Photorespiration is a light-dependent metabolic pathway in plants where the oxygenation of ribulose-1, 5 5-bisphosphate (RuBP) to phosphoglycolate, a two-carbon metabolite, is catalysed by the enzyme RuBisCO, and this compound cannot directly enter the Calvin cycle.
Consequently, this forces the plant to undergo a multi-organelle salvage pathway, which retrieves a portion of carbon at the expense of energy and release of earlier fixed CO2.

Photorespiration is therefore a competitive pathway to photosynthesis, which implies that in most aspects it is a wasteful mechanism that reduces net carbon gain, photosynthetic efficiency, and consumes ATP and reducing equivalents. This occurs primarily in C-3 plants and is particularly prominent when the environmental conditions are favourable to the oxygenation activity of RuBisCO and not the carboxylation activity of the same.
Photorespiration takes place in the stroma of the chloroplasts.
The Role of RuBisCO in Photorespiration
RuBisCO (Ribulose-1,5-bisphosphate carboxylase/oxygenase) is the centrally located enzyme that is involved in the fixation of CO2 in the Calvin cycle. The RuBisCO, however, comes with a major drawback since it is dual-specific to CO2 and O2.
Reaction of RuBisCO with CO2 fixes carbon and forms two molecules of 3-phosphoglycerate (3-PGA), which is then provided to the Calvin cycle to form sugars. It is the necessary and productive response known as carboxylation.
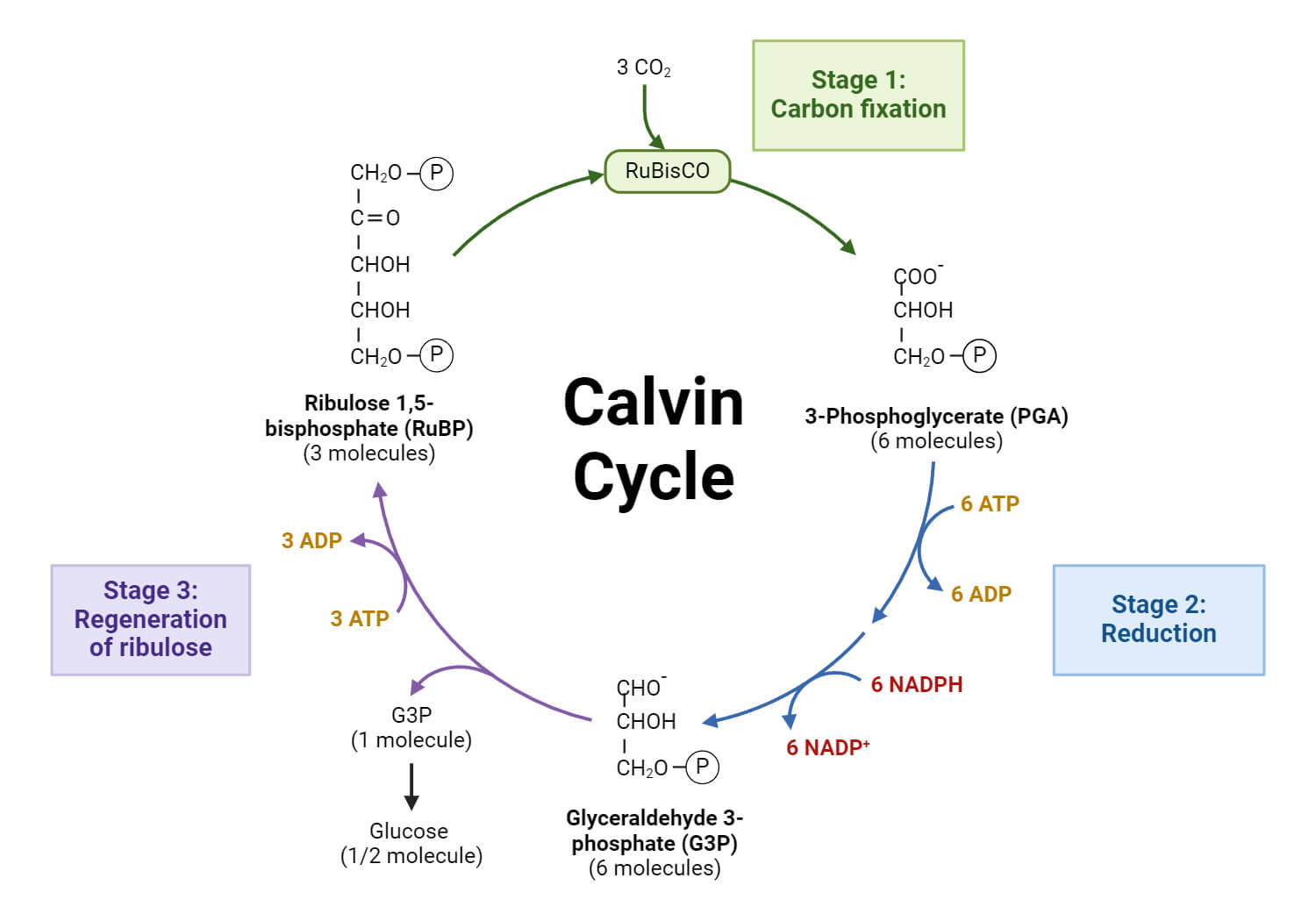
Conversely, the reaction of RuBisCO with O2 causes the oxygenation of RuBP, resulting in one molecule of 3-PGA and one molecule of 2-phosphoglycolate (PG). Phosphoglycolate is not metabolised by the Calvin cycle and is toxic when it is accumulated. To recycle the PG, the plant needs to consume energy, emitting CO2 and NH3. This is the biochemical basis of photorespiration. Temperature, salinity, and the ratio of CO2-O2 affect the affinity of RuBisCO towards CO2-O2 relative to O2 (specificity factor), and thus, photorespiration is inevitable in the natural environment.
Factors affecting Photorespiration
Photorespiration happens when the oxygenase activity of RuBisCO is preferred over the carboxylase activity of RuBisCO. This balance is strongly affected by several environmental conditions:
High Light Intensity
When there is high light, the photosynthesis rate accelerates and quickly uses CO2 within the leaf. As the concentration of CO2 decreases and the concentration of O2 increases as a result of the process of photo-lysis, RuBisCO is faced by more O2 than CO2, resulting in oxygenation.
High Temperature
The strong issue is that temperature has a very strong influence on the solubility of the gases. As the temperature increases, CO2 is less soluble than O2. The affinity of RuBisCO towards CO2 reduces, yet the affinity towards O2 does not change. Stomata will partially close to minimise water loss and lower internal CO2 even more. So on hot summer days, photorespiration is commonly at its maximum.
Low CO₂ Concentration
The low CO2, high altitude, and drought conditions or stomatal closure reduce the level of CO2 in the interstitial leaves. A low CO2/O2 ratio is highly inclined towards oxygenation.
Drought and Water Stress
In the case of water shortage, the stomata are shut to avoid the loss of water through the process of transpiration. It decreases internal CO2 and permits O2 that is generated during light reactions to build up. This makes the pathway of photorespiration dominant.
Photorespiratory Pathway: Mitochondria, Peroxisomes, and Chloroplasts
Photorespiration is among the most complicated processes in plants due to the fact that it involves three organelles, which include the chloroplast, the peroxisome and the mitochondrion. This is a multi-step salvage cycle, which tries to salvage carbon and reform 3-PGA in order to proceed with the Calvin cycle.
In the Chloroplast
It starts with RuBisCO oxygenation of RuBP, to yield: 3-phosphoglycerate (useful), 1,5-bisphosphoglycolate (toxic wastes). Phosphoglycolate is changed to glycolate. Glycolate is then transported out of the chloroplast to the peroxisome.
In the Peroxisome
Glycolate oxidase oxidises glycolate to glyoxylate at the expense of hydrogen peroxide (H2O2 ), a reactive oxygen species that has to be countered by catalase. The glyoxylate is then transformed into glycine, and it is transported to the mitochondrion.
