B cell or B lymphocyte is a type of lymphocyte (white blood cells) involved in the humoral immunity of the adaptive immune system as they differentiate into plasma and produce antibodies.
- Besides, B cells are also considered professional antigen-presenting cells as these can detect antigens present on the surface of bacteria and viruses.
- Like in all white blood cells, B lymphocytes are also generated in the bone marrow, which is then exported to the periphery.
- Every day, millions of B lymphocytes are produced in the bone marrow, and the rapid generation of these cells is carefully monitored by a regulated sequence of events.
- B cells are fewer in number than T cells as these account for about 20% of the blood lymphocytes in the body, whereas the rest are T cells.
- B cells are essential cells of the immune system since these are a part of the humoral or antibody-mediated immunity essential for the successful removal of antigens from the body.
- These cells have receptors on the surface known as B cell receptors. The B cell receptor is a macromolecular complex that is built with the help of IgM and IgD.
- Since the receptors are present on the surface, these can detect antigens that occur on the surface of pathogens.
- Another distinguishing feature of B lymphocytes is that these mature within the bone marrow, unlike T lymphocytes that are moved to the thymus for maturation.
- The mature B cells occur outside the lymph nodes, and these have a shorter lifespan and thus immediately mature to produce antibodies.
- B cells do not act against tumors or transplants and also do not move to the site of infection. Instead, they produce immunoglobulins that move to the site of action.
B cell (B lymphocyte) Types
B cells have been differentiated into four distinct groups; transitional, naïve, plasma, and memory cells.
1. Transitional B cells
- Transitional B cells are the intermediate B cells that are the link between the immature B cells in the bone marrow and mature B cells in the lymphoid organs.
- Transitional B cells are the cells that are differentiated from the myeloid progenitor cells in the bone marrow but have not matured.
- Transitional B cells can be found in the bone marrow, peripheral blood as well as spleen but only a small fraction of the immature B cells can survive the transitional phase before maturing into matured cells.
- After leaving the bone marrow, these cells are subjected to a number of checks to ensure that they do not produce autoantibodies.
- Transitional B cells can be found in two transitional stages; T1 and T2. T1 stage is the period between the migration of the cells from the bone marrow to the point of entry into the spleen, whereas the T2 stage occurs within the spleen, where the cells develop into mature B cells.
2. Naïve B cells
- Naïve B cells are the mature B cells that haven’t yet been exposed to antigens.
- Naïve B cells are the B cells at a stage of differentiation where the cells either develop into plasma cells or memory cells after exposure to a specific antigen.
- These cells occur in the secondary lymphoid organs and have passed the transitional stage of B cell development.
- Naïve B cells have recently divided into a new subcategory of B cells called Breg cells or regulatory B cells. These cells are paired with naïve T cells and act in regulating the response of T cells.
3. Plasma Cells
- Plasma cells or plasma B cells, or effector B cells are the white blood cells that are differentiated from naïve B cells that secrete large quantities of antibodies in response to activation by antigens.
- The production of plasma cells requires that the naïve B cells present an antigen to a helper T cell. This activates the T cell, which in turn activates the B cell.
- Some plasma cells can be also be activated by a second process called T-cell independent antigen stimulation. The plasma cells produced by this method can only secrete IgM antibodies.
- The antibodies produced by plasma cells are specific to the antigen initially processed by the naïve T cell during activation. A plasma cell, thus, cannot secrete more than one type of antibody.
- Plasma cells have a shorter lifespan as compared to the memory B cells, and these move through the body according to the distribution of cytokines to produce antibodies closer to the site of infection.
- Plasma cells are usually large in size with abundant cytoplasm and a characteristic eccentric nucleus. The differentiated plasma cells express fewer surface antigens like CD27++ but do not express CD19 and CD20.
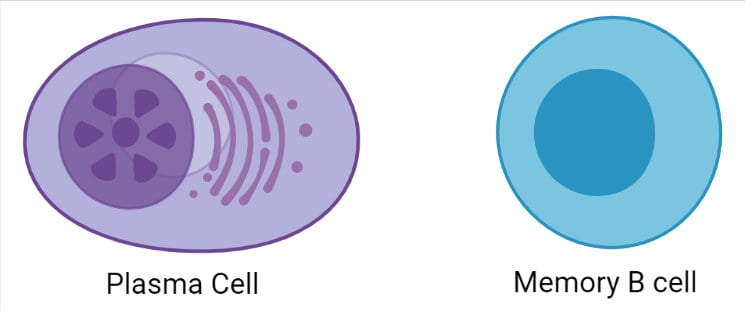
4. Memory cells
- Memory B cells are lymphocytes that are differentiated from naïve B cells during maturation and circulate through the bloodstream in a stationary phase.
- The cause of differentiation of naïve cells into plasma cells and memory cells is not yet understood.
- Memory B cells have a much longer lifespan as these can move through the bloodstream for years.
- Memory B cells are distributed throughout the body, and like plasma cells, these also have an affinity for one type of antigen.
- The primary function of memory cells is that these cells memorize the characteristics of the antigen that activated the parent B cell so that if the memory cell encounters the antigen again, it can trigger a stronger secondary immune response.
- Memory B cells are developed within the germinal centers of lymphoid organs where the differentiation can occur either by a T cell-dependent mechanism or a T cell-independent mechanism.
- Following differentiation, memory B cells might remain concentrated at certain areas in the body where they are more likely to detect the antigens.
B cell (B lymphocyte) Development
- The initial stages of B cell development occur within the complex microenvironments of the stromal cells of the bone marrow called niches.
- The process is activated by stimuli and factors that initiate a series of cell signals that cause the expression of different target genes that modulate cell survival, proliferation, and development.
- The development of B cell initiates with hematopoietic stem cell, which is then transformed into an early lymphoid progenitor and then a common lymphoid progenitor.
- The prerequisite for the development of B cells is the absence or suppression of protein Notch-1 signaling in the bone marrow.
- The overall development of B cells occurs following the stages of maturation, activation, differentiation, and memory generation.
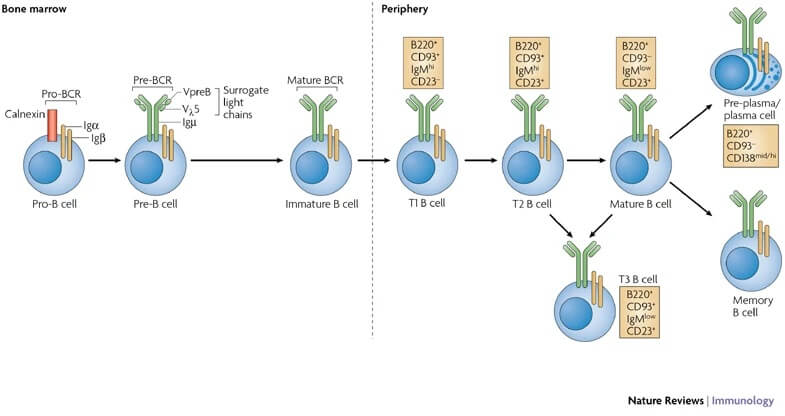
1. Maturation of B cell
- Maturation is the first step of B cell development within the bone marrow before traveling to other lymphoid organs like the spleen and lymph nodes.
- The development of immature B cells in the bone marrow can be described into different stages, each of which is characterized by various gene expression patterns and immunoglobulin H chain and L chain gene arrangements.
- During the development, B cells generate various B cell receptors as a part of the selection process.
- The selection occurs via one of two mechanisms; positive mechanisms occur via antigen-independent signaling where if the receptors on the B cells do not bind to their ligands, the cells do not receive proper signals and cease to develop.
- A negative selection mechanism occurs by the binding of self-antigen to the BCR, where if the BCR can bind strongly to a self-antigen, the development of B cells is ceased.
- In order to complete development, immature B cells migrate from the bone marrow into the spleen as transitional B cells through the two stages; T1 and T2.
- The cells are considered T1 B cells through their migration to the spleen and after entry into the spleen. In the spleen, the T1 B cells mature into T2 B cells.
- The T2 B cells differentiate either into follicular B cells or marginal zone B cells depending on the signals received by the receptors on the cells.
- The cells are considered mature B cells or naïve B cells after differentiation in the spleen.
2. Activation of B cell
- B cell activation usually occurs in the spleen or other secondary lymphoid organs like lymph nodes.
- After maturation in the bone marrow, the cells migrate to lymphoid organs as they tend to have a constant supply of antigen with the help of the circulating lymph. The migration is induced by chemokine interaction between CXCL13 and CXCR5.
