Antibodies are protein molecules naturally produced or synthesized by the B-lymphocytes.
They are also known as Immunoglobulins. The use of the term antibody defines an Immunoglobulin molecule that has specificity for an epitope of the molecules that make up antigens.
Produced and secreted by plasma cells, antibodies are soluble molecules that travel throughout the body to find and bind to their targets which are foreign substances known as antigens. The binding of an antibody to an antigen or a microbial epitope can inhibit or prevent microbial spread by several means: immobilization, prevention of microbial attachment to host cells, promotion of increased phagocytosis, and targeting microbes for destruction by other soluble molecules or by leukocytes such as natural killer (NK) cells and eosinophils.
Antibodies’ major function is to recognize and bind antigen molecules on their very specific antigen-binding sites.
Upon detection and recognization of an antigen, the B-lymphocytes undergo cell proliferation and differentiation to form the plasma cells which secrete large amounts of antibodies to attack the antigen.
Their specialized structure allows them to have antigen specificity, allowing only specific antigens to bind onto the antigen-binding sites.
Forms of Antibodies
These antibodies are of two classes;
- Membrane-bound antibodies that make up the B-cell receptors (BCR); also known as surface immunoglobulins (sIg) or membrane-bound immunoglobulin (mIg). The antibody-bound B-cells have receptors on its surface allowing the B cell to detect a specific antigen that is present in the body, which triggers B cell activation, which proliferates and differentiates to plasma cells that produce the circulating antibodies. The B-cell receptor (BCR) is normally composed of surface-bound IgD or IgM antibodies and associated with Ig-α and Ig-β heterodimers, that have the ability to induce signal transduction. a single B-cell has about 50,000-100,000 antibodies bound to its surface.
- Antibodies circulating freely in the blood offering the effector mechanisms of humoral immunity by searching and neutralizing antigens or marking them for elimination by immune cells like the antigen-presenting cells.
Antigen-Antibody (Ag-Ab) Interaction
- Antibodies have specific sites that recognize antigen epitopes known as antigen-binding sites. these are the regions on the antibodies where the antigen binds on the antibody, and antigens have special surface structures known as epitopes or antigen determinants which are arranged discontinuously and they vary or are different.
- During an encounter with an antibody, the antigen is bound spatially on the antigen-binding site, and their interaction is similar to that of the lock and key reaction of enzymes and substrate interactions.
- This interaction involves a number of weak forces including the hydrogen bonds, hydrophobic interactions, electrostatic forces, and the van der Waals forces. The weakness of these forces can only mean that antibody affinity to an antigen is relative and therefore, the antigen-antibody interaction can be reversible since it is not absolute. In addition to this, it means that the weak forces make it possible for the antibody to cross-react with other different antigens of different relative affinities.
- When the antigen and the antibody bind, they form an immune complex with functions as a unit, either as an antigen on its own that is attacked by other circulating antibodies or by provoking an immune response, by acting as carriers to the antigens (haptens), making them immunogenic.
- Antigen-antibody complexes have been for the diagnosis of infections caused by pathogens such as bacteria, viruses.
Antibody Structure
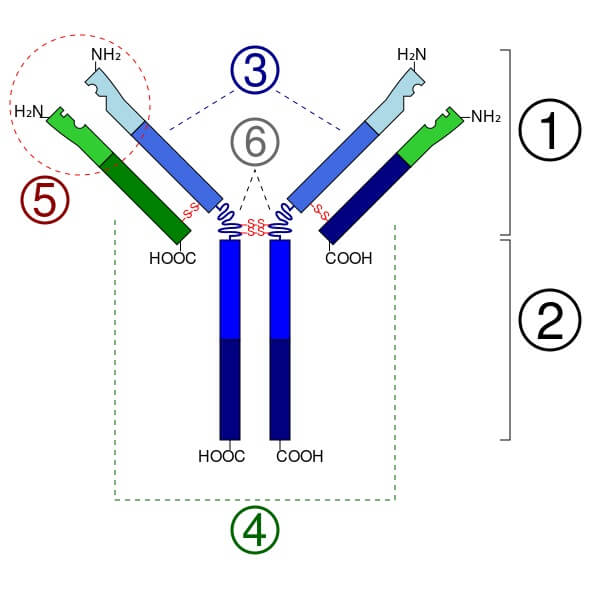
Figure: 1- Fab region. 2- Fc region. 3- Heavy chain (blue) with one variable (VH) domain followed by a constant domain (CH1), a hinge region, and two more constants (CH2 and CH3) domains. 4- Light chain (green) with one variable (VL) and one constant (CL) domain. 5- Antigen binding site (paratope). 6- Hinge regions. Image Source: Y_tambe (Wikipedia).
