The chemiosmotic hypothesis is a principal theory of bioenergetics according to which living cells produce energy in the form of the universal currency of energy called ATP. The hypothesis was proposed by Peter Mitchell in 1961, in opposition to earlier assumptions that ATP was synthesised by use of high-energy chemical intermediates. Rather, Mitchell expected that in biological membranes, electron transport chains forged the movement of protons to build an electrochemical potential.
This chemical energy is then used by ATP synthase in the production of ATP. For this, he received a Nobel Prize in Chemistry in 1978. The chemiosmotic mechanism is now considered to be at the centre of mitochondrial respiration, chloroplast photophosphorylation, and bacterial energy metabolism.
What is the Chemiosmotic Hypothesis?
The chemiosmotic hypothesis is defined as the process of production of ATP due to the flow of protons across an electrochemical gradient through a biological membrane. When transferring the electrons, certain protein complexes pump the protons between the sides of the membrane. The selective permeability of the membrane causes the concentration gradient (differences in the levels of protons) as well as an electrical gradient (differences in charge). A combination of these gradients creates the proton-motive force, which is stored energy.
Once the protons come back through ATP synthase, the energy is captured by the enzyme and is utilised to change ADP and inorganic phosphate into ATP. Therefore, the hypothesis connects the electron transport with the ATP production by a proton gradient across the membrane as opposed to a chemical intermediate.
Key Postulates of the Chemiosmotic Theory
Several important postulates support Mitchell’s theory. The first is that electron transport chains act as proton pumps.
As electrons pass through carriers having greater and greater electron affinities, part of the energy is utilised to pump protons across the membrane.
The membrane is impermeable to protons.
Due to the lipid bilayer, protons cannot freely diffuse across the membrane. As a result, a gradient forms and can be maintained. This proton gradient stores potential energy.
The proton-motive force is produced by a combination of pH difference and charge separation. ATP synthase constitutes the only return pathway for protons. As the protons move through ATP synthase, the enzyme converts the energy of proton movement directly into ATP. It requires an intact membrane and a stable proton gradient to synthesise ATP. If the gradient is lost through uncouplers or membrane damage, then ATP production stops instantly. These postulates develop a clear framework relating electron transport, proton gradients, and ATP synthesis.
Proton-Motive Force (PMF): The driving energy for ATP synthesis is the proton-motive force. PMF has two components:
A chemical gradient- ΔpH: a difference in proton concentration across the membrane.
Electrical gradient- ΔΨ: difference in charge caused by protons being pumped to one side.
These gradients together exert a powerful force to return protons across the membrane. This flow occurs through ATP synthase, serving as the channel. As protons move through the enzyme, kinetic energy is converted to mechanical rotational energy, utilised by ATP synthase to catalyse ATP formation. The magnitude of PMF varies with metabolic activity, oxygen availability, and electron transport efficiency. A high PMF is essential in mitochondria for efficient oxidative phosphorylation.
The Role of Electron Transport Chains in Membrane Proton Pumping
Electron transport chains are responsible for establishing the proton gradient that chemiosmosis utilises. They contain carriers that are arranged in a sequential manner, embedded in membranes such as the inner mitochondrial membrane, thylakoid membrane of chloroplasts, or bacterial plasma membrane.
In mitochondria, Complexes I, III, and IV are proton pumps that pump protons from the matrix into the intermembrane space. NADH and FADH₂ donate electrons that ultimately reduce oxygen to water.
In chloroplasts, the thylakoid ETC includes Photosystem II, cytochrome b6f, and Photosystem I. Proton accumulation inside the thylakoid lumen is the result of electron transfer.
In bacteria, the plasma membrane contains simplified ETCs that still pump protons to generate PMF. In all systems, the ETC does not directly provide ATP; its primary role is to establish the proton gradient that will be utilised by ATP synthase for ATP synthesis.
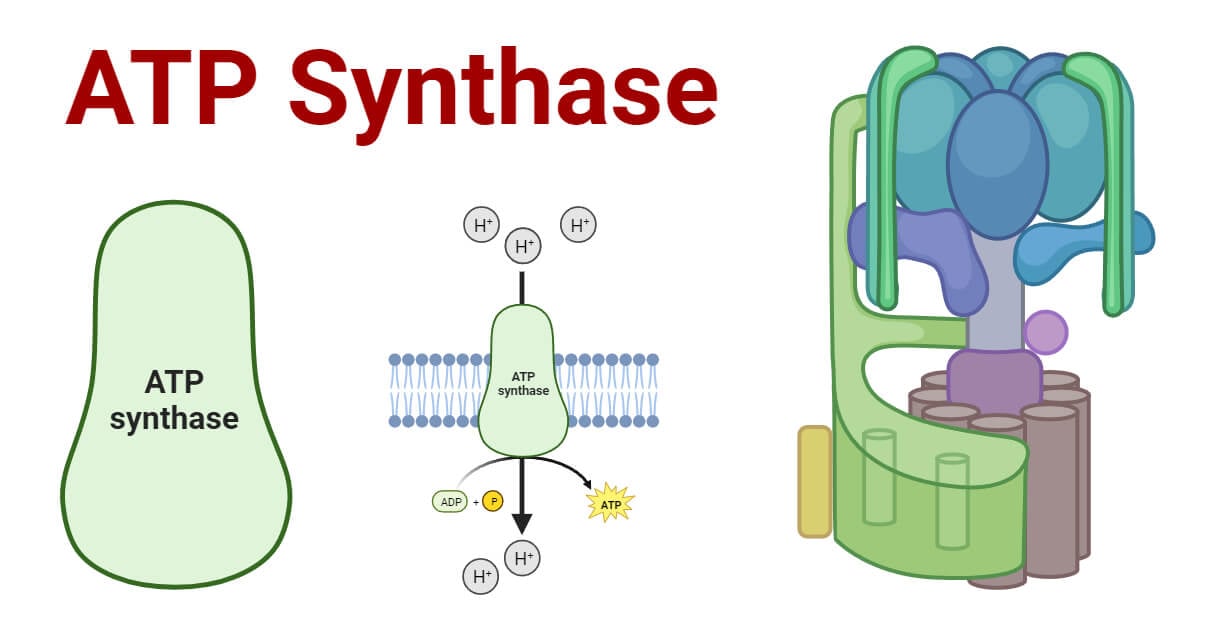
ATP Synthase Mechanism: F₀–F₁ Complex and Rotary Catalysis
ATP synthase is a highly conserved molecular machine of two major components:
F₀ Subunit- Embedded in the membrane.
Forms a proton channel
Contains the C-ring, which rotates as protons pass through it.
F₁ Subunit- Projects into the matrix or stroma
Contains three catalytic β-subunits responsible for ATP synthesis
The central γ-shaft rotates within the β-subunits
Rotary Catalysis- As protons move through F₀, the C-ring rotates, which thus turns the γ-shaft. This induces a shift of the β-subunits through three conformations: open, loose, and tight. During the tight conformation, ADP and phosphate are converted into ATP. This rotary mechanism is reversible, meaning ATP synthase can also hydrolyse ATP to pump protons when necessary. The efficiency and precision of this enzyme make it one of the most remarkable examples of biological nano-machinery.
Applications of the Chemiosmotic Hypothesis
Applications in Mitochondria
- Oxidative Phosphorylation: Oxidative phosphorylation is a process in which mitochondria use the energy derived from the oxidation of nutrients to generate ATP. Key points include:
Electrons from NADH and FADH₂ join the ETC.
Complexes I, III, and IV pump protons to the intermembrane space.
A strong PMF develops across the inner membrane.
ATP synthase utilises this gradient to synthesise ATP.
This process provides the majority of the ATP for cellular activities, including muscle contraction, biosynthesis, ion transport, and signalling. Defects in oxidative phosphorylation are responsible for severe metabolic diseases.
Applications in Chloroplasts
- Photophosphorylation in Plants- In chloroplasts, chemiosmosis drives photophosphorylation in the light-dependent reactions of photosynthesis. Light energises electrons in Photosystem II. Water is split to replace lost electrons, releasing oxygen and protons into the thylakoid lumen. The ETC pumps additional protons into the lumen. This steep proton gradient powers ATP synthase, an enzyme that generates ATP in the stroma.
The produced ATP is used in the Calvin cycle for carbon fixation and the synthesis of sugar. Hence, chemiosmosis plays a crucial role in the conversion of solar energy to chemical energy. Importance in Bacterial Respiration and Bioenergetics
Given that bacteria have no internal organelles, they rely heavily on chemiosmosis. All functions associated with the ETC and ATP synthase occur in their plasma membrane.
The protons are pumped outside the cell, generating PMF. The energy from the PMF is used to drive ATP synthesis, nutrient uptake, waste removal, and various transport systems. Many bacteria also use PMF to rotate their flagella. Some species produce sodium ion gradients instead of proton gradients. This flexibility enables bacteria to survive under various extreme conditions. Chemiosmosis, therefore, is important to microbial energy management and adaptation. Implications for cellular metabolism, evolution, and membrane biology
Ramifications of the Chemiosmotic Hypothesis:
Cellular Metabolism- By coupling ATP synthesis with proton gradients, chemiosmosis provides a common mechanism for energy conservation in cells. It influences metabolic rate, energy allocation, and adaptive responses.
Evolution- The universality of chemiosmosis in bacteria, mitochondria, and chloroplasts provides evidence for the endosymbiotic theory. Early efficient proton-pumping prokaryotes became ancestors of modern organelles.
Membrane Biology- The hypothesis emphasises the role of membrane structure, proton impermeability, and protein organisation. The loss of membrane integrity instantly stops ATP production. This underlines how membranes developed not only as barriers but as active participants in energy transformation. Overall, chemiosmosis illustrates basic concepts of physics and chemistry, forming the foundation for biological energy systems.
Conclusion
The chemiosmotic hypothesis put forward an all-encompassing explanation of how cells convert energy from the flow of electrons into ATP by means of a proton gradient. It united work on mitochondrial respiration, photosynthesis, and bacterial metabolism by demonstrating that ATP synthesis is dependent upon a proton-motive force originating from electron transport chains. Additionally, it became clear with the rotary mechanism of ATP synthase how very sophisticated biological energy conservation really is. Today, chemiosmosis remains one of the most influential and elegant principles in modern biology, shaping our views on metabolism, evolution, and membrane function.
References
- Admin. (2020, August 13). Chemiosmotic hypothesis. BYJUS. https://byjus.com/biology/chemiosmotic-hypothesis/
- Bhatia, J. (2025, February 5). Chemiosmotic Hypothesis, history and its Process. Physics Wallah. https://www.pw.live/school-prep/exams/chemiosmotic-hypothesis
- Braun, H. (2020). The Oxidative Phosphorylation system of the mitochondria in plants. Mitochondrion, 53, 66–75. https://doi.org/10.1016/j.mito.2020.04.007
- Chemiosmotic regeneration of ATP during respiration, fr the Ff1 model of ATPase & role in ATP synthesis. (n.d.). Slideshare. https://www.slideshare.net/slideshow/chemiosmotic-regeneration-of-atp-during-respiration-fo-f1-model-of-atpase-role-in-atp-synthesis/110009892
- Chemiosmotic theory | biochemistry | Britannica. (n.d.). Encyclopedia Britannica. https://www.britannica.com/science/chemiosmotic-theory
- Chemiosmotic theory. (n.d.). Slideshare. https://www.slideshare.net/slideshow/chemiosmotic-theory-249390132/249390132
- Fiveable. (2025, August 22). Chemiosmotic theory and proton-motive force. Fiveable. https://fiveable.me/biophysics/unit-8/chemiosmotic-theory-proton-motive-force/study-guide/xutpC6f2J1TVI93D
- Ghifari, A. S., Saha, S., & Murcha, M. W. (2023). The biogenesis and regulation of the plant oxidative phosphorylation system. PLANT PHYSIOLOGY, 192(2), 728–747. https://doi.org/10.1093/plphys/kiad108
- Mitchell, P. (2011). Chemiosmotic coupling in oxidative and photosynthetic phosphorylation. Biochimica Et Biophysica Acta (BBA) – Bioenergetics, 1807(12), 1507–1538. https://doi.org/10.1016/j.bbabio.2011.09.018
- Mitchell, P., & Moyle, J. (1967). Chemiosmotic hypothesis of oxidative phosphorylation. Nature, 213(5072), 137–139. https://doi.org/10.1038/213137a0
- Zharova, T. V., Grivennikova, V. G., & Borisov, V. B. (2023). F1·FO ATP Synthase/ATPASE: Contemporary View on Unidirectional Catalysis. International Journal of Molecular Sciences, 24(6), 5417. https://doi.org/10.3390/ijms24065417
