Gas chromatography-mass spectrometry (GC-MS) is a method that combines the separation ability of gas chromatography (GC) with the detection ability of mass spectrometry (MS) for the qualitative and quantitative analysis of compounds in mixtures.
The combination of chromatographic peak information and mass spectral information helps identify unknown compounds and analyze complex mixtures accurately.

Fred McLafferty and Roland Gohlke of Dow Chemical Company were the first to combine GC and MS in the 1950s. GC-MS is now widely used as a standard method in many fields for detecting and quantifying different chemicals. This method is used in fields including environmental monitoring, forensics, pharmaceuticals, food safety, and even space exploration.
Principle of GC-MS
The principle of GC-MS involves combining GC and MS for accurate separation and identification of compounds in complex mixtures. At first, the GC separates the components of a sample, and then the MS identifies the separated compounds.
In GC, the sample mixture is first vaporized in the heated inlet. This vaporized sample is carried by an inert carrier gas into the GC column, which is coated with a stationary phase. Each compound in the sample interacts differently with the stationary phase based on its properties and elutes from the column at a different speed. This separates the compounds.
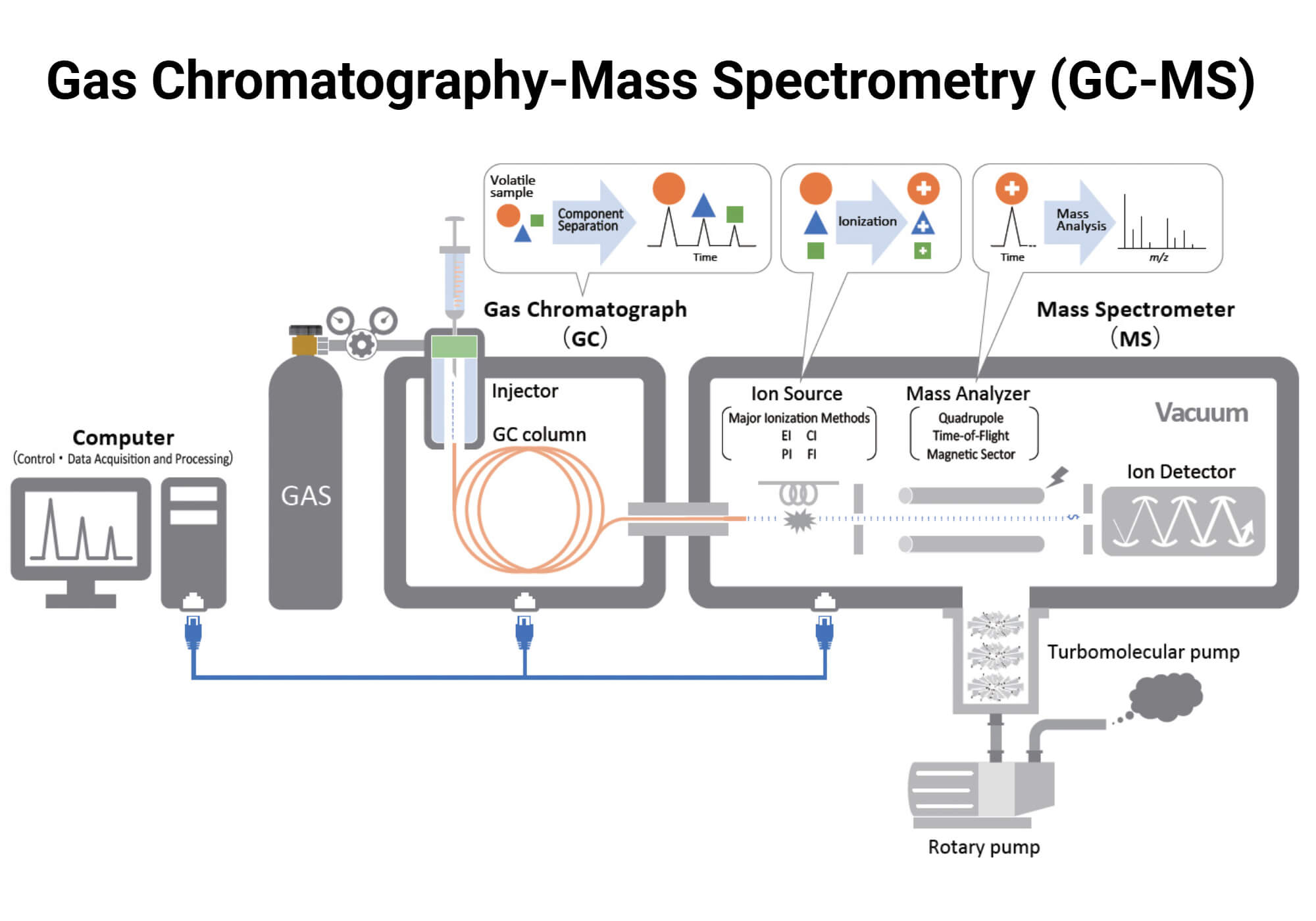
The separated compounds from the GC enter the MS through a heated transfer line that connects the two systems. In the MS, molecules are ionized using electron or chemical ionization sources. The resulting ions are separated by mass analyzers according to their mass-to-charge ratio (m/z). These separated ions are detected by ion detectors, which produce a unique mass spectrum that can be compared with known spectral libraries for accurate identification and quantification. The combined ion signals are plotted as a total ion chromatogram (TIC), where each peak corresponds to a specific compound.
Components of GC-MS
- GC inlet: It is the injection port for the introduction of the sample into the GC system. It is also used for vaporization of samples and to transport these vaporized analytes into the column using carrier gas. Different types of inlets can be used in GC-MS. Common inlet types include split/splitless, direct or on-column, and headspace inlets.
- Carrier gas: It is the mobile phase that moves the sample through the column. The carrier gas is stored in high-pressure cylinders, and its flow is regulated by valves, flow meters, and gauges. Commonly used carrier gases include helium, nitrogen, hydrogen, and argon.
