Gas Liquid chromatography (GLC) is a type of gas chromatography (GC) that uses a mobile gas phase and a stationary liquid phase to separate and analyze volatile compounds in a mixture.
It is also known as gas liquid partition chromatography (GLPC), as it separates compounds based on their partition between the gas phase and the liquid phase.

GLC was first developed in the early 1950s by Archer J. P. Martin and Anthony T. James. It is based on the principle of partition chromatography introduced by Martin and Richard L.M. Synge, who were awarded the Nobel Prize in Chemistry in 1952 for their work.
Principle of Gas Liquid Chromatography
GLC separates sample components based on their partitioning between the mobile phase and the stationary phase. Here, the mobile phase is an inert carrier gas, and the stationary phase is a liquid film coated on a solid support within a column.
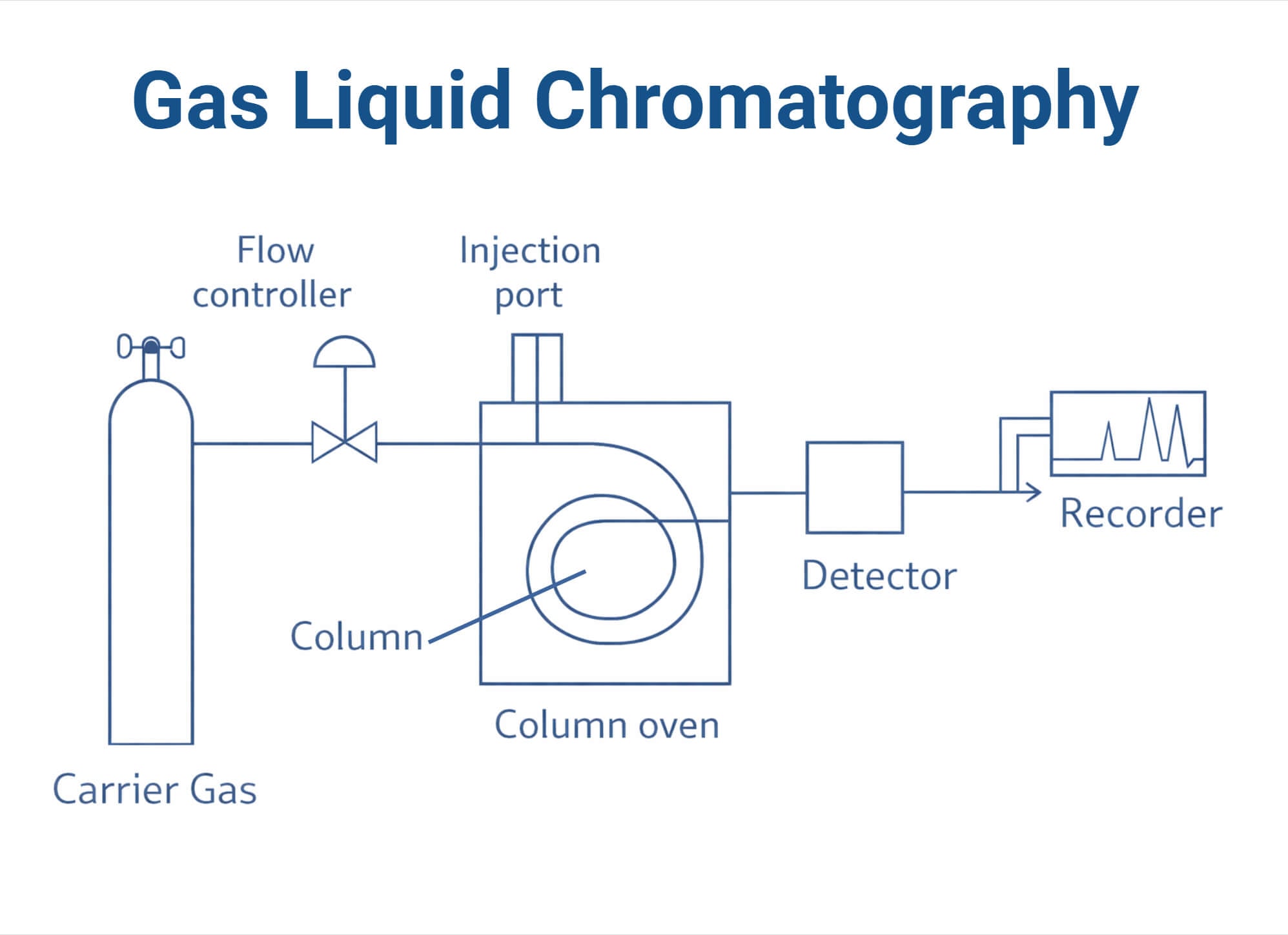
When the sample is introduced into the GLC system, it is vaporized and carried through the column by the mobile carrier gas. As the sample moves through the column, each component interacts differently and distributes between the gas and liquid phases based on its properties, such as boiling points, polarity, and volatility. More volatile compounds remain in the gas phase longer and move faster through the column to elute first. Less volatile compounds spend more time in the liquid phase, which results in slower movement and longer retention times. Each compound elutes at a unique retention time, which is detected and recorded by the system as a chromatographic peak. The peak position and peak area are used to identify and quantify the compounds.