- The activation of B cells begins with exposure to antigen via different receptors present on the surface like the BCR receptors.
- The response of B cells upon detection of antigen depends on the structure of the antigens.
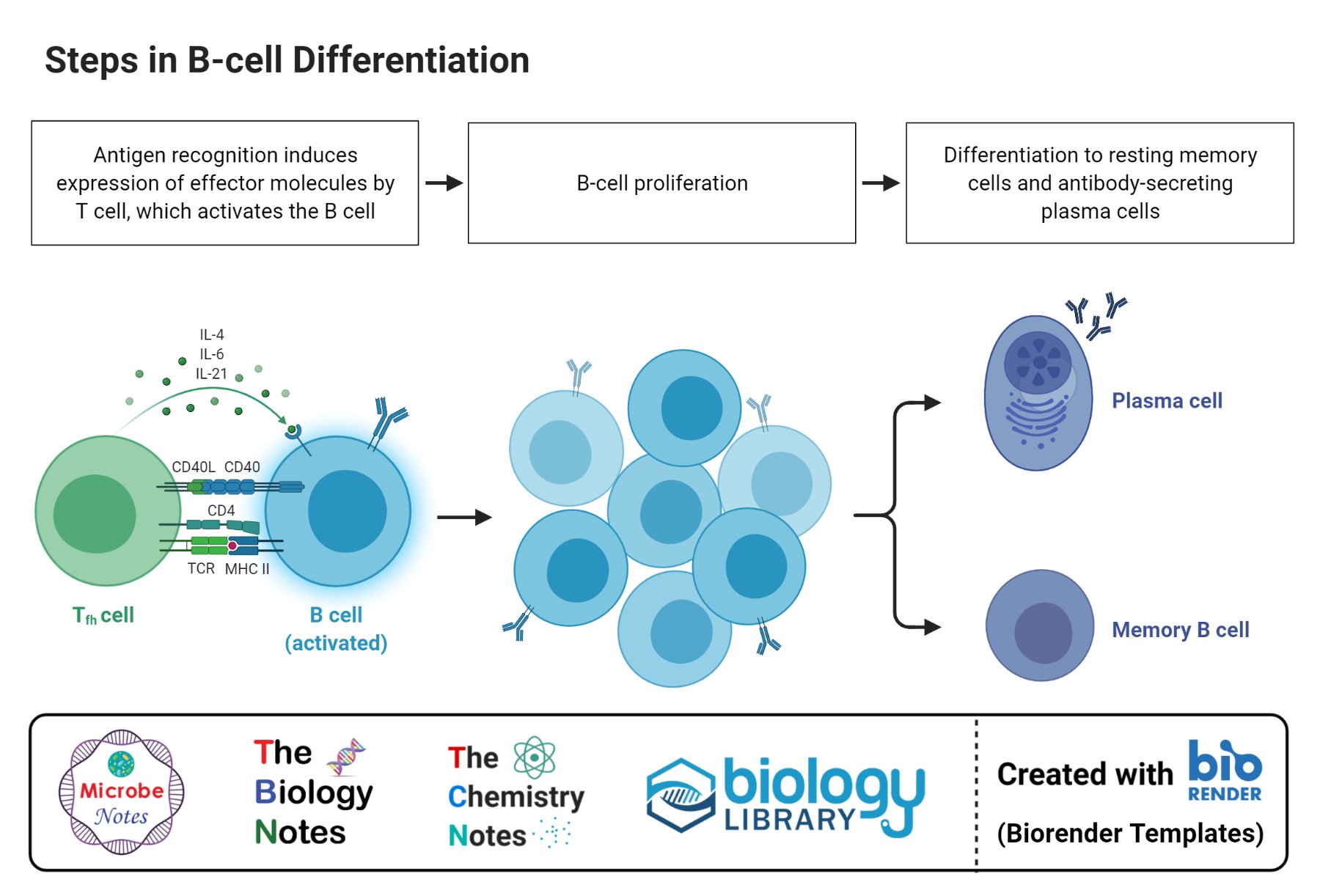
a. T cell-dependent B cell response
- At the beginning of the T-dependent B cell response, the B cells bind to the antigen via the Ig receptors. Some of the antigens are internalized into specialized vesicles within the B cells.
