 3D printing to create chromatography columns improves separation and also reduces cost.
Conclusion
Column chromatography is a widely used method for separating, purifying, and analyzing compounds from complex mixtures in both research and industry. This is a simple and versatile method that can be used for both analytical and preparative applications. It is commonly used in research, pharmaceuticals, environmental testing, and biochemical analysis. However, it can be time-consuming and requires large amounts of solvent. Despite its limitations, advances like automation and improved columns have made column chromatography faster and more efficient.
References
- Abbas, A. (2023, February 21). Column Chromatography-Principle, Types, Applications. Retrieved from https://themasterchemistry.com/column-chromatography-principle-types-applications/
- Column Chromatography 101: A Clear and Simple explanation. (2024, December 9). Retrieved from https://uhplcs.com/column-chromatography-101/
- Column chromatography procedures. (n.d.). Retrieved from https://www.orgchemboulder.com/Technique/Procedures/Columnchrom/Procedure.shtml
- Column chromatography. (2025, May 12). Vedantu. Retrieved from https://www.vedantu.com/chemistry/column-chromatography
- Column chromatography. (n.d.). Retrieved from https://organiclab.welderco.host.dartmouth.edu/CC/index.htm
- Column Chromatography: Definition, Purpose, Types & Applications. (2024, February 27). Chemistry Learner. Retrieved from https://www.chemistrylearner.com/chromatography/column-chromatography
- Hroncich, C. (2024, October 10). New advances in column technology spotlighted at the International Symposium on Chromatography. Chromatography Online. Retrieved from https://www.chromatographyonline.com
- Introduction to Column Chromatography Methods and instrumentation. (n.d.). Retrieved from https://www.bio-rad.com/en-np/applications-technologies/introduction-column-chromatography-methods-instrumentation?ID=MWHB7PIVK
- Lewis, S., & Lenehan, C. (2013). Liquid and Thin-Layer chromatography. In Elsevier eBooks (pp. 586–589). https://doi.org/10.1016/b978-0-12-382165-2.00246-4
- Pan, S. (2025, March 4). Column Chromatography – Principle, steps, parts, uses, advantages – Biology Notes online. Retrieved from https://biologynotesonline.com/column-chromatography-principle-steps-parts-uses/
- Sample Preparation Techniques | Thermo Fisher Scientific – NP. (n.d.). Retrieved from https://www.thermofisher.com/np/en/home/industrial/chromatography/chromatography-learning-center/chromatography-consumables/sample-prep.html

A clear and concise presentation. Keep up the good work
amazing content , really helpful in exam preparation
notes are very useful to undergraduates students who are in the field of chemitry and pharmacy
Really, it was very helpful. I understand it very well for first time…. Thanks
Yes, the website provide clear elocution about column chromatography!
Best notes
it is very helpful to me a very nice website