Dye ligand chromatography is a type of affinity chromatography that uses synthetic dyes as ligands to purify target biomolecules, such as proteins and enzymes, from complex mixtures.
Unlike traditional affinity chromatography which uses biological ligands like antibodies, dye ligand chromatography uses synthetic dyes which are cheaper and more stable. These dyes act as pseudo-ligand that structurally resemble natural ligands and allow binding to target biomolecules. So, this method is also called pseudo-affinity chromatography.
- Dye ligand chromatography was first discovered in 1968 when researchers observed that Blue Dextran, which was originally used as a gel filtration marker, could bind specific proteins like pyruvate kinase.
- It was later identified that the binding affinity was due to the Cibacron Blue F3GA dye component in blue dextran.
- Cibacron Blue dye resembles the structural features of ADP-ribose in nicotinamide adenine dinucleotide (NAD+) which allows it to interact with nucleotide-binding sites in proteins.
- This finding led to the development of dye ligand chromatography as a protein purification method.
- Over time, many other synthetic dyes were developed. These dyes can bind proteins not just by mimicking biological ligands but also through hydrophobic and ionic interactions.
Principle of Dye Ligand Chromatography
Dye ligand chromatography works on the principle of selective interactions between synthetic dye molecules and target proteins. The synthetic dyes mimic the structure and binding characteristics of natural ligands such as cofactors or substrates. This allows the dye to interact with specific regions of the target molecule. This affinity between the dye and target molecule is used to separate and purify the molecules from a mixture.
This chromatographic method involves loading the sample mixture of target biomolecules onto a column packed with the dye-immobilized matrix. The dye is immobilized to a solid support matrix like agarose or cellulose. The target molecule binds specifically to the immobilized dye ligands and non-specific components are washed away. The bound target molecules are eluted by using an appropriate elution buffer containing high salt, altered pH, or other competing agents that disrupt the dye-protein interactions. The eluted fractions containing purified target molecules are collected for downstream applications.
Components of Dye Ligand Chromatography