In the Mitochondrion
Glycine decarboxylase complex (GDC) decarboxylates two molecules of glycine to yield Serine, CO₂ is lost to the atmosphere, NH₃ requires reassimilation, and NADH. The serine is reintroduced into the peroxisome, where it is again converted to hydroxypyruvate and finally to glycerate.
Return to the Chloroplast
Glycerate is taken back to the chloroplast, and it is phosphorylated to 3-PGA. This cycle is the salvage cycle, and the recovered carbon enters the Calvin cycle. It is a wasteful pathway that releases previously fixed carbon, although it is also an important part of plant metabolism.
Energy cost of Photorespiration
Carbon Loss
One molecule of CO2 is emitted for every two molecules of glycine that undergo the processes in the mitochondria. In this manner, carbon that is already fixed is lost, and it decreases net photosynthesis.
Energy Consumption
This pathway utilises ATP to lyse glycerate phosphorylate, reducing steps by using NADH and NADPH, and Extra ATP to reabsorb lost ammonia in the process of glycine decarboxylation. The decrease in Photosynthetic Efficiency.
Under ideal conditions, CO2 is fixed effectively by RuBisCO. However, when conditions conducive to photorespiration occur, photosynthetic efficiency can decrease as much as 25-50 per cent in C3 plants.
Ammonia Re-assimilation
Photorespiration emits ammonia, which is poisonous. The GS-GOGAT cycle of NH 3 re-assimilation needs additional ATP. Therefore, the cost of energy that is used in photorespiration has a major influence on the productivity of plants, particularly those grown in agriculture.
Effects on C₃, C₄ and CAM Plants
C₃ Plants
Plants with C3 (e.g. rice, wheat, potato, soybean) use only RuBisCO in CO2 fixation. Therefore, they get the greatest rates of photorespiration. They are less productive in high temperatures, light, or low CO2. In extreme conditions, loss of up to 40% of fixed carbon may occur.
C₄ Plants
C4 plants (e.g., maize, sugarcane, sorghum) reduce photorespiration by using a CO2-concentrating mechanism. They spatially isolate the initial fixation of CO2 and Calvin cycle- First fixation of CO2 occurs without oxygenase activity by PEP carboxylase. RuBisCO is surrounded by CO2 in bundle sheath cells. Due to this, there is a near absence of photorespiration in C₄ plants, and their productivity becomes significantly less sensitive to heat and light stress.
CAM Plants
CAM plants (e.g., cactus, pineapple, agave) open their stomata at night to concentrate CO2, which is deposited as malic acid. CO2 is also released internally, and during the day, there is a high CO2 concentration around RuBisCO. This time lag effectively inhibits photorespiration during the day and saves water.
Therefore, C4 and CAM plants have devised ways in which to surmount the drawbacks of RuBisCO, but C3 plants are very susceptible to photorespiration.
Physical Processes and Potential Applications of Photorespiration
Historically, photorespiration has been considered a wasteful process only; however, recent studies indicate that it does provide several valuable functions-
Protection vs. Photoinhibition
When the light is abundant, the photosynthetic electron transfer chain can be overloaded. Photorespiration is an electron sink, which dissipates the excess ATP and NADPH to avoid PSI and PSII damage.
Maintenance of Metabolic Balance
Photorespiration helps in avoiding excessive reduction of the chloroplast stroma, which keeps the redox equilibrium.
Metabolite Detoxification
Photorespiration eliminates phosphoglycolate, which is an inhibitor of major Calvin cycle enzymes.
Role in Nitrogen Metabolism
The conversion of glycine to serine also emits NH 3, which connects photorespiration to nitrogen assimilation pathways.
Defence and Stress Tolerance
Photorespiration is induced in case of drought, salt stress, high temperature, and oxidative stress. It helps plants to survive in harsh environmental conditions.
Evolutionary aspects
Photorespiration is said to have been an ancient process that occurred when the O2 in the atmosphere was low and the CO2 was high. With the increase in oxygen, the oxygenase activity of RuBisCO was problematic, yet with photorespiration, a salvage mechanism was available to sustain plant life.