- Antibodies are heavy globular plasma proteins of about ~150 kDa and a size of about 10nm. They are made up of sugar chains known as glycans which are added to conserved amino acid residues, and therefore, antibodies are glycoproteins.
- The glycans play a principal role in stabilizing the antibody structure and its function. They also modulate the antibody affinity to the FcR (s).
- The basic functional unit of an antibody is known as an immunoglobulin (Ig) which is monomeric, while the secreted antibodies can be dimeric or tetrameric or pentameric or polymeric.
- The antibody is made up of variable V regions, and constant C regions.
Heterodimer structure of antibodies
- The antibody structure was first determined by Edelman who isolated it from the blood sample of Multiple Myeloma, using myeloma proteins. They identified two chains, with molecular weights of 20 kDa (light chain) and 50 kDa (heavy chain). Their relative concentrations suggested that the basic antibody unit consisted of two heavy chains and two light chains. In conclusion, antibodies are heterodimers.
- This four-chain structure is seen in IgG, IgD, and IgE. By contrast, IgA occurs in both monomeric and polymeric forms (comprising more than one basic four-chain unit structure) whilst IgM occurs as a pentamer with five basic units.
Immunoglobulin domains
- The Immunoglobulin monomer has a “Y” – shaped molecule which has 4-polypeptide chains; two identical heavy (H) chains and two identical light (L) chains which are connected by a disulfide bond.
- Each of the chains is made up of globular domains known as immunoglobulin domains formed by intra-chain disulfide bonds. The domains containing 70-110 amino acids and are classified into different types depending on size and function such as the variable domain (IgV) and the constant domains (IgC).
- The single variable region or domains (IgV) is of two forms, VL and VH consisting of 100 amino acids and the constant domains (CH and CL).
- The domains have a specific immunoglobulin fold which has two beta-sheets sheets form a sandwich shape, that is held together by the forces between the interaction of conserved cysteines and other amino acids.
The Heavy Chain of the antibodies
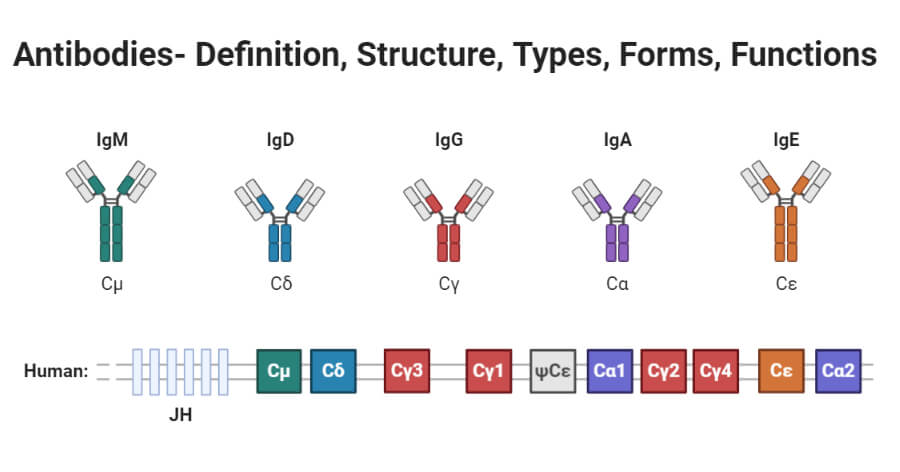
- There are five types of the mammalian immunoglobulin heavy chains that are denoted by greek letters α, δ, ε, γ, and μ
- The heavy chains dictate the class of immunoglobulin and therefore they are found on each immunoglobulin isotype, e.g. µ chains are present in IgM, γ chains in IgG, etc.
- The heavy chain is distinct in its size and composition, such that the α and γ contain approximately 450 amino acids, whereas μ and ε have approximately 550 amino acids.
- Each heavy chain has two regions, the constant (C)region, and the variable (V) region; the constant region is identical in all antibodies of the same isotype but different in the antibodies of other isotypes.
- The heavy chains γ, α, and δ have a constant region that is made up three tandem immunoglobulin domains, and a hinge region, which functions in maintaining the antibody flexibility, while the heavy chains μ and ε have a constant region that is made up of four immunoglobulin domains.
- The heavy chain variable region is different in the antibodies that are produced by different B-cells, but it is the same in the antibodies produced by a single B-cell or a B-cell clone.
- The variable region of each heavy chain is about 110 amino acids long, and it is made up of a single immunoglobulin domain.
The Light Chain of the antibodies
- Mammals have two types of light chains known as lambda (λ) and kappa (κ) with two domains; a constant domain and a variable domain.