- The internalized antigens are processed and re-expressed in the form of peptides presented in the antigen-binding groove of class II MHC molecules.
- The T cells that have been previously exposed to antigen-bearing dendritic cells can now bind to the MHC-presented peptide on the surface of the B cell.
- The binding is further enhanced by the interaction of accessory molecules on the T- and B-cell surfaces.
- Some of the T-cell-activated B cells now move into specialized regions of the lymph node or spleen to begin the process of differentiation.
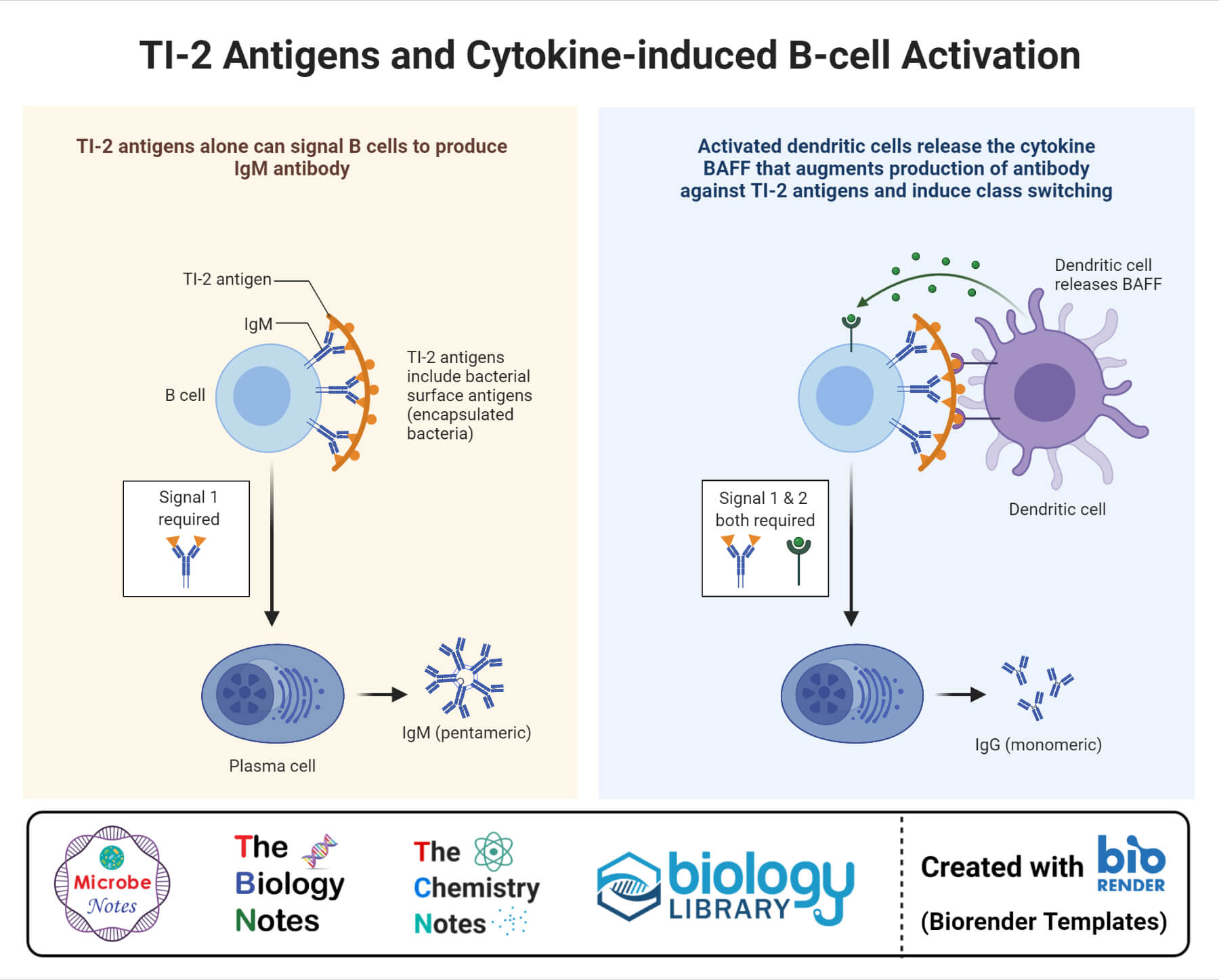
b. T cell-independent B cell response
- Activation of B cells can also occur without the participation of T cells, and it produces a particular subset of B cells that respond with antibody production to particular classes of antigens.