Components of Gas Liquid Chromatography
- Sample Injection Port: This is the inlet for introducing the sample into the system. It is heated to vaporize the sample. Samples are injected using micro syringes or autosampler through a septum into the heated injector. The injection method depends on the properties of the sample.
- Column: It is the tube where the separation occurs. Two types of columns are used for GLC: packed columns and capillary columns. Packed columns are filled with inert solid support materials coated with a liquid stationary phase. Capillary or open-tubular columns are tubes with a stationary phase coated on the inner wall. The column is placed within an oven that maintains a controlled temperature, which can be kept constant (isothermal) or gradually increased during analysis (temperature programming).
- Mobile Phase or Carrier Gas System: GLC uses inert carrier gases as a mobile phase to move the vaporized sample through the column. Some of the commonly used carrier gases are helium, nitrogen, argon, and hydrogen. They are stored in high-pressure cylinders, and their flow rate is controlled using flow meters and pressure gauges.
- Stationary Phase: The stationary phase in GLC is a film of liquid coated on an inert support or on the inner surface of the column. Stationary phases can be polar or non-polar. Polar stationary phases include polyethylene glycol and polyesters. They separate solutes based on polarity and functional groups. Non-polar stationary phases separate compounds based on their boiling points.
- Detector: Detectors are used to identify and quantify the compounds eluted from the column. They can be grouped into concentration or mass detectors. Concentration detectors measure the concentration of analytes, while mass detectors measure the total mass or the mass flow. Detectors can also be universal or selective. Universal detectors respond to all compounds, while selective detectors detect specific groups. Some of the commonly used detectors in GLC are the flame ionization detector (FID), electron capture detector (ECD), and thermal conductivity detector (TCD).
FID is the most widely used detector due to its high specificity. Here, the sample is burned in a hydrogen-air flame, which produces ions from organic compounds. These ions generate an electrical current proportional to the amount of carbon present, which is measured as a peak.
TCD detects changes in the thermal conductivity of the carrier gas. When a sample component passes through the detector, it changes the heat loss from a heated filament, and this change is detected as a signal. It is a universal detector that responds to all compounds.
ECD detects compounds that capture electrons, like halogenated or organometallic compounds. A radioactive source emits electrons, and when an electronegative compound passes through, it captures some electrons, which reduces the current. The difference in current is recorded.
- Data System: The detector signals are sent to an amplifier, chart recorder, or computer system and converted into a chromatogram, which shows peaks for each separated compound. Each peak represents a specific compound, with its retention time indicating the identity of the compound and peak area indicating its concentration.
Procedure or Steps of Gas Liquid Chromatography
Sample Preparation for Chromatography
Sample preparation is necessary to make the sample compatible with the separation process. Proper sample preparation allows accurate analysis of compounds. It removes impurities and interferences that could affect the quality of the separation. Common steps of sample preparation include dilution, extraction, filtration, concentration, and derivatization. Dilution and filtration are basic methods that remove impurities. More advanced techniques include extraction methods like solid-phase extraction (SPE), solid-phase microextraction (SPME), liquid-liquid extraction (LLE), headspace sampling, and the purge-and-trap method. For non-volatile compounds, derivatization is used to modify them into a volatile and thermally stable form.
Sample Injection and Vaporization
- Suitable volume of sample mixture is introduced into the column through the inlet using a micro syringe or autosampler. The syringe needle passes through a rubber disc on the injection port called a septum.
- The port is maintained at a high temperature to vaporize the sample instantly when injected. This vaporized sample is carried through the column by the carrier gas.
- Different methods of injection can be used, like split/splitless injection, direct injection, or on-column injection. The injection method depends on the sample concentration and volatility.