- The light chain is 211 to 217 amino acids long.
- Each antibody has two light chains that are identical and only one of the two (lambda (λ) and kappa (κ)) light chains is present per antibody in mammals; therefore either of the chains can occur in any of the 5 types of heavy chains.
- Therefore, the heavy and light chains are antigenically distinct and only one type of the light chain can be present in any single antibody molecule.
The Cluster of differentiation, Fv, Fab, and Fc regions
- Each part of the antibody has a role it plays in antigenicity.
- The arms of the antibody shape up in a V-shape which is structurally functional for binding of specific molecules which enables the recognition of antigens is known as the Fab (fragment, antigen-binding) region. It is made up of one constant and one variable domain from each heavy and light chain of the antibody.
- The antigen-binding site also known as the paratope at the amino-terminal end on the monomeric antibody is shaped by the variable domains from the heavy and light chains known as the Fv region. It plays a pivotal role in the binding of antigens.
- The variable regions form variable loops of both the light (VL) and the heavy (VH) chains that are responsible for the binding to the antigen. These loops are known as the complementarity-determining regions (CDRs).
- Fc region which is the tail section of an antibody, made up of the constant heavy chain domains, cooperates with cell surface receptors called Fc receptors and some proteins of the complement system. This property permits antibodies to activate the immune system.
- This allows them to bind to specific antigens and mediate the antigen presentation mechanisms.
Functions of Antibodies
- Antibodies are produced when B-cells have been activated and differentiated into plasma cells that are responsible for the secretion of soluble antibodies or memory cells that can survive in the body for a long time, as long as years. The memory cells give the immune system the capability to remember an antigen it had previously encountered and produces a quick immune response on exposure.
- The antibodies found during the prenatal and neonatal phase of life are those that have been passively passed on from the mother by passive immunization. The early endogenous antibody production depends on the type of antibody, and they appear during the first year of life.
- The existence of antibodies freely circulating in the bloodstream, they form part of the humoral immune system. The freely circulating antibodies are produced by clonal B-cells that respond to a specific antigen. The antibodies functions are vast but can be generalized as follows:
- Neutralization whereby, the neutralizing antibodies block bacterial cells or virions, preventing them from attacking the host, by making them ineffective in their mechanism.
- Agglutination: This is where the antibodies play a role in the clumping of foreign cells attracting them to phagocytic cells, for phagocytosis.
- Precipitation: similar to agglutination but In this case, serum soluble antigens are clumped so at to attract phagocytic cells for phagocytosis.
- Complement activation: This is the mechanism of antibodies that are bound to a foreign cell, initiates the activation of complements to attack the foreign cells by forming a membrane attack complex. The membrane attack complex can lead to cell lysis, attracting inflammatory cells that cause inflammation.
The role of antibodies in immunity however includes:
- To prevent pathogens from entering and damaging the host cells, by binding to the antigens
- By stimulating the removal of pathogens by macrophages and other immune cells. This is enabled by the antibodies coating themselves on the pathogen, which triggers the production of these immune cells.
- By triggering the destruction of pathogens. This is enhanced by stimulating the functions of other immune responses including complement activation pathways.
- By triggering vasoactive amine degranulation which contributes to providing immunity against certain antigens such as allergens, helminths.
Overall, the functions of antibodies are three:
- Activation of complements: When antibodies bind to the antigen, the complex (Ag-ab) attracts the components of the complement cascade with the Fc region. this initiates the activation of the classical pathway of the complement system. The pathway kills the antigen (bacteria) by:
- binding of the antibody and the complement molecules, marking the antigen for ingestion by phagocytes, a mechanism known as opsonization: due to the attraction of the complement elements byte complex which consequently attract the phagocytic cells.
- elements of the complement system form a membrane attack complex that assists in the antibodies killing the bacterial antigens directly by bacteriolysis.
- Activation of the effector cells: antibodies normally have at least two paratopes on their surface. When they recognize an antigen (pathogen) outside the cell coated on the Fc region, they bind identical antigens on their paratopes’ Fc region causing the coating/clumping of more than two antigens (pathogens). This links the antigens together, by agglutination process. The agglutinin activates the effector mechanisms of the cells that recognize the Fc region of the antibodies. This includes the activation of phagocytic cells to phagocytosis the complex, mast cells, and neutrophils to degranulate the complex and the natural killer cells which induce the Antibody-dependent Cytotoxic activity (ADCC). The induction of the ADCC is a mechanism that has been applied in studying the efficacy of monoclonal antibody use in cancer therapies. The Fc receptors are isotype-specific (IgA, IgG, and IgE) and therefore it gives the antibodies great flexibility to the immune system, therefore it invokes the appropriate immune mechanism for distinct pathogens
- Natural antibodies: these are antibodies that are naturally produced by the body without prior exposure to an antigen or even vaccination or even passive immunization. They are found in human serum before exposure to a viral particle. These antibodies have been studied and they have the ability to induce a classical complement pathway that lyses enveloped viruses before the activation of the adaptive immune responses. studies have linked xenotransplant rejection to the mechanism of the natural antibodies that circulate in the recipient serum.