- Column: The chromatography column holds the stationary phase and provides a controlled environment for separation. It can range from small-scale columns used for screening to larger preparative columns.
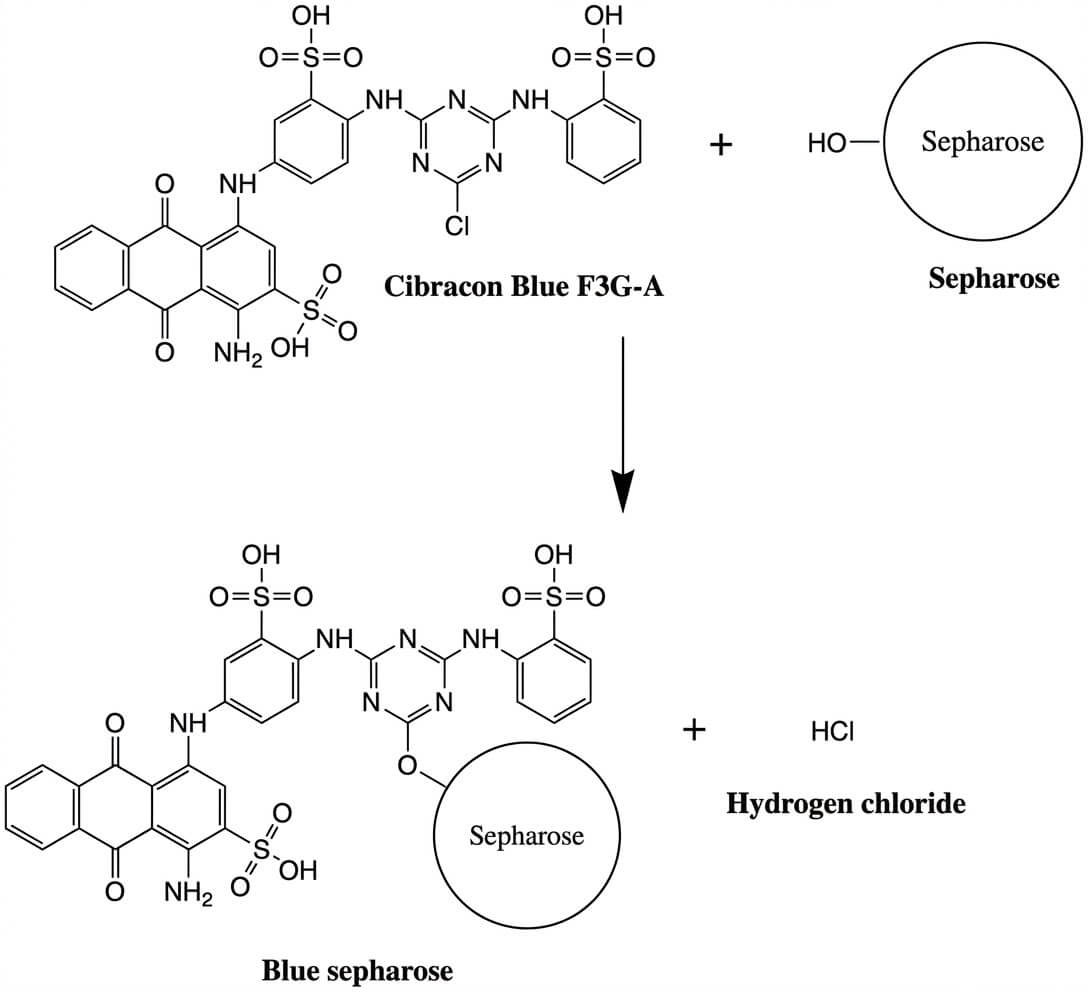
- Stationary Phase/Solid Matrix: The stationary phase in dye ligand chromatography consists of a porous support matrix such as agarose onto which synthetic dyes are covalently immobilized. It is hydrophilic and porous to allow protein diffusion and interaction. Functional groups like hydroxyl on agarose surface allow covalent attachment to the dye through reactive linkers like triazine ring systems.
- Mobile Phase/Buffer systems: The mobile phase consists of aqueous buffer solutions used for different steps like equilibration buffer that stabilizes the column, sample application buffer that carries the sample solution into the column, wash buffer that removes unbound components, and elution buffer to release the target molecule from the dye-ligand complex. Chaotropic agents like urea are also sometimes used to weaken non-covalent interactions during elution. They can help elute tightly bound proteins or reduce non-specific binding.
- Dye Ligands: These are synthetic molecules that mimic natural ligands and selectively bind target molecules through ionic, hydrophobic, and hydrogen bonding interactions. The dye molecule contains a chromophore which gives the color of the dye and a reactive group like chlorotriazine ring which allows covalent attachment to the solid matrix. Triazine-based dyes are widely used in this method. Some commonly used dyes are Cibacron Blue F3G-A, Procion Red HE-3B, Reactive Green 19, and Reactive Yellow 86.
- Spacer Arm: This is the linker molecule that connects the dye ligand to the solid matrix. It reduces steric hindrance and makes it easier for target molecules to reach and bind to the dye without being blocked.
- Sample: The sample contains a mixture of target molecules that need to be separated or purified. It is usually filtered or centrifuged to remove particulates before separation.
- Detectors: They are used to detect the presence and quantity of target molecules in each fraction. UV-Visible detectors are commonly used to monitor the elution. Additional methods like enzyme activity assays, SDS-PAGE, or Western blotting are also used.
- Fraction Collector: This is used to collect the eluted fractions that comes out of the column at the end of the chromatography system. This makes it easier to separate and analyze fractions containing target molecule.
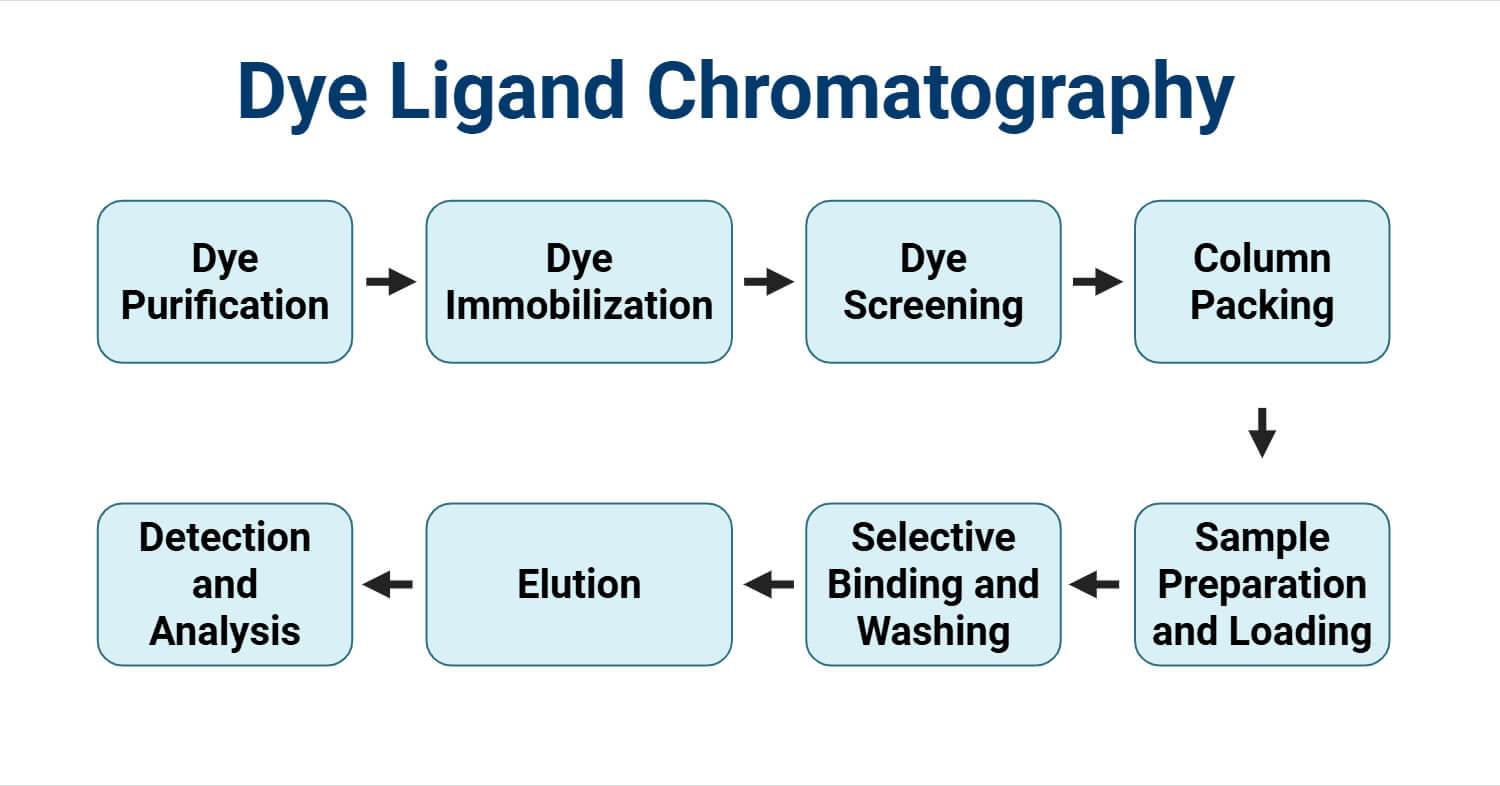
Procedure or Steps of Dye Ligand Chromatography
1. Dye Purification
Synthetic dyes are impure and may contain stabilizers or organic byproducts that can interfere with ligand binding. So, the dye must first be purified to ensure high specificity. Methods like gel filtration or column chromatography can be used to remove unwanted additives and contaminants.
2. Dye Immobilization
After purification, the dye is covalently attached to a solid matrix like agarose to create the stationary phase. Dye immobilization can be done in two ways:
- Direct coupling using reactive groups: In this method, the dye is reacted directly with a matrix that allows interaction between the reactive group of the dye like chlorotriazine ring and the hydroxyl group on the matrix. After coupling, the matrix is washed to remove unreacted dye.
- Indirect method using spacer arm: In this method, the dye is first modified with a spacer molecule. Spacer arm like hexamethyldiamine reduces steric hindrance and improves dye selectivity. The resulting dye-spacer conjugate is then attached to a pre-activated matrix. After immobilization, the matrix is washed to remove unbound or excess dye.
3. Dye Screening
It is important to screen multiple dye-immobilized matrices to identify the best ligand for binding the target protein. Small-scale columns can be packed with different dye-ligand gels and the target molecule is tested to determine the best dye for a specific molecule.
4. Column Packing
The dye-immobilized matrix is degassed to remove air bubbles and then packed into a suitable chromatography column carefully. A buffer wash is done to equilibrate the column and create a stable environment in the column before use.
5. Sample Preparation and Loading
The sample mixture is pretreated to remove salts, low molecular weight components, and other insoluble materials to prevent clogging. The prepared sample mixture is then loaded onto the equilibrated column at a controlled flow rate.
6. Selective Binding and Washing
Specific molecules bind to specific immobilized dye through affinity interactions. Non-target molecules or unbound components are washed off from the column by using wash buffer and the fraction is collected.
7. Elution
The target molecules bound to the ligand are eluted from the column using an elution buffer. Elution can be done by modifying the buffer pH, increasing ionic strength, or adding a competing ligand that displaces the target from the dye-ligand complex. The eluted fractions containing the purified target molecules are collected for downstream applications.
8. Detection and Analysis
The collected fractions are analyzed to determine the target molecules. Techniques like UV absorbance, SDS-PAGE, and other functional assays can be performed to analyze the fractions.
Factors Affecting Dye Ligand Chromatography
- The solvents used in the process is an important factor. Using non-denaturing solvents like glycerol and ethylene glycol can improve protein recovery.
- The pH of the buffer can affect the interactions between dye ligands and target proteins.
- Ionic strength or salt concentration also affects the interactions. Low ionic strength increases binding affinity but very low ionic strength can reduce protein solubility.
- Chaotropic agents and detergents can change protein conformation and interactions. Agents like urea and guanidine hydrochloride can help in disrupting non-specific interactions and improving elution.
- Factors such as dye concentration, buffer composition, flow rate, and column dimensions also affect the process.
- The type of dye ligand is also important as different dyes have different affinity towards biomolecules.
- The properties of the solid matrix such as pore size and surface area are also important factors affecting the separation process.
Common Products and Manufacturers of Dye Ligand Chromatography
| Common Products | Manufacturers |
| HiTrap Blue HP, Blue Sepharose 6 Fast flow | Cytiva |
| Affi-Gel Blue Gel, CM Affi-Gel Blue Gel, EconoFit Affi-Gel Blue Column | Bio-Rad Laboratories |
| Cibacron Blue 3GA Agarose, Reactive Blue 4, Procion Red, Hexamethylenediamine | Sigma-Aldrich (Merck) |
Applications of Dye Ligand Chromatography
- Dye ligand chromatography is used to separate and purify proteins and enzymes from complex mixtures.
- It is useful in proteomics research and biomarker discovery.
- It can be used in the pharmaceutical industry to purify drug intermediates or active pharmaceutical ingredients.
- It has applications in isolating specific proteins or enzymes as biomarkers for disease which is useful in research, diagnostic, or therapeutic use.
- It has industrial and bioprocess applications for large-scale purification of therapeutic proteins.
- It can be used to study protein-ligand interactions.
Advantages of Dye Ligand Chromatography
- Synthetic dyes are usually more stable and less expensive than biological ligands. They are also easy to immobilize onto solid matrices. This makes dye ligand chromatography suitable for large-scale purification applications.
- Elution can be done under gentle conditions like slight changes in pH or ionic strength which helps to preserve the biological activity of the molecule.
- The column can be reused multiple times due to the stability of the immobilized dyes.
- It may be used to purify proteins that have weak interactions with biological ligands which are difficult to isolate using traditional affinity chromatography methods.
Limitations of Dye Ligand Chromatography
- Dye ligand chromatography does not always provide high specificity. The immobilized dye may bind to multiple proteins that have similar properties which can lead to co-elution and reduced purity of the target molecule.
- While dye ligands are cheaper than biological ligands, they may be more expensive than other standard chromatographic resins.
- Due to strong binding interactions with the dye or elution conditions, some proteins may suffer from partial denaturation or inactivation.
- Binding and elution conditions including pH, ionic strength, and buffer composition must be carefully optimized which can be time-consuming.