- Liquid samples are usually diluted and introduced using micro syringes. Solid samples are dissolved in a suitable solvent and introduced into thin glass ampoules that can be vaporized in the injector. Gaseous samples are introduced using a gas-tight syringe.
Separation
- The vaporized sample enters the GLC column, which is either a packed column or a capillary column that contains a stationary liquid phase.
- Each sample compound partitions between the stationary and mobile phases according to its volatility and solubility.
- More volatile compounds remain longer in the gas phase and elute earlier, while less volatile compounds will spend more time absorbed into the liquid stationary phase and are retained longer.
- So, individual sample components are eluted from the column at a different retention time.
Detection
- The compounds eluted from the column pass through a detector that monitors the elution.
- These detectors generate electrical signals that are processed by a data system to generate a chromatogram.
- The most widely used detectors in GLC are FID, TCD, and ECD. Each has its own advantages and limitations, so detector selection depends on the specific application.
Data Analysis
- The resulting chromatogram is a graph of signal intensity versus time.
- It shows a series of peaks where each peak represents a distinct compound in the sample, and the area under each peak represents the concentration of that compound.
Factors Affecting Gas Liquid Chromatography
- The flow rate of carrier gas can affect the speed and resolution of separation. Higher flow rates lead to shorter analysis times but reduce resolution, while lower flow rates improve separation but take longer. An optimal flow rate of carrier gas is necessary to ensure sharp peaks and better separation.
- Sample size and amount can affect the separation process. Too large a sample can overload the column and distort peaks.
- Column type and dimensions, like length and diameter, are also important factors. Longer and narrower columns improve resolution but increase analysis time, while shorter and thicker columns allow faster analysis but can reduce resolution. Capillary columns are most commonly used due to their high resolution.
- Column temperature also affects the separation. Temperature directly affects the volatility and elution rate of the compounds. Higher temperatures reduce retention time but can also reduce resolution, while lower temperatures improve separation but take longer.
- The nature of the stationary phase is also important. The polarity and thermal stability of the liquid stationary phase determine the resolution of the separation. Suitable phase should be selected to match the polarity and volatility of the sample.
Common Products and Manufacturers of Gas Liquid Chromatography
| Common Products | Manufacturers |
| 8850 GC system, Intuvo 9000 GC system, 8890 GC system, 7693A Automatic Liquid Sampler, 8697 Headspace Sampler, GC Autosampler Syringes, J&W GC Columns | Agilent |
| TRACE 1600 series GC, TriPlus RSH SMART Autosampler, TraceGOLD columns, iConnect injectors | Thermo Fisher Scientific |
| Brevis GC-2050, GC-2010 Pro, GC-2014, SH series GC columns, AOC-30 Autoinjector/Autosampler series | Shimadzu |
| Clarus GC series, GC 2400 system, TurboMatrix HS 16 | PerkinElmer |
| Zebron GC columns, Inlet liners, GC syringes | Phenomenex |

Applications of Gas Liquid Chromatography
- GLC is used in environmental testing to analyze pollutants like volatile organic compounds, including pesticide residues and chlorinated compounds in the environment.
- It is used in pharmaceuticals for quality control and analysis of drug formulations.
- It is used for quality control and safety testing in the food industry. It can also be used to identify flavor compounds.
- It is used in the petrochemical industry to analyze petroleum products and processes.
- It has applications in forensic investigations to identify illicit compounds and analyze samples in crime scenes.
Advantages of Gas Liquid Chromatography
- GLC can resolve complex mixtures into individual components with high accuracy.
- It provides high sensitivity, so very small quantities of compounds can also be detected.
- It also provides fast analysis compared to other chromatographic methods.
- It is versatile and can be used to analyze a wide range of volatile compounds.
- A small amount of sample can be used for complete analysis.
- It can be coupled with other analytical techniques like mass spectrometry for more accurate identification and quantification.
Limitations of Gas Liquid Chromatography
- GLC is only suitable for analyzing volatile and thermally stable compounds.
- Some samples cannot be directly analyzed and need additional preparation steps, like derivatization, to make them suitable for separation.
- GLC instruments and consumables can be expensive and less accessible for small laboratories.
- Factors like temperature and phase selection should be carefully optimized.
- GLC is highly sensitive to contaminants, which can affect the accuracy of the analysis.
- Operating a GLC system and interpreting the results may require specialized training.
Gas Liquid Chromatography Troubleshooting and Safety Considerations
Troubleshooting in any system begins by isolating the source of the problem. Most issues in GLC systems arise from these components: injector, flow system, column, detector, and electronics.
- Ghost peaks or carryover peaks are unexpected peaks or peaks from compounds left from previous injections. They may be caused by sample carryover (back flash) and a contaminated injector, column, or carrier gas. These unwanted peaks can be removed by regular maintenance, proper cleaning or replacement of contaminated parts, verifying injector conditions, and checking carrier gas purity.
- Peak fronting occurs when the peak has an asymmetrical leading edge. It may be caused by column overload, improper injection, column contamination or solubility mismatch. This can be solved by diluting the sample, cleaning or replacing the column, changing the solvent, and ensuring proper injector performance.
- Peak tailing occurs when a peak has an asymmetrical trailing edge. This can occur due to column contamination or activity, poor column installation, or improper injection. Solutions include cleaning or replacing the column, reinstalling the column correctly, and ensuring proper injection.
- Peak splitting occurs when one compound produces two peaks. This can be caused by manual injection, improper column installation, incorrect injector temperature, or incompatible solvent. This can be resolved by using an autosampler, reinstalling the column correctly, adjusting the injector temperature, and using an appropriate solvent.
- High baseline noise occurs due to a contaminated detector, column bleed, or electronic interference. Regular maintenance of the detector and using high-purity gases and solvents can help reduce noise.
- Retention time shifts are caused by column overload, leaks, injection or temperature issues, and can be corrected by diluting the sample, replacing the septum, proper injection, checking leaks, and adjusting column temperature.
- GLC involves high temperatures, pressurized gases, and volatile chemicals, so proper safety practices are important.
- Gas cylinders must always be secured and regularly checked to ensure there are no leaks.
- Volatile and flammable solvents should always be handled in a fume hood or well-ventilated area. Protective equipment should be worn at all times to avoid exposure.
- All waste materials must be disposed of according to waste regulations.
Gas Liquid Chromatography Recent Advances and Innovations in GLC
- Advanced columns and novel stationary phases like porous polymers and monolithic columns have improved separation and enhanced selectivity, especially for complex volatile compounds.
- GLC can be integrated with advanced detectors like mass spectrometry (MS) for better qualitative and quantitative analysis.
- Sustainable and cost-effective practices are being adopted, such as using hydrogen or alternative carrier gases to reduce helium use.
- Use of advanced software and data analysis tools is also increasing, which improves peak detection and compound identification.
- Miniaturized and portable systems like micro-GC devices have been developed that allow on-site analysis in environmental and forensic applications.
- Multidimensional GC is also increasingly used to separate highly complex mixtures.
Conclusion
Gas-liquid chromatography (GLC) is an important analytical method that separates and identifies compounds in a mixture based on how they partition between the mobile gas phase and the stationary liquid phase. It is useful in many scientific and industrial applications, including checking drug purity and detecting environmental pollutants. Although GLC was developed many years ago, it is still relevant due to its high resolution and sensitivity for analyzing volatile compounds. Recent advances and innovations in GLC have further enhanced its performance.
References
- Acevedo, A. (2024, December 26). A Year in Review: Key Trends in Gas Chromatography for 2024. LCGC International – Separation Science News &Amp; Chromatography Insights. Retrieved from https://www.chromatographyonline.com
- Basic troubleshooting for GC systems. (2022, November 10). Retrieved from https://community.agilent.com/knowledge/gc-portal/kmp/gc-articles/kp533.basic-troubleshooting-for-gc-systems
- Drawell. (2025, July 7). GC sample preparation – Techniques and challenges – Drawell. Retrieved from https://www.drawellanalytical.com/gc-sample-preparation-techniques-and-challenges/
- Factors affecting chromatographic separation | Solubility of things. (n.d.). Retrieved from https://www.solubilityofthings.com/factors-affecting-chromatographic-separation
- Gas Liquid Chromatography (GLC): instrumentation. (n.d.). Retrieved from https://www.brainkart.com/article/Gas-Liquid-Chromatography-%28GLC%29%E2%80%93Instrumentation_30940/
- Libretexts. (2023, August 29). D. Gas-Liquid chromatography. Retrieved from https://chem.libretexts.org/Bookshelves/Analytical_Chemistry/Supplemental_Modules_(Analytical_Chemistry)/Instrumentation_and_Analysis/Chromatography/V._Chromatography/D._Gas-Liquid_Chromatography
- Sharma, K. (2024, October 17). Gas-Liquid Chromatography: instrumentation, applications. Science Info. Retrieved from https://scienceinfo.com/gas-liquid-chromatography-applications/