Antibody Isotypes: Structure and functions
- Antibodies can come in different varieties known as isotypes or classes
- There are five isotypes or classes of antibodies differentiated by the aminoacid sequences in the heavy-chain constant regions that confer class-specific structural and functional properties of antibody molecules: IgG, IgM, IgA, IgE, and IgD. They are characterized by the type of heavy chain they contain.
- They also differ in their biological properties, functional locations, and how they interact with different antigens,
Immunoglobulin G (IgG)
- Immunoglobulin G (IgG) antibodies are the most abundant class found in serum, making up to 80% of the total serum Immunoglobulin.
- They are larger globular proteins with a molecular weight of about 150 kDa composed of four peptide chains, consisting of two identical γ (gamma) heavy chains of about 50 kDa and two identical light chains of about 25 kDa, thus a tetrameric quaternary structure.
- It is the main immunoglobulin produced during a secondary immune response and is the only antibody with antitoxin activity.
- IgG is the only antibody to be transported across the placenta and provides long term protection because it persists for months and years after the presence of the antigen that has triggered their production.
- IgG protects against bacteria, viruses, neutralizes bacterial toxins, triggers complement protein systems and binds antigens to enhance the effectiveness of phagocytosis.
- There are four subclasses of IgG: IgG1, IgG2, IgG3, and IgG4, differentiated by the γ-chain sequences.
- IgG1, IgG3, and IgG4 readily cross the placenta and play an important role in protecting the developing fetus.
- IgG3 is the most efficient complement activator, followed by IgG1; IgG2 is lesser, and IgG4 is not able to activate complement at all.
- IgG1 and IgG3 bind with high affinity to Fc receptors on phagocytic cells and thus mediate opsonization.
- IgG4 has a transitional affinity for Fc receptors and IgG2 has an extremely low affinity.
Immunoglobulin M (IgM)
- IgM comprises about 5-10% of circulating immunoglobulins. It has a molecular weight of 970 kDa. Monomeric IgM, is found as a membrane-bound antibody on B cells.
- IgM secreted by plasma cells is pentameric, in which five monomer units are held together by disulfide bonds that link their 2- carboxyl-terminal heavy chain domains and two light chain domains, and the whole structure is stabilized the Joining (J)chain.
- The J chain is required for polymerization of the monomers to form pentameric IgM; it is produced just before the secretion of the pentamer.
- This immunoglobulin is largely confined to the peripheral circulation and is the principal antibody produced in a primary response.
- It is produced early in a secondary response, and with certain antigens is the sole antibody produced, e.g. natural blood group antibodies, therefore, it’s involved in the ABO blood group antigens on the surface of RBCs.
- IgM enhance ingestions of cells by phagocytosis
Immunoglobulin A (IgA)
- Although IgA constitutes only 10%–15% of the total immunoglobulin in serum, it is the predominant immunoglobulin class in external secretions such as breast milk, saliva, tears, and mucus of the bronchial, genitourinary, and digestive tracts.
- It consists of heavy (H) and light (L) chains. Each H chain is comprised of the constant region (Cα1, Cα2, Cα3), hinge region, and the Variable (V) region.
- Light chains consist of the CL and Vκ or Vλ elements. In serum, IgA exists primarily as a monomer, but polymeric forms (dimers, trimers, and some tetramers) are sometimes seen, all containing a J-chain polypeptide.
- The major function of IgA is to bind antigens on microbes before they attack tissues. It aggregates the antigens and keeps them in the secretions so when the secretion is expelled, so is the antigen.
- IgA is also the first defense for mucosal surfaces such as the intestines, nose, and lungs.
Immunoglobulin E (IgE)
- Immunoglobulin E (IgE) antibodies have only been found in mammals.
- IgE is synthesized by plasma cells. Monomers of IgE consist of two heavy chains (ε chain) and two light chains, with the ε chain containing 4 Ig-like constant domains (Cε1-Cε4).
- IgE antibodies facilitate the immediate hypersensitivity reactions that are responsible for the symptoms of hay fever, asthma, hives, and anaphylactic shock. The presence of a serum component responsible for allergic reactions.