Troubleshooting and Safety Considerations
- Using incorrect pH or ionic strength of buffer and incompatible dye can cause problems like low binding or no target retention. This can be solved by optimizing buffer conditions and screening for best dye.
- Strong interactions between the target molecule and dye can lead to poor elution of target molecules. Increasing the salt concentration or using chaotropic agents or competing ligands can solve this problem.
- Columns may be clogged by air bubbles or particulates. Sample should be prefiltered or centrifuged to remove debris and the buffer must be degassed to avoid air bubbles.
- Chemicals used in this method like dyes, chaotropic agents, and organic solvents can be toxic. So, it is necessary to carefully handle these chemicals and always use protective equipment.
- Dye-containing solutions must be disposed as chemical waste. Other chemical waste must also be disposed according to standard biosafety protocols.
Recent Advances and Innovations of Dye Ligand Chromatography
- Development of biomimetic and tailored dye ligands have increased specificity of the process.
- Advancements in solid supports like monolithic columns and nanoporous materials.
- Integration with other chromatography systems like HPLC.
- Use of environmentally friendly dyes immobilized on renewable support matrices derived from sustainable sources.
- Miniaturization of chromatographic system such as microcolumns or lab-on-chip platforms which reduce sample and reagent consumption.
- Use of solvent-free or low solvent elution methods to reduce chemical waste.
- Use of Artificial Intelligence (AI) in rapid screening of multiple dye-ligand matrices and optimization of process parameters.
Conclusion
Dye-ligand chromatography is an advancement in protein purification method which combines the principles of affinity chromatography with synthetic dyes. It can selectively bind and purify target biomolecules which is important in both research and industrial applications.
References
- Angal, S. (1988). Dye-Ligand Chromatography. In: Walker, J.M. (eds) New Protein Techniques. Methods in Molecular Biology, vol 3. Humana Press, 111–122. https://doi.org/10.1385/0-89603-126-8:111
- Boyer, P. M., & Hsu, J. T. (1993). Protein purification by dye-ligand chromatography. Advances in Biochemical Engineering, Biotechnology, 1–44. https://doi.org/10.1007/bfb0046571
- Chronopoulou, E. G., Premetis, G., Varotsou, C., Georgakis, N., Ioannou, E., & Labrou, N. E. (2020). Synthesis and evaluation of Dye-Ligand affinity adsorbents for protein purification. Methods in Molecular Biology, 201–215. https://doi.org/10.1007/978-1-0716-0775-6_15
- Debnath, S., Das, M., Mondal, S., Sarkar, B. K., & Babu, G. (2025). Advances in chromatography: contemporary techniques and applications. Essential Chem, 2(1), 1–27. https://doi.org/10.1080/28378083.2025.2466624
- Denizli, A., & Pişkin, E. (2001). Dye-ligand affinity systems. Journal of Biochemical and Biophysical Methods, 49(1–3), 391–416. https://doi.org/10.1016/s0165-022x(01)00209-3
- Gallant, S. R., Koppaka, V., & Zecherle, N. (2008). Dye Ligand Chromatography. Humana Press eBooks, 61–70. https://doi.org/10.1007/978-1-59745-582-4_5
- Labrou, N.E. (2000). Dye-Ligand Affinity Chromatography for Protein Separation and Purification. In: Bailon, P., Ehrlich, G.K., Fung, WJ., Berthold, W. (eds) Affinity Chromatography. Methods in Molecular Biology, vol 147, 129–139. Humana Press. https://doi.org/10.1007/978-1-60327-261-2_13
- Protein affinity chromatography. (n.d.). Retrieved from https://www.sigmaaldrich.com/NP/en/technical-documents/technical-article/genomics/dna-and-rna-purification/affinity-chromatography
- Worrall, A. F. (2022) Factors Effecting Dye Ligand Affinity Chromatography: A Purification Technique. Bio Bulletin, 8(4): 05-06.
- Yokoyama, M. (2022, December 1). Dye-ligand Affinity Chromatography for Protein Purification. News-Medical. Retrieved on June 19, 2025 from https://www.news-medical.net/life-sciences/Dye-ligand-Affinity-Chromatography-for-Protein-Purification.aspx.
