Hydrodynamic chromatography (HDC) is a liquid chromatography method used to separate particles in solution based on their size under laminar flow conditions.
Separation occurs according to how particles of different sizes sample the parabolic velocity field that develops in laminar flow. HDC is especially useful for particles in the micron-to-sub-micron range or for high-molar-mass polymers, including colloids, nanoparticles, emulsions, and macromolecules.

- The first experimental paper on HDC was published by Hamish Small in 1974. This showed the separation of polymer mixtures using columns packed with non-porous solid particles.
- This experimental work by Small was based on the theory developed by E. A. DiMarzio and C. M. Guttman in the late 1960s. They introduced the separation-by-flow theory, which described the size-based transport of particles in laminar flow fields.
- While HDC was practically demonstrated as a separation method by Small, the first HDC-type separation had already been reported by Pedersen in 1962, but it was not recognized as HDC at that time.
- HDC has many practical advantages, but it has remained only as a specialized method compared to other size-based separation methods like size-exclusion chromatography (SEC) and field-flow fractionation (FFF), which are more commonly used.
- While the introduction of multi-detector system resurged the interest in HDC during the period from 2005 to 2015, there seems to be a decline in publications in recent years.
- HDC is still considered an underutilized size-based separation method.
- The limited use of HDC can be due to less awareness and availability. Commercially available columns for HDC are not readily available. It is also practically used mostly in cases when the SEC is unsuitable. So, HDC has remained only as a niche method.
- If these limitations are well addressed, HDC with a multi-detector approach has huge potential for polymer and particle characterization.
Principle of Hydrodynamic Chromatography
HDC separates particles based on their size in solution and their behavior in a parabolic velocity profile that develops under laminar flow conditions. Separation occurs due to hydrodynamic effects and does not depend on chemical interactions or externally applied fields.
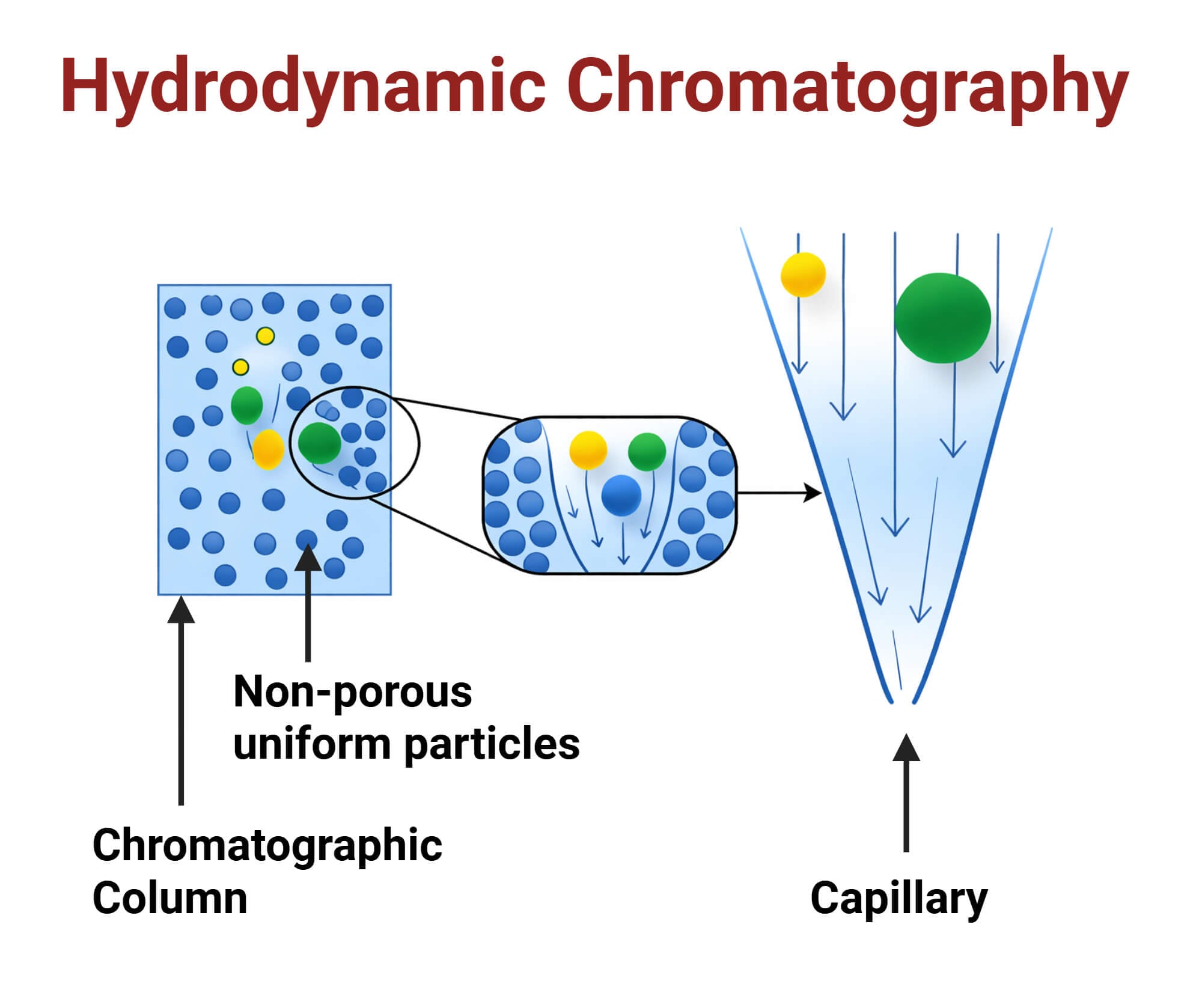
In HDC, the sample is introduced into an open capillary tube or into a column packed with non-porous particles. The sample moves through the system with a liquid mobile phase flowing under laminar flow conditions. In laminar flow, the liquid develops a parabolic or Poiseuille-like velocity profile, which means the fluid velocity is highest at the center and decreases toward the walls. Larger particles have a larger radius, so their center cannot approach the wall of the tube as closely as the center of a smaller particle.