- IgE mechanism involves binding to Fc receptors on the membranes of blood basophils and tissue mast cells. Cross-linkage of receptor-bound IgE molecules by antigen (allergen) induces basophils and mast cells to translocate their granules to the plasma membrane and release their contents to the extracellular environment, a process known as degranulation, resulting in a variety of pharmacologically active mediators are released and give rise to allergic manifestations.
- Localized mast-cell degranulation induced by IgE also may release mediators that assist in the buildup of various cells necessary for antiparasitic defense.
Immunoglobulin D (IgD)
- IgD comprises only less than 1% of serum, and its expressed in the plasma membranes of immature B-lymphocytes, during differentiation to mature IgM. IgD is also produced in a secreted form that is found in small amounts in blood serum.
- IgD may be involved in humoral immune responses by regulating B cell selection and homeostasis.
- IgD may also play a role in the induction of antibody production.
Table: Antibodies isotypes, Structure, and Functions
| Antibody isotype | Subclasses | Structure | Functions |
| IgG | 4 (IgG1, IgG2, IgG3, IgG4) | Pentamer; Molecular weight; 146kDa |
|
| IgM | 1 (IgM) | Monomeric and/or Pentameric
Molecular weight; 970 kDa
|
|
| IgA | 2 (IgA1 and IgA2) | Monomeric and/or Polymeric forms e.g. dimers
Molecular weight; 385 kDa
|
|
| IgE | 1 (IgE) | Monomeric |
|
| IgD | – | Monomeric
Molecular weight;185kDa |
|
Antigenic Determinants (Epitopes) on Immunoglobulins
As glycoproteins, antibodies can act as immunogens hence inducing an antibody response. However part of the immunoglobulin can act as an immunogen and not the entire antibody, only the specific sites known as the antigenic determinants (epitopes). This understanding has paved way for studies of the B-cell development and humoral immune responses. Based on the location of these epitopes on the antibody molecule, they can be classified into three: isotypic, allotypic, and idiotypic.
A. Isotypic determinants
- The antibody molecules each have one type of heavy chain(γ, or α, or μ, or ε, or δ) and one type of light chain (k or λ).
- The differences in the structure of the constant region of a heavy chain or light chain determine immunoglobulin (Ig) class, its subclasses, types, and subtypes within a given species.
- Therefore the difference in the constant region defines the isotypic determinants (isotypes).
- Each isotype is encoded by a separate constant region gene.
- Different species inherit different constant-region genes and therefore express different isotypes while members of the same species have the same constant region gene.
- When an antibody of a different species is injected into another species, the new isotypic determinants will be recognized as foreign, forming an anti-isotypic antibody.
B. Allotypic determinants
- Members of the same species inherit the same isotypic genes however, some genes have the existence of multiple alleles and these alleles encode small amino acid differences. This means that the products of the allelic forms of the genes will have some different amino acid sequences in the constant regions known as the allotypic determinants.
- The total number of allotypic determinants displayed by an antibody determines its allotype.
- In humans, allotypes have been characterized for the four (4) subclasses of IgG, the IgA2 subclasses, and the Kappa light chain.
- The gamma-chain allotypes are known as the Gm markers and there are about 24 Gm allotypes that have been identified. They are designated by the class, subclass, and allele number. For example G1m (1), G2m (23), G3m (11), G4m (4a).
- The IgA2 subclass has two allotypes (A2m (1) and A2m (2)).
- The Kappa (κ) light chain has 3 allotypes (Km (1), Km (2), and Km (3)).
- Each of these allotypic determinants represents differences in one to four amino acids that are encoded by different alleles.
- Production of antibodies to allotypic determinants can be done by injecting the antibodies from one member of a species into another, carrying the allotypic determinants. For example, a pregnant mother produces antibodies to the paternal allotypic determinants present on the fetal immunoglobulin. Additionally, anti-allotypic determinants can also be produced during blood transfusion.
C. Idiotypic determinants
- The amino acid sequence of the VH and VL domains of an antibody can act as both an antigen-binding site and as an antigen determinant.
- The idiotypic determinants arise from the sequence of the Heavy and Light-chain variable regions and each individual antigenic determinant of the variable region is known as idiotope.
- The idiotope can act as an antigen-binding site and can also comprise the variable-region sequence outside the antigen-binding site.
- Each antibody will present multiple idiotopes and the total number of individual idiotopes is known as the idiotype of the antibody.
- Because the antibodies produced by individual B cells derived from the same clone have identical variable-region sequences,
they all have the same idiotype. - The idiotype refers to a specificity that is associated with the variable region. Idiotype markers are found on the hypervariable region of the immunoglobulin.