Some of the strategies to reduce photorespiration in crop plants
The aim of contemporary breeding and biotechnology of plants is to minimise the wasteful nature of photorespiration.
Engineering RuBisCO
This can be done with precision. Enhancing the turnover of catalysts, substitution of plant RuBisCO by cyanobacterial or algal counterparts can be done. However, RuBisCO is highly complicated and not an easy one to alter effectively.
Implementation of CO2
Scientists are trying to develop CO2-like properties in CO2 crops like rice and wheat. Strategies include-
Addition of PEP carboxylase
These would involve changing the anatomy of the leaves to produce a Kranz-like structure.
Photorespiratory Bypass Pathways
To avoid native photorespiration, a number of synthetic pathways have been introduced into plants, in many cases resulting in lower carbon loss, reduced energy cost, and increased biomass. These are the glycolate dehydrogenase pathway and other shortcuts created-
Stomatal Manipulation
Internal CO2 can be optimised by changing stomatal density or behaviour.
Genetic Selection
Naturally, breeding for varieties less prone to photorespiration. These plans play important roles in enhancing food security in the world, particularly in the issue of climate change.
Climate Change Effect on Photorespiration
The changes in climate are likely to have several impacts on photorespiration-
Increasing Temperature
Increased temperature enhances the oxygenase activity of RuBisCO and the CO2 dissolution and promotes photorespiration.
Elevated CO₂ Levels
An increase in atmospheric CO2 may suppress photorespiration to a degree, since an increase in CO2 will be competing with O2. But this could be neutralised by,
High Drought Frequency–
Increased drought leads to closed stomata, reduced CO2, and photorespiration.
Heat Stress– Severe heat waves cause losses in photorespiration of wheat, rice, and other crops by large margins.
Rapid Plant Metabolism
An Increase in the intensity of light and temperature increases metabolism, and thus, photorespiration is a significant constraint on global agriculture.
Therefore, the effects of warming and water scarcity will possibly amplify the process of photorespiration in the next several decades.
Photorespiration and agricultural implications of photoperiodism and productivity in plants
The implications of photorespiration on agriculture are massive because-
- The production of food worldwide is dominated by C3 crops.
- Photorespiration makes their productivity highly impaired.
- Carbon will be further depleted due to climate change.
Impact on Crop Yields
Research findings project that photorespiration lowers yields by:
- 20–50% in wheat
- 30–40% in rice
- 15–30% in soybean
The losses are more intensive in the case of heat stress.
Inefficiency in nitrogen utilisation
Photorespiration emits ammonia, which means that plants use more energy to reabsorb nitrogen. This makes the use of nitrogen less efficient, raising the fertiliser requirements.
Less efficiency in water use
Photorespiration with high rates is typical of the drought period, when stomata are closed, and the growth is already restricted by water stress.
Since the demand for food is increasing, the main aim of sustainable food production is to decrease photorespiration.
Photorespiration as measured and experimentally demonstrated
Photorespiration has been examined in considerable detail through several experimental methods-
Gas Exchange Measurements
Infrared gas analysers determine the uptake of CO2 and release of O2 under different conditions. The rate of photorespiration is reflected in the ratio of CO2 assimilation to the uptake of O2.
Isotope Labelling
Using isotopes such as ¹⁴C-labelled CO₂, ¹⁸O₂ assists in monitoring reactions of carbon flow and oxygenation.
Measurements of enzyme activity
Assessing RuBisCO oxygenase activity, Glycolate oxidase, GDC activity, and crook evidence of the pathway biochemically.
Mutant studies
Photorespiratory mutants, which lack some enzymes like glycolate oxidase or GDC, are toxic and accumulate, and exhibit chlorosis in the presence of normal air. They are only survivable in high CO2, which testifies that the pathway is necessary in the conditions of a normal atmosphere.
Transcriptomics and Proteomics
The fact that photorespiration is extensively controlled and stress-responsive through modern technologies also adds weight to its physiological importance.