- GC column: It is the tube where separation occurs. The column is placed within an oven that maintains the column temperature. GC-MS uses packed columns or capillary columns. Capillary columns are more widely used as they provide higher resolution.
- Stationary phase: It is the fixed material inside the column that interacts with the sample. It can be solid particles or a liquid coating on an inert support inside the column.
- Interface: This is the heated transfer line that connects the GC and MS. It maintains the analytes in the vapor phase to ensure that the analytes eluting from the GC column enter the MS ion source without degradation.
- Vacuum system: The vacuum system in MS is necessary for the collision-free flight of ions from source to detector. Different pumping setups can be used like single pump system and a differential pumping system.
- Ion source: It ionizes the sample molecules and directs the resulting ions into the mass analyzer. Two ionization methods used are electron ionization (EI) and chemical ionization (CI). EI forms ions through electron bombardment, while CI generates ions through reaction with a reagent gas. EI is more commonly used.
- Mass analyzer: It separates the ions based on their mass-to-charge (m/z) ratio. Commonly used mass analyzers are quadrupole, ion trap/orbitrap, time-of-flight, and magnetic sector analyzers. These are single mass analyzers. Some systems use multiple mass analyzers, like a triple quadrupole MS/MS system. Mass analyzers can also be grouped into continuous and pulsed analyzers based on how ions are introduced into the system. Continuous analyzers allow a constant flow of ions, while pulsed analyzers introduce ions in pulses or batches.
- Detector: The detector records the ions exiting from the mass analyzer and converts them into electrical signals for analysis. These signals are processed into a mass spectrum and combined with chromatographic retention time data for compound identification and quantification. Ion detectors include electron multipliers and multi-channel plates.
- Data system: It collects, processes, and displays the chromatogram and mass spectra. It allows analysis by comparing spectra with reference libraries.
Types of GC-MS
Based on the type of mass spectrometer used, some of the main types of GC-MS are:
Single Quadrupole GC-MS
- It uses quadrupole mass analyzers that have four parallel rods inside a vacuum chamber.
- They act as mass filters by allowing only target ions of certain m/z values through the quadrupole to reach the detector, while unstable non-target ions are filtered out.