- The anti-idiotype antibody is produced by injecting antibodies that have minimal variation in their isotypes and allotypes so that the idiotypic difference can be recognized. Often a homogeneous antibody such as myeloma protein or monoclonal antibody is used. Injection of such an antibody into a recipient who is genetically identical to the donor will result in the formation of an anti-idiotype antibody to the idiotypic determinants.
Antigen Binding Sites (B-cell Receptors)
- B-cell receptor (BCR) is a transmembrane protein complex composed of mIg and disulfide-linked heterodimers called Ig-α/Ig-β.
- Molecules of this heterodimer associate with a mIg molecule to form a BCR.
- The Ig-α chain has a long cytoplasmic tail containing 61 amino acids; the tail of the Ig-β chain contains 48 amino acids.
- The tails in both Ig-α and Ig-β are long enough to interact with intracellular signaling molecules.
- The Discovery of the Ig-α/Ig-β heterodimer by Michael Reth and his colleagues in the early 1990s has substantially furthered understanding of B-cell activation
Antibody (Immunoglobulin) diversity
- The immune system has the ability to generate a high level of diversity in order to recognize a very vast range of unique molecules estimated to be in billions. With this capability, foreign structural, foreign antigens can be recognized and eliminated.
- The B-cells antigenic specificity is determined by the membrane-bound antigen-binding receptor (antibody). The antibodies are expressed on the surface of the B-cells, which matures in the bone marrow.
- During maturation, the B-cells’ specificity is generated by the random arrangement of a series of gene segments that encode the antibody molecule.
The various diversities are discussed below:
Domain variability
- Several distinct gene loci that encode the antibody on the chromosomes are large and they code for each domain of the antibody.
- This is the chromosomal region that contains the heavy chain genes (IGH@), and it is found on the 14th chromosome and the loci containing lambda and kappa light chain genes (IGL@ and IGK@) are found on chromosomes 22 and 2 in humans.
- One of these domains is called the variable domain. It is present in each heavy and light chain of every antibody but can differ in different antibodies generated from distinct B cells.
- Differences that arise from the variable domains are located on three loops known as hypervariable regions (HV-1, HV-2, and HV-3) or complementarity-determining regions (CDR1, CDR2, and CDR3).
- CDRs are supported within the variable domains by conserved framework regions.
- The heavy chain locus contains about 65 different variable domain genes that all differ in their CDRs.
- The combination of these genes with a range of genes for other domains of the antibody does generate a largeCombining these genes with an array of genes for other domains of the antibody mounts a large troop of antibodies with a high degree of variability.
- This combination is known as the V(D)J recombination.
V(D) J recombination
- Antibodies and T-cell receptors are made up of two polypeptide chains, each of which contributes to the antigen-binding domain.
- The exons that encode the antigen-binding domains are assembled from the V (variable), D (diversity), and J (joining) gene segments by “cut and paste” DNA rearrangements. This process is termed as V(D)J recombination.
- The process chooses a pair of segments, introduces double-strand breaks adjacent to each segment, deletes or inverts the intervening DNA, and ligates the segments together.
- Therefore V(D)J recombination is a somatic recombination process of immunoglobulins, that involves generating unique immunoglobulin variable regions.
- Each of the variable regions of the antibody, heavy and light chain is encoded in several pieces known as gene segments also known as subgenes. These are the variable, V, diversity, D, and joining segments.
- V, D, and J segments are found in Ig heavy chains, but only V and J segments are found in Ig light chains.
- Several copies of the V, D, and J gene segments exist and are tandemly arranged in the genomes of mammals.
- In the bone marrow, each developing B cell will assemble an immunoglobulin variable region by randomly selecting and combining one V, one D, and one J gene segment (or one V and one J segment in the light chain).
- Because of the existence of multiple copies of each type of gene segment with different combinations of the gene segments that can be used to generate each immunoglobulin variable region, the V(D)J recombination can be able to generate a large number of antibodies, each with different paratopes, and hence different antigenic specificities.
- Recombination Activating Gene (RAG) proteins play an important role with V(D)J recombination in cutting DNA at a particular region. These proteins necessitate V(D)J recombination mechanism and without them, these recombination processes may not occur.
- After a B cell produces a functional immunoglobulin gene during V(D)J recombination, it cannot express any other variable region (a process known as allelic exclusion) thus each B cell can produce antibodies containing only one kind of variable chain.
Somatic hypermutation and affinity maturation
- Somatic hypermutation is a process that allows B cells to mutate the genes that they use to produce antibodies. This enables the B cells to produce antibodies that are better able to bind to bacteria, viruses, and other infections.
- When B-cells become activated by encountering an antigen, they proliferate rapidly, and during this phase of proliferation, the genes that encode for the variable domains of the heavy and light chain, undergo a high rate of point mutations. This is the process that is referred to as somatic hypermutation (SHM).
- SHM diversifies B-cell receptors used to recognize foreign antigens, allowing the immune system to adapt its response to new threats during the lifetime of an organism.
- The mutations caused by SHM result in an estimated one nucleotide per variable gene, per cell division, and therefore the daughter cells produced acquire some slight amino acid differences in the variable domains of their antibody chains.
- This increases the diversity of the antibody pool and hence impacting the antibody’s antigen-binding affinity.
- Some point mutations will result in the production of antibodies that have a weaker interaction (low affinity) with their antigen than the original antibody, and some mutations will generate antibodies with a stronger interaction (high affinity).
- B cells that express high affinity antibodies on their surface will receive a strong survival signal during interactions with other cells, whereas those with low-affinity antibodies will not, and will die by apoptosis.
- Thus, B cells expressing antibodies with a higher affinity for the antigen will outcompete those with weaker affinities for function and survival allowing the average affinity of antibodies to increase over time.
- The process of generating antibodies with increased binding affinities is called affinity maturation.
- Affinity maturation occurs in mature B cells after V(D)J recombination and is dependent on help from helper T cells.
Class switching
- Class switching, also known as isotype switching is a biological process occurring after activation of the B cell, which allows the cell to produce different classes of antibody (IgA, IgE, or IgG), by a mechanism known as Class Switching Recombination (CSR).
- The different classes of antibody, and thus effector functions, are defined by the constant (C) regions of the immunoglobulin heavy chain and therefore during class switch recombination the constant region portion of the antibody heavy chain is changed, but the variable region of the heavy chain stays the same; thus, class switching does not affect antigen specificity. This means that the antibody retains an affinity for the same antigens, but can interact with different effector molecules.
- Initially, immature naive B- cells express only cell-surface IgM and IgD, with identical antigen-binding regions.
- When the immature B-cell is activated majorly by cytokines, it encounters specific signaling molecules via their CD40 and cytokine receptors (both modulated by T helper cells). They then undergo antibody class switching to produce IgG, IgA, or IgE antibodies that have defined roles in the immune system.
- Each isotype is adapted for a distinct function; therefore, after activation, an antibody with an IgG, IgA, or IgE effector function might be required to effectively eliminate an antigen.
- Class switching allows different daughter cells from the same activated B cell to produce antibodies of different isotypes.
- Thus the progeny of a single B cell can produce antibodies, all specific for the same antigen, but with the ability to produce the effector function appropriate for each antigenic challenge.
- Class switching is triggered by cytokines; the isotype generated depends on which cytokines are present in the B cell environment.
- Mechanism of class switching: Repetitive areas of DNA known as ‘switch regions’ are found in the introns upstream of each isotype gene, which is used to guide AID and other enzymes to the site. These then create nicks in the DNA sequence, allowing the μ genes to be excised and then a repair enzyme to come along and join the VDJ segment back on to the new constant region.
Specific designations
- An antibody can be called monospecific if it has specificity for the same antigen or epitope, or bispecific if they have an affinity for two different antigens or two different epitopes on the same antigen.
- A group of antibodies can be called polyvalent (or unspecific) if they have an affinity for various antigens or microorganisms.
- Intravenous immunoglobulin consists of a variety of different IgG (polyclonal IgG).
- In contrast, monoclonal antibodies are identical antibodies produced by a single B cell.
Medical and Research Applications of Antibodies
- Serological testing aim at detecting specific antibodies produced against particular antigens. The application methods vary, for example, a biochemical assay for the diagnosis of Epstein- Barr Virus, Lyme disease detects the presence of antibodies against these diseases.
- Nephelometry/turbidimetry is a measure of the level of individual classes of immunoglobulin. It gives an antibody profile of the patient and an increase on the level of immunoglobulin classes could be an indicator of liver damage in patients whose diagnosis is not clear, for example, increased levels of IgA indicates alcoholic cirrhosis, elevated IgM indicated viral hepatitis and primary biliary cirrhosis while IgG is elevated in viral hepatitis, autoimmune hepatitis, and cirrhosis.
- Coombs test is an antibody test used to detect the presence of autoantibodies directed against the surface of Red blood cells, and it is used test for auto-mediated hemolytic anemia. It is also used for blood screening prior to a blood transfusion.
- Antibodies are a major component for several immunological diagnostic tests for detection of the presence of an antigen such as ELISA, western blotting, immunofluorescence, immunodiffusion, electrophoresis, etc.
- Antibody therapy has also been directed to treat diseases and disorders associated with low or deficiencies of antibodies such as X-linked agammaglobulinemia, hypogammaglobulinemia
- Monoclonal antibodies are used for the treatment of diseases such as rheumatoid arthritis, multiple sclerosis, psoriasis, and many forms of cancer including non-Hodgkin’s lymphoma, colorectal cancer, head and neck cancer, and breast cancer, using monoclonal antibodies targeted therapy.