Conclusion
Photorespiration is an obligatory, yet metabolically expensive, phenomenon due to the bivalency of RuBisCO to use both CO2 and O2. Though still considered eventually as wasteful in energy usage and carbon loss, it has essential roles in sustaining plant metabolism, in safeguarding photosystems, and in enabling stress resistance. C3 plants are also highly susceptible to photorespiration, but C4 and CAM plants have adapted to avoid it. Photorespiration has turned out to be an even more important limitation to crop productivity in the context of climate change, with an increase in temperature, droughts, and varying CO2 levels. Contemporary plant science intends to mitigate the adverse effects of photorespiration using genetic engineering, modification of RuBisCO, and the CO2 -concentrating systems. Knowledge in this intricate pathway would be critical to enhance agricultural production and food security in the world.
References
- Peterhansel, C., Horst, I., Niessen, M., Blume, C., Kebeish, R., Kürkcüoglu, S., & Kreuzaler, F. (2010). Photorespiration. The Arabidopsis Book, 8, e0130. https://doi.org/10.1199/tab.0130
- Smith, E. N., Van Aalst, M., Tosens, T., Niinemets, Ü., Stich, B., Morosinotto, T., Alboresi, A., Erb, T. J., Gómez-Coronado, P. A., Tolleter, D., Finazzi, G., Curien, G., Heinemann, M., Ebenhöh, O., Hibberd, J. M., Schlüter, U., Sun, T., & Weber, A. P. (2023). Improving photosynthetic efficiency toward food security: Strategies, advances, and perspectives. Molecular Plant, 16(10), 1547–1563. https://doi.org/10.1016/j.molp.2023.08.017
- Chikov, V. I., & Akhtyamova, G. A. (2019). Photorespiration and its role in the regulation of photosynthesis and plant productivity. American Journal of Plant Sciences, 10(12), 2179–2202. https://doi.org/10.4236/ajps.2019.1012154
- Betti, M., Bauwe, H., Busch, F. A., Fernie, A. R., Keech, O., Levey, M., Ort, D. R., Parry, M. a. J., Sage, R., Timm, S., Walker, B., & Weber, A. P. M. (2016). Manipulating photorespiration to increase plant productivity: recent advances and perspectives for crop improvement. Journal of Experimental Botany, 67(10), 2977–2988. https://doi.org/10.1093/jxb/erw076
- Dusenge, M. E., Duarte, A. G., & Way, D. A. (2018). Plant carbon metabolism and climate change: elevated CO2 and temperature impacts on photosynthesis, photorespiration and respiration. New Phytologist, 221(1), 32–49. https://doi.org/10.1111/nph.15283
- Aroca, A., García-Díaz, I., García-Calderón, M., Gotor, C., Márquez, A. J., & Betti, M. (2023). Photorespiration: regulation and new insights on the potential role of persulfidation. Journal of Experimental Botany, 74(19), 6023–6039. https://doi.org/10.1093/jxb/erad291
- 2.2.3 – Energetics of C4 photosynthesis | Plants in Action. (n.d.). https://rseco.org/content/223-energetics-c4-photosynthesis.html
- Libretexts. (2025, July 28). 4.1.7: Photorespiration and Photosynthetic Pathways. Biology LibreTexts. https://bio.libretexts.org/Bookshelves/Botany/Botany_(Ha_Morrow_and_Algiers)/04%3A_Plant_Physiology_and_Regulation/4.01%3A_Photosynthesis_and_Respiration/4.1.07%3A_Photorespiration_and_Photosynthetic_Pathways
- Photosynthesis in C3, C4 and CAM plants. (2025, September 12). Student Academic Success.
- https://www.monash.edu/student-academic-success/biology/photosynthesis/photosynthesis-in-c3,-c4-and-cam-plants
- Pandey, S. N., & Sinha, B. K. (2009). Plant physiology. Vikas Publishing House.
- Verma, S. K., & Verma, M. (2010). Textbook of plant physiology, biochemistry and biotechnology. S. Chand Publishing.