- The ions that reach the detector are converted into electrical signals, and the mass spectrum data are captured.
- It can be operated in two modes:
Full scan mode scans across a range of m/z values and provides full spectral data. This is especially useful for unknown compounds.
Selected Ion Monitoring (SIM) focuses on only specific ions and is useful for targeted quantitative analysis.
Triple Quadrupole GC-MS/MS
- It combines GC with a tandem mass spectrometry (MS/MS) that contains three quadrupoles arranged in series (Q1, Q2, and Q3).
- The first quadruple isolates the target ion, the second acts as a collision cell to fragment the selected ions, and the third analyzes the resulting ions.
- It can operate in different modes like product ion scan, precursor ion scan, neutral ion scan, and selected reaction monitoring (SRM).
Time-of-Flight (TOF) GC-MS
- In this method, ions are accelerated by high voltage into a high-vacuum chamber called a flight tube.
- The ions are separated based on how quickly they travel through the flight tube to reach the ion detector. Lighter ions travel faster than heavier ones. The time of flight is measured accurately and converted into m/z values.
Magnetic sector GC-MS
- It uses a strong magnetic field to separate ions based on their m/z values.
- Here, ions are accelerated into the mass analyzer and deflected according to their m/z. Lighter ions are deflected more strongly. A complete mass spectrum is generated by scanning the magnetic field across the m/z range.
Ion Trap GC-MS
- Ion traps use electric fields to trap ions temporarily in orbits before sequentially releasing them to the detector.
- It provides high sensitivity as all trapped ions can be detected during scanning; however, only a limited number of ions can be trapped at once.
Sample Preparation for GC-MS
GC-MS is mainly used for small and volatile molecules like environmental pollutants, food contaminants, and drug metabolites, which are usually complex and contain unwanted components that could interfere with the analysis. So, samples need to be cleaned or processed before analysis. Proper sample preparation is necessary for accurate analysis of sample components, as it ensures that the samples are in a suitable form for separation and ionization. Different sample preparation techniques can be used depending on the type of sample. Sample preparation steps may include extraction, filtration, dilution, concentration, or derivatization.
Procedure or Steps of GC-MS
Sample introduction and vaporization
The sample is introduced and vaporized in the heated GC inlet. Samples can be injected using a syringe or autosampler. The vaporized sample is carried into the GC column by an inert carrier gas.
Separation (GC)
Inside the GC column, sample components interact with the stationary phase according to their properties, such as volatility and polarity. Compounds that have weaker interactions with the stationary phase move faster through the column and elute first, while those with stronger interactions have longer retention times. This difference in movement or retention time separates the sample components.
Transfer to MS
The separated compounds elute from the GC column and are passed into the ion source of MS through a heated transfer line. This interface is maintained at a high temperature to prevent the degradation of the sample.
Ionization
The MS ion source ionizes and fragments the molecules using electron ionization or chemical ionization. The ionized molecules and fragments carry specific m/z ratios.
- Electron ionization (EI) is a hard ionization method where high-energy electrons are generated by a heated filament and accelerated into the ion source. These electrons collide with the sample molecules eluting from the GC and form molecular ions. The excess energy from the collision causes fragmentation of these ions.
- Chemical ionization (CI) is a soft ionization method that produces molecular ions with less fragmentation. It uses a reagent gas to transfer charge instead of directly ionizing the sample with electrons. There are two modes of CI: positive (PCI) and negative (NCI). In PCI, electrons ionize the reagent gas molecules to form positive reagent ions. Sample molecules collide with these ions and produce charged analyte ions. In negative CI, electrons collide with reagent gases and produce low-energy electrons that attach to sample molecules, forming negative ions.
Mass Analysis
The resulting ions are separated based on their mass-to-charge ratio (m/z) by using a mass analyzer. Quadrupole, ion trap, TOF, and magnetic sector are commonly used analyzers. Single quadrupole mass analyzers are the most widely used due to their compact design, easy operation, and low cost.