- The Rh D antigen found on the red blood cells, also known as the Rh factor. An Rh-positive (Rh+) individual has the Rh antigen on their red blood cells and an Rh-negative
- Rh factor, also known as Rh D antigen, is an antigen found on red blood cells; individuals that are Rh-positive (Rh+) have this antigen on their red blood cells and individuals that are Rh-negative (Rh–) do not. At birth, a child’s blood can enter the circulating system of the mother, and due to incompatibilities of the Rh antigen (Rh- mother and Rh+ baby), the mixing of the blood can sensitize the Rh- mother to the Rh antigen of the blood cells of the Rh+ baby which posses a problem to the subsequent pregnancies due to the risks of hemolytic disease of the newborn. However thought research, immune globulin specific for the human anti-RhD antibodies known as the Rho (D) immune antibodies have been produced which aid in prenatal treatment preventing the sensitization of the Rh-negative and Rh-positive interactions between the mother and the baby/fetus. Treatment of a mother with Anti-RhD antibodies prior to and immediately after trauma and delivery destroys Rh antigen in the mother’s system from the fetus. These are the stages that initially stimulated the memory B-cells to remember the Rh antigen, and therefore the Rho immune antibodies prevent these activations.
Research Applications of Antibodies
- Antibodies can be produced by injecting an antigen into a lab animal such as a mouse, rat, rabbit, or horse. This enables a large production of antibodies from the blood of these animals.
- The blood that is isolated from these animals contains polyclonal antibodies in their serum (antiserum). These are multiple antibodies that bind to the same antigen.
- Antigens can also be injected into the chicken to enable the generation of polyclonal antibodies in the egg yolk. The specific antibodies for single epitopes of an antigen can be obtained by isolation from animals and fusion into cancer cell lines. The fused cells are known as hybridomas. They have the ability to continuously grow and secrete antibodies in culture.
- Single hybridoma cells can be isolated by dilution cloning to generate cell clones that produce the same antibody, and these antibodies are known as monoclonal antibodies.
- Polyclonal and monoclonal antibodies are purified using antigen-affinity chromatography, and they can be used for many research applications such as the identification of intracellular and extracellular proteins.
- Antibodies are used in flow cytometry to differentiate cell types by the proteins they express.
- They are also used in immunoprecipitation to separate proteins and anything bound to them (co-immunoprecipitation) from other molecules in a cell lysate, Western blot to identify proteins separated by electrophoresis, and in immunohistochemistry or immunofluorescence to examine protein expression in tissue sections or to locate proteins within cells with the assistance of a microscope.
- Proteins can also be detected and quantified with antibodies, using ELISA and ELISpot techniques.
References and Sources
- Kuby Immunology 5th Edition
- Kuby Immunology, 7th Edition
- Lippincott Immunology Review
- https://www.livescience.com/antibodies.html#:~:text=Antibodies%20are%20specialized%2C%20Y%2Dshaped,and%20marking%20it%20for%20destruction.
- https://www.britannica.com/science/antibody
- https://en.wikipedia.org/wiki/Antibody
- https://www.genome.gov/genetics-glossary/Antibody
- https://med.libretexts.org/Bookshelves/Anatomy_and_Physiology/Book%3A_Anatomy_and_Physiology_(Boundless)/20%3A_Immune_System/20.6%3A_Humoral_Immune_Response/20.6B%3A_Structure_and_Function_of_Antibodies
- https://study.com/academy/lesson/what-are-antibodies-definition-function-types.html
- https://www.nature.com/subjects/somatic-hypermutation
- https://bio.libretexts.org/Bookshelves/Microbiology/Book%3A_Microbiology_(Boundless)/11%3A_Immunology/11.07%3A_Antibodies/11.7D%3A_Isotype_Class_Switching
- Stavnezer, J., Guikema, J. E., & Schrader, C. E. (2008). Mechanism and regulation of class switch recombination. Annual review of immunology, 26, 261–292. https://doi.org/10.1146/annurev.immunol.26.021607.090248
- https://www.sciencedirect.com/topics/immunology-and-microbiology/somatic-hypermutation
- Roth D. B. (2014). V(D)J Recombination: Mechanism, Errors, and Fidelity. Microbiology spectrum, 2(6), 10.1128/microbiolspec.MDNA3-0041-2014. https://doi.org/10.1128/microbiolspec.MDNA3-0041-2014
- https://www.cell.com/fulltext/S0092-8674(02)00675-X

Good