- Antigens that elicit a T cell-independent antibody response tend to be polyvalent with repeating determinants shared among many microbial species. These antigens are called Tl antigens.
- The responses to these antigens are usually rapid even though antibodies generated by this method have lower affinity and are less functionally versatile than those activated by T cell-dependent methods.
- Like in the case of T cell-dependent activation, B cells activated by Tl antigens also require additional signals to attain complete activation.
- However, these cells receive the signals either by recognition and binding a microbial constituent to toll-like receptors or by extensive cross-linking of the B cell receptor epitopes to the bacterial or viral surfaces.
- The B cells activated by the T cell-independent method proliferate outside the lymphoid centers and undergo immunoglobulin class switching and differentiation.
3. Differentiation of B cell
- The differentiation of activated B cells is stimulated by the interaction of the B cell receptors to specific antigens.
- Some activated cells are moved into regions at the border of the T cell and B cell areas known as primary foci.
- At the primary foci, the cells undergo differentiation into plasma cells in about four days post-stimulation.
- The differentiation cells then migrate to the medullary cord regions of the node, where they secrete large quantities of antibodies.
- Post differentiation, some of the plasma cells die after the primary response, whereas others remain in the bone marrow or the gut as long-lived plasma cells.
- Some antigen-stimulated B cells, however, do not enter the primary foci but rather migrate to follicles on the lymph nodes or the spleen.
- As the B cells begin to differentiate, the follicles swell with antigen-specific lymphocytes, resulting in a germinal center’s appearance.
- At the end of the immune response, memory B cells remain that are the daughter cells of the cells stimulated during the response.
B cell (B lymphocyte) Applications
The applications or functions of B cells can be explained in threefold;
1. Antigen Presentation
- Even though the primary function of B cells is an antibody-mediated immune response, these cells also function as professional antigen-presenting cells.
- B cells have antigen-MHC complexes as well as T cell receptors involved in T-cell activation.
- B cell lymphocytes have also been associated with the inactivation of T cells in the case of the non-specific immune system or innate immune system.
2. Cytokine Secretion
- B lymphocytes are also known to produce cytokines which are essential for cell-cell communication, especially during an immune response.
- The cytokine production invites white blood cells to induce phagocytosis on antigens attached to the B cell antibodies.
3. Antibody production
- Antibody production is the most important function of B cells as these are involved in the antibody-mediated humoral immune response.
- The antibodies fight against multiple antigens of different origins to protect the body against possible harm.
4. B cell-based immunotherapy
- Besides being the producers of antibodies, B cells also contribute to immune regulation via cytokine production and antigen presentation.
- The use of B cells as APCs has increased over the years as these can be consistently generated from peripheral blood and are relatively insensitive towards tumor-derived immunosuppressive mechanisms.
- These also do not induce tolerance by themselves and are well tolerated in terms of toxic side effects.
References
- Peter J. Delves, Seamus J. Martin, Dennis R. Burton, and Ivan M. Roitt(2017). Roitt’s Essential Immunology, Thirteenth Edition. John Wiley & Sons, Ltd.
- Judith A. Owen, Jenni Punt, Sharon A. Stranford (2013). Kuby Immunology. Seventh Edition. W. H. Freeman and Company
- Cano RLE, Lopera HDE. Introduction to T and B lymphocytes. In: Anaya JM, Shoenfeld Y, Rojas-Villarraga A, et al., editors. Autoimmunity: From Bench to Bedside [Internet]. Bogota (Colombia): El Rosario University Press; 2013 Jul 18. Chapter 5. Available from: https://www.ncbi.nlm.nih.gov/books/NBK459471.
- Su, Kuei-Ying et al. “Efficient Culture of Human Naive and Memory B Cells for Use as APCs.” Journal of immunology (Baltimore, Md. : 1950) vol. 197,10 (2016): 4163-4176. doi:10.4049/jimmunol.1502193
- Wennhold K, Shimabukuro-Vornhagen A, von Bergwelt-Baildon M: B Cell-Based Cancer Immunotherapy. Transfus Med Hemother 2019;46:36-46. doi: 10.1159/000496166.

Thank you. Great website and information.
it is good information