Citric acid (CA) has been widely produced as essential weak organic acid with extensive use in the pharmaceutical and food industries due to being a common metabolite found in animals and plants alike. Citric acid can be produced on a mass scale via synthetic or natural methods of extraction process or manufacturing the biomolecule. The natural process involves the extraction of citric acid from citrus plants like oranges, lime, lemons, etc. Whereas in the synthetic method, citric acid is produced by two approaches, i.e.
- The chemical approach of citric acid synthesis with the use of enzymes.
- A biological approach involving fermentation by the use of microorganisms like bacteria, yeast, etc.
Industrial production of CA using fermentation by microorganisms uses the ‘submerged’ fermentation technique where species like Aspergillus niger (fungi) and Candida (yeast) metabolize various carbon sources such as molasses and starch-based culture media. Other fermenting techniques like surface fermentation and solid-state fermentation take use of agro-based residues as the carbon source for the mass production of citric acid.
Properties of Citric Acid
- The citric acid (CA) is a naturally occurring weak acid with a molecular formula C6H8O7 and IUPAC molecular name being ‘2-hydroxy-1,2,3 propane tricarboxylic acid’.
- This tricarboxylic acid has a molecular weight of 210.14 g/mol.
- They naturally occur in citrus plants like lemon and also is a commonly found metabolite in animals.
- Citric acid is a common primary metabolite derived from carbohydrates, proteins, and fats of the Krebs (tricarboxylic) cycle for energy needs.
- The melting point of CA is 153 degrees Celsius.
- When CA is in the pure state, it is soluble in water and other polar solvents like acetone, ethanol, etc.
- At room temperature, citric acid is present in the solid state.
- Produced massively via chemical (enzymatic) biosynthesis or biologically by the microbial process of fermentation.
Historical Events
- Karl Scheels in the year 1874, was the first scientist to isolate citric acid by crystallization process from lemon juice.
- In 1880, two scientists named, Adams and Grimaux synthesized citric acid from glycerol.
- In 1893, Wehmer tested fermentation carried out by the Penicillum gaucum species and observed the accumulation of CA as a by-product of calcium oxalate.
- In 1917, James Curie a food chemist demonstrated that Aspergillus niger yielded a larger amount of citric acid than other organisms at the time in a sugar-based medium.
- Millard in 1922, accumulated citric acid by limiting nutrients in the bioreactors where fermentation was carried out by Aspergillus niger.
- By the year 1965, yeasts were introduced along with alkanes and carbohydrates as substrates.
- In 1984, the ‘Submerged fermentation technique’ was introduced to produce large quantities of citric acid in the US industries.
Citric Acid Biochemistry and Accumulation
In 1953, Hans Adolf Krebs was awarded Nobel Prize in Physiology or Medicine for the discovery of the Citric Acid cycle. Citrate or Citric acid is an intermediate metabolite in the Krebs cycle (also known as Tricarboxylic acid or Citric acid cycle) found in bacteria, plants, and animals. To produce citrate, the condensation of acetyl CoA to oxaloacetate is catalyzed by the citrate synthase enzyme. Citrate acts as a substrate for the downstream reaction of the TCA cycle and the cycle ends with the renewal of oxaloacetate. This citrate is exported to the cytoplasm from mitochondria to serve as a substrate and regulate various biochemical synthesis of biomolecules.
It suggested that enzymes responsible for the degradation of CA in the Krebs cycle must be deactivated to accumulate Citric Acid. But it is also observed that the Krebs cycle is active for biomass production. Another hypothesis is that tricarboxylate transporter activity competes with aconitase for binding to CA. The transporter has a higher affinity for CA where it ejects the citrate out of mitochondria to cytoplasm without inhibiting kreb cycle enzymes.
Microorganisms Used in CA Production
Aspergillus niger is the highly recommended industrial producer of citric acid. They flourish in sugar medium containing salts at pH 2.5-3.5 and excrete large volumes of CA. An estimated practical yield of CA is 70 % of the theoretical estimate which is 112g per 100g of sucrose.
Many organisms like Penicillin sp., Eupenicillin sp., Botrytis sp., Absidia sp., Ustulina vulgaris, and more can also accumulate citric acid in a sugar and inorganic salt medium. Yeast species belonging to the genera of Hansenula, Candida, Torula, Saccharomyces, Pichia, etc, can produce CA from carbohydrates and n-alkanes. However, the production of CA via these organisms may not seem economical due to the accumulation of unwanted by-products like isocitric acid. A remedial approach could be the development of a mutant strain with lower aconitase activity.
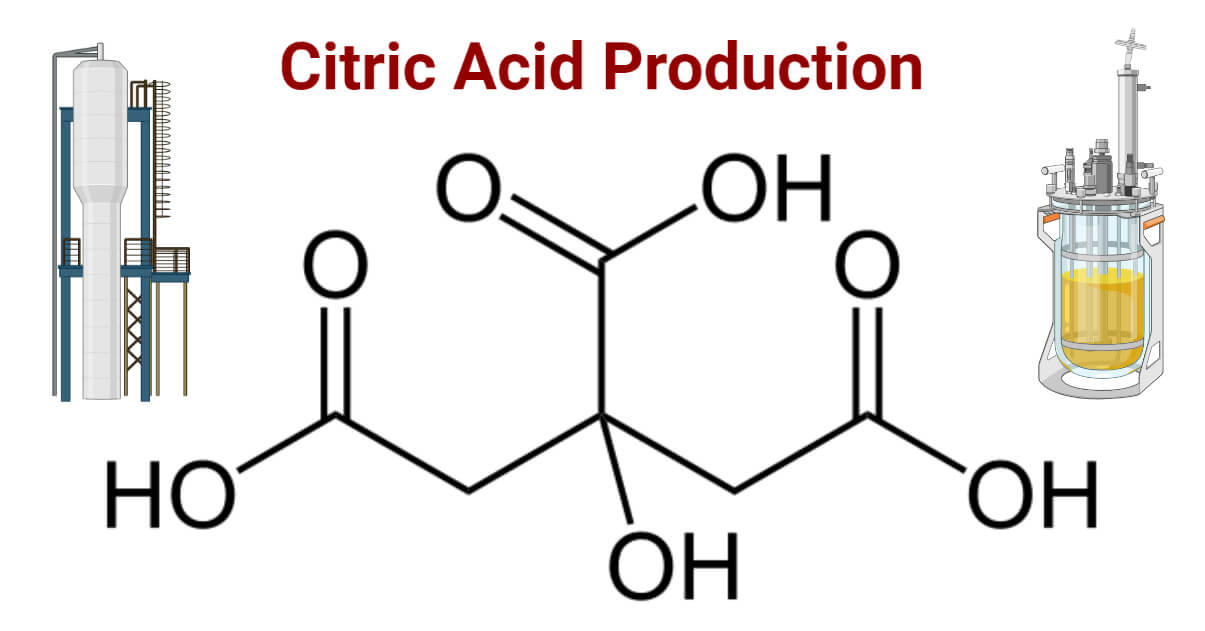
Industrial Citric Acid Production Approach
An estimated 99% of CA mass production worldwide is carried out by microbial processes, surface or submerged culture methods. The final product obtained is marketed as a monohydrate acid or an anhydrous salt. About 70% of total CA is used in the beverages and food industries as an acidifier or antioxidant for the preservation and enhancement of flavors, and aromas in ice cream, marmalades, and fruit juices. 20% of total CA produced is utilized by pharmaceutical industries as blood preservatives, vitamin preservant as antioxidants, effervescent, and iron citrate in cosmetics, tablets, and ointments. In the chemical industry, it is used as a foaming agent for softening textiles and a phosphate substitute for hardening cement.
a. Surface Culture
Surface culture is divided into solids and liquids, though the liquid approach provides higher economic production.
- The culture medium is put on a shallow aluminium tray 5-20 cm deep and maintained at 5-6 pH.
- Uniform air circulation and maintenance of temperature and humidity are ensured in the fermentation chamber.
- Then A. niger spores are blown onto the culture medium surface for 5-6 days with uniform passage of dry air.
- The pH of the medium is changed to 1.5 to 2.
- After 24 hours of incubation, the spores germinate and the white mycelium forms on the medium surface.
- The moulds start to utilize the sugar content of the medium and after complete utilization, the residual liquid is extracted from the mycelium mesh.
- This residual liquid contains CA and A. niger produces a small volume of CA as the main metabolite in this method.
Inoculum Preparation
- A suitable and high-yielding A.niger strain is selected from the stock culture.
- Glass vials that are incubated for 10-14 days and then sporulating media are infected with the selected stock culture.
- In the sporulating media, trace elements like zinc, iron salts, and manganese should be maintained.
- To suspend the mature spores, the appropriate diluent is used like water with the wetting chemical sodium lauryl phosphate.
Media Preparation
- For CA production, the medium must contain a carbon source such as sucrose and inorganic salts.
- Sucrose and beet molasses are preferred carbon sources for CA fermentation media.
- To achieve a high CA yield, a culture medium with less than 15% sucrose is beneficial.
- The use of fructose or glucose instead of sucrose leads to lower citric acid output.
- On a commercial scale, beet molasses are the preferred carbon source for A. niger.
- Beet molasses are rich in inorganic salts, hence they are pre-treated with ferricyanide or ferrocyanide to diminish excess inorganic salt.
- Other elements added to the medium for optimum growth of microorganisms and CA production are nitrogen, phosphorus, potassium, and magnesium.
- Salts like ammonium nitrate, magnesium sulfate, potassium mono hydrogen phosphate, etc, are also added in small amounts.
- pH is adjusted by the addition of HCl to 3.4 – 3.5 as lower pH facilitates sterilization, less contamination, suppression of oxalic acid formation, and good yield of citric acid synthesis.
Fermentation
- The prepared media of 1 to 2.5 cm thick is placed in a shallow pan.
- The inoculum spores are spread onto the medium surface with uniform aeration to allow even distribution of spores in the media.
- Incubation is done at 30-40 degree celsius temperature.
- The temperature is held constant during fermentation with air current ventilation to allow efficient gaseous exchange.
- After 24 hours of inoculation, the germinating spores produce a thin layer of mycelium on the medium surface.
- the pH of the medium drops to 1.5 – 2.0 as ammonium ions are formed.
- Presence of oxalic acid and yellow pigment after 30 hours of fermentation is an indication of high iron concentration and halted citric acid production.
- Fermentation ceases after 8-14 days with thick mature white mycelium floating atop of liquid media.
- The ratio of surface area to medium volume is proportional to the rate of sugar conversion to CA.
- In the shallow pan method, the ratio is reduced and the yield of CA increases.
- Under appropriate conditions, sugar is exponentially converted to citric acid.
- This method yields between 1.2 and 1.5 kilogrammes of citric acid monohydrate per square metre of fermentation surface per hour.
Recovery
- Mycelium is separated from the mycelium broth.
- Mycelium is pressed to extract any intracellular citric acid present.
- The filtered broth is treated with Calcium hydroxide and then treated with sulphuric acid in equivalent volume to obtain CA and calcium sulfate is generated as a precipitate.
- Filtration is employed to isolate precipitate, and impure citric acid is generated by demineralizing and decolorizing it with activated carbon.
- After evaporation, pure CA crystals are obtained.
b. Submerged Culture
This fermentation approach recruits Aspergillus japonicus, a black aspergillus. Submerged culture is done in stainless steel bioreactors with a cooling system, aeration, impeller, etc, equipment. Carbon source substrates include maize starch, beet molasses, etc, and for nitrogen supplements, ammonia is utilized. The culture medium is maintained at 30 degrees celsius when A. japonicus is added to the culture. The use of batch bioreactors generates 1500 kg of CA and 500 kg of biomass from using 2500 kg of glucose and 860 kg of O2. The quality of the metal recruited to produce fermenters, mycelium structures, and oxygen delivery systems, greatly influences the production rate. An alkane metabolizing fungus, Candida lypolytica is employed in a continuous fermentation approach for citric acid production, yielding 45% more CA than standard procedure.
Inoculum Preparation
- A suitable and high-yielding A. japonicus strain is extracted from the stock culture.
- As an inoculum for fermentation, mycelial mats are used known as pellets.
- In the seed fermenter, 15% molasses sugar is used to induce spore germination.
- To stimulate high mycelial production, the appropriate concentration of cyanide ions is introduced to the medium.
- To avoid regular mycelium production which is seen in the case of lower cyanide concentration, high cyanide concentration is required for mycelial pellet formation.
- In 24 hours and at 32 degrees Celsius, the spores germinate forming 0.2 to 0.5 mm pellets.
- The pH drops to 4.3, and pellets are used as a starter culture for production fermenters.
Media Preparation
Same preparatory protocol as the surface media culture is made.
Fermentation
- Stainless steel fermenters are used to avoid heavy metal leaching.
- Mycelial pellets that are grown in a seed fermenter are transferred to the steel production fermenter with a minimum holding capacity of 1000 litres and maintained at 30 degrees Celsius temperature.
- The loose and few branched filamentous mycelia with no chlamydospores are observed to produce lower citric acid.
- The iron-to-copper ratio is essential for mycelial growth and induces pellet form, which causes optimum CA production.
- The optimum oxygen concentration for high CA yield is 20 – 25 % of the saturation value during fermentation.
- The aeration rate should be maintained at 0.2 – 1 volume per minute during the acid production phase.
- To avoid foaming issues, lard oil, an antifoaming agent is utilized.
Recovery
Similar protocols as applied in the surface culture media.
Limiting Factors during Citric Acid Production
Fungal Species
Many fungi species such as A. niger, Penicillium luteum, Ustulina vulgaris, etc., have been demonstrated to produce citric acid, hence it becomes crucial to select quality and high-yield strains for optimum CA production. A. niger is the widely preferred strain because they are highly productive, simple to cultivate, and produce a very low amount of unnecessary by-products like oxalic acid.
Carbon Source
To produce massive amounts of quality citric acid, sugars that are easily assimilated and metabolized by the microorganisms used. If the microbe produces hydrolytic enzymes effective at the lower pH required for fermentation, polysaccharides can be a useful raw material. Sucrose is preferred over glucose as a carbon source because A. niger possesses an invertase enzyme in the mycelium which catalyzes the hydrolysis of sucrose into glucose and fructose.
Widely utilized industrial carbon sources are sugar beet molasses, lower-quality sugarcane byproducts with levels of cations, and glucose syrup from starch hydrolysis. The highest productivity is seen in the medium containing 15 – 22% sugar.
Nitrogen and Phosphate
Complex media containing beet molasses are rich in nitrogen and laboratory-prepared media are supplemented with ammonium sulfate and nitrates, which favours lower pH essential for fermentation. Urea can also be used as a nitrogen source for media preparation. The use of phosphate promotes biomass growth due to enhanced secondary reactions.
Culture Medium pH
There are two stages of pH fluctuation seen. For spore germination, the pH of the medium should be lower than 5. And during the production phase, the pH drops to 2 or less due to the absorption of ammonium ions by germinating spores which leads to the release of protons, aiding in citric acid production. Lower pH also decreases the chance of pathogen contamination and inhibits the production of undesirable organic acids like oxalic and gluconic acids.
Aeration
Fluctuations in the aeration can have a detrimental impact on CA production. Lower levels of CO2 are caused due to high aeration rate. CO2 is an important substrate for pyruvate carboxylase enzyme that replenishes oxaloacetate for citrate synthase affecting CA output. Higher levels of carbon dioxide are also not beneficial for optimum citrate biomass.
Trace Elements
Balanced levels of trace elements Mn, Fe, Zn, Cu, etc, are necessary for fungal growth and high CA output. Levels to maintain for :
- Zinc – 0.3 ppm
- Iron – 1.3 ppm
- Manganese – 3 mg/l
Manganese regulates many cellular functions like sporulation, cell wall synthesis, and the production of secondary metabolites.
Conclusion
Citric acid or CA is a weak organic acid commonly found in all organism and commercially they are used as a preservative, acidulant, and flavouring agent in beverages, foods, cosmetics, and pharmaceutical drugs. Produced industrially via fermentation approach by microorganisms of fungal and bacterial species like Aspergillus niger, Escherichia coli, etc.
The fermentation process involves several regulated steps like sterilization, inoculation, media preparation, fermentation, recovery, and downstream processes. The sterilized substrate (any carbon source like molasses, sucrose, etc.) is inoculated with CA-producing microorganisms in a culture medium. The inoculated substrate is transferred to a bioreactor where further fermentation is achieved under optimum conditions of temperature, aeration, and pH. Microorganism metabolizes substrate molecules and releases citric acid as a by-product. After the completion of the fermentation process, the fermented broth is sent for recovery and purification of CA which involves steps like filtration, crystallization, and drying to form anhydrous white crystals to be sold for commercial purposes.
References
- Production of Citric Acid – https://biologyreader.com/production-of-citric-acid.html
- Vandenberghe, Luciana PS, et al. “Microbial production of citric acid.” Brazilian Archives of Biology and Technology 42 (1999): 263-276.
- Max, Belén, et al. “Biotechnological production of citric acid.” Brazilian journal of Microbiology 41 (2010): 862-875.
- Citric Acid Production – https://microbiologynote.com/citric-acid-production/
- Citric acid – https://en.wikipedia.org/wiki/Citric_acid
- Solvents – https://www.thermofisher.in/chemicals/en/brands/qualigens/solvents.html