Detection
The separated ions reach the ion detector, which measures the intensity of each ion and converts them into electrical signals. These signals are processed to generate a mass spectrum showing ion intensity at different m/z values. The spectrum can be compared with spectral libraries to obtain detailed molecular information.
Identification and Quantification
All detected ion signals can be combined to form a total ion chromatogram (TIC), which plots signal intensity against retention time. Each peak in the TIC represents a separate compound, and the peak area can be used to quantify them.
Factors Affecting GC-MS
- The type of carrier gas used can affect the resolution of separation. The flow rate and pressure of the gas are also important factors.
- Column characteristics like length, internal diameter, temperature, and stationary phase are also factors affecting GC-MS.
- Proper sample preparation is necessary to avoid contamination and column damage.
- Sample characteristics like volatility, thermal stability, and concentration can also affect the process.
- The method of ionization used also affects GC-MS.
- Mass analyzer and detector settings can determine the accuracy of the mass spectra.
Common Products and Manufacturers of GC-MS
| Common Products | Manufacturers |
| GCMS-TQ8050 NX, GCMS-QP2020 NX, SH Series GC Columns, AOC-6000 Plus series Autosampler, OPTIC-4 Multimode Inlet, TD-30 Series Thermal Desorption Systems, Twin Line MS System, GCMSsolution, LabSolutions GCMS, Smart Aroma Database | Shimadzu |
| ISQ 7610 Single Quadrupole GC-MS, TSQ 9610 Triple Quadrupole GC-MS/MS, Orbitrap Exploris GC 240 MS, DFS Magnetic Sector GC-HRMS, TriPlus RSH SMART Autosampler | ThermoFisher |
| 5977C GC/MSD, 7000E triple quadrupole GC/MS, 7010D Triple Quadrupole GC/MS, 7250 GC/Q-TOF, GC/MS Ion Sources, Ultra Low-Bleed (Q) GC/MS Columns | Agilent |
| Waters Atmospheric Pressure Gas Chromatography (APGC) Source, Xevo TQ-GC Mass Spectrometry System | Waters |

Applications of GC-MS
- GC-MS is used in environmental monitoring to track pollutants like pesticides and volatile organic compounds.
- It is used in forensic labs for crime scene investigations. Biological samples from suspects or victims can be screened to detect illicit drugs like cocaine, heroin, methamphetamine, and poisons using GC-MS.
- It is also used in anti-doping laboratories to detect prohibited performance-enhancing substances like anabolic steroids in athletes.
- It has been used in security checkpoints at airports or sensitive locations to detect explosives or other chemical warfare agents.
- It has also been used in space research, including Viking landers in the 1970s to search for organic molecules in Martian soil, Pioneer Venus to analyze the atmosphere of Venus, and the Rosetta mission to analyze the comet’s material.
- It has applications in diagnostics and metabolic research. It has been used in newborn screening for early diagnosis and treatment of several congenital metabolic diseases.
Advantages of GC-MS
- GC-MS is used for both qualitative and quantitative analysis of compounds in complex mixtures.
- It provides accurate information on compounds using mass spectral data.
- It is effective for detecting trace-level compounds that are difficult to analyze with other techniques.
- Most GC-MS systems allow rapid separation and detection, which saves time in high-throughput applications.
- It is widely used in different fields, including food safety, forensic science, pharmaceuticals, and environmental monitoring.
Limitations of GC-MS
- It is expensive to operate and maintain GC-MS systems for some applications.
- Sample preparation can be complex and time-consuming.
- System leaks and contamination can occur frequently, which increases background noise and reduces sensitivity.
- It is limited to volatile organic compounds and thermally stable analytes. Other substances cannot be directly analyzed and require derivatization.
- Operation and data interpretation of GC-MS can be complex and may require skilled personnel.
Troubleshooting and Safety Considerations for GC-MS
- Poor peak shape can result from column overloading, leaks, contamination, or an unsuitable stationary phase. All fittings should be checked, and leak detectors or filters can be installed to prevent leaks. The stationary phase must also be suitable, and sample overloading should be avoided.
- An unstable baseline or high background noise occurs due to contamination or column bleed. Using high-purity gases and regularly cleaning or replacing filters and inlets can help minimize background contamination.
- Injection or system issues like blocked or empty syringes, broken columns, and an inappropriate detector can result in no peaks. This can be resolved by cleaning or replacing the syringe, checking the sample in the vial, replacing or reinstalling the column, and ensuring the detector is suitable.
- Split peaks are caused by incompatible solvents or a stationary phase, incomplete vaporization, or sample overloading. This can be solved by using a compatible solvent or stationary phase, using proper inlet temperature, or reducing injection volume.
- Carrier gases should be properly secured in cylinders to avoid gas leaks or explosions. Special leak detection and ventilation systems should be used for flammable carrier gases like hydrogen.
- Direct contact with hot surfaces like the injector, oven, or transfer line must be avoided.
- Toxic or reactive samples should be handled properly using personal protective equipment (PPE), fume hoods, and proper waste disposal protocols.
Recent Advances and Innovations
- Improved ion source technologies and detectors have been introduced that provide high sensitivity to detect compounds at trace levels. High-resolution mass spectrometry (HRMS) has improved mass accuracy and allowed more accurate compound identification.
- Advanced mass spectrometers like triple quadrupole systems and ultra-fast mass spectrometry systems provide greater sensitivity.
- Smaller and more compact instruments like micro-GC systems have been developed, which allow on-site analysis.
- The use of smart and automated systems in GC-MS instruments is increasing, which reduces manual effort and time.
- Modern GC-MS systems also include sustainability to use less resources, including energy, water, and carrier gas.
- Multidimensional techniques and a combination of GC-MS with other analytical methods have been used, which improves separation and identification.
Conclusion
GC-MS is a widely used analytical method for the accurate separation and identification of volatile and semi-volatile compounds. It provides advantages like high sensitivity and accurate qualitative and quantitative detection, but it also has limitations like high cost, complex sample preparation, and suitability for volatile compounds. Recent innovations and advances are helping to overcome these challenges.
References
- Botcherby, L., & Matheson, A. (2022, April 15). Trends and Developments in GC and GC–MS. Chromatography Online. Retrieved from https://www.chromatographyonline.com
- Fundamentals of GCMS: Understand the basics, key trends, and latest developments. (n.d.). Retrieved from https://www.shimadzu.com.sg/an/resources/resource-library/gas-chromatography-mass-spectrometry-gc-gcms/gas-chromatography-mass-spectrometry-gcms-fundamental-guide.html
- Gas Chromatograph-Mass Spectrometry (GC-MS). (2024, January 1). Retrieved from https://www.shimadzu.com/an/products/gas-chromatograph-mass-spectrometry/index.html
- Gas chromatography mass spectrometry basic principles | Agilent. (n.d.). Retrieved from https://www.agilent.com/en/product/gas-chromatography-mass-spectrometry-gc-ms/gcms-fundamentals
- Gas Chromatography Mass Spectrometry: Principle, instrumentation, Advantages, and 10 Reliable Applications – Chemistry Notes. (2023, April 19). Retrieved from https://chemistnotes.com/analytical_chemistry/gas-chromatography-mass-spectrometry-principle-instrumentation-advantages-and-10-reliable-applications/
- History of the combination of gas chromatography and mass spectrometry – American Chemical Society. (n.d.). Retrieved from https://www.acs.org/education/whatischemistry/landmarks/gas-chromatography-mass-spectrometry.html
- Mathias, J. (2024, June 7). How stuff works: GC/MS analysis. Retrieved from https://www.innovatechlabs.com/newsroom/642/stuff-works-gcms-analysis/
- Qualitative Methods of GC/MS Analysis:Retention time and Retention index. (n.d.). Retrieved from https://www.shimadzu.com/an/service-support/technical-support/gas-chromatograph-mass-spectrometry/analysis-results/retention/retention.html#anchor4
- Technology Networks. (2024, February 16). GC-MS Principle, Instrument and Analyses and GC-MS/MS. Retrieved from https://www.technologynetworks.com/analysis/articles/gc-ms-principle-instrument-and-analyses-and-gc-msms-362513