So, larger particles remain in the central region of the tube while smaller particles can sample both fast and slow streamlines. As a result, larger particles move more rapidly and elute from the column before smaller particles, and separation occurs.
Components of Hydrodynamic Chromatography
- Mobile phase: It is the liquid that carries the sample through the capillary or column. The mobile phase composition is carefully selected to fully dissolve the sample and to minimize unwanted interactions with the column packing or capillary wall. Salts or surfactants can also be added to the mobile phase to suppress the interactions.
- Pump: It is used to deliver the mobile phase through the column or capillary at a constant and controlled flow rate. Separation depends on the velocity profile, which directly depends on the flow rate. So, it is important to maintain a stable flow in HDC.
- Injector: It introduces a small and well-defined volume of sample into the flowing mobile phase. Small injection volumes are used, as excessive volumes can degrade resolution or broaden bands.
- HDC column or capillary: Separation in HDC can occur in either an open capillary tube or a column packed with inert and non-porous beads. The separation principle is the same in both systems, as the interstitial channels in a packed column can be considered as a network of small open capillaries.
- Detector: The detector measures the components eluting from the column. Commonly used detectors in HDC are refractive index (RI), UV-visible absorbance, differential viscometry (VISC), multi-angle light scattering (MALS), and dynamic or quasi-elastic light scattering (DLS/QELS). Initially, only a single concentration-sensitive detector, like RI or UV, was used. The single-detector method needed calibration curves made from reference standards to obtain particle information, but these standards were unreliable. So, a multi-detector approach was introduced that involved coupling multiple detectors with HDC.
- Data system: It records and processes the raw detector signals to obtain particle information, including molar mass, particle shape, size, and structure.
Types of Hydrodynamic Chromatography
HDC can be classified into the following types according to the type of column used:
Open-tubular HDC
In open-tubular HDC, separation occurs in a narrow open or empty capillary tube. This is especially suitable for biomacromolecules. The absence of packing material removes interactions with solid packing materials and reduces stress on the sample. So, it is suitable for the analysis of sensitive macromolecules. Modern miniaturized forms of open-tubular HDC, including microcapillary HDC and nanocapillary HDC, have been useful for DNA analysis.
- Microcapillary HDC uses capillaries with micrometer-scale inner diameters. This is used for bioanalytical applications, especially DNA analysis.
- Nanocapillary HDC systems are smaller than microcapillary systems and use capillaries with sub-micrometer diameters. It is especially useful for the analysis of large DNA molecules.
Packed-column HDC
In packed-column HDC, separation occurs in a column packed with inert and non-porous spherical particles. The packing particles must be chemically inert to minimize the interactions between the sample and the stationary phase. The interstitial spaces between the packing particles act as capillaries, which support laminar flow. Packed-column HDC is used for particles and polymers.
Procedure or Steps of Hydrodynamic Chromatography
1. Sample Preparation
Most samples are not ready for direct analysis and need to be carefully prepared before analysis. Sample preparation makes the sample compatible with the separation and free of interferences. This may include sample collection, storage and preservation, transport, processing, dilution, removal of particulates through filtration, centrifugation, or extraction, and derivatization.
2. Mobile phase and column preparation
A suitable liquid or solvent is prepared to fully dissolve the sample particles and to minimize unwanted interactions. Buffers or surfactants can be used to adjust the ionic strength of the mobile phase and minimize unwanted interactions. The solvent moves the sample through the separation unit. A suitable separation unit is selected based on the sample and separation requirements. It can be either an open capillary tube or a packed column. The capillary or column is flushed and equilibrated with the selected solvent to maintain stable flow conditions.
3. Sample injection
The prepared sample is injected into the capillary or packed column using manual injection loops or autosamplers. Small sample injection volumes are used to avoid band broadening. The sample should also be diluted enough to avoid interactions between the particles. It should also be noted that the solvent used to dissolve the sample is compatible with the mobile phase.
4. Separation
The mobile phase flows through the narrow open channel or the interstitial spaces of the packed column under laminar flow conditions. This creates a parabolic velocity profile needed for separation in which the fluid moves fastest at the center and slowest near the walls. Sample particles are distributed across the flow channels according to their size. Larger particles cannot approach the walls, so they remain in the faster central streamlines and move with higher average velocity. Smaller particles can approach the walls more closely, so they can sample both the fast central streamlines and the slower wall regions. As a result, larger particles elute earlier than smaller particles.
5. Detection
The sample eluting from the column is detected using one or more detectors. The most widely used detector combinations include MALS, QELS/DLS, VISC, and a concentration detector like RI or UV.
- MALS + RI can be used to determine absolute molar mass, molar mass distributions, and radius of gyration.
- QELS + RI can be used to obtain the hydrodynamic radius.
- MALS + VISC + RI can be used to determine the viscometric radius and the structure of the particle.
- MALS + QELS can be used to obtain the shape and compactness of the particle.
6. Data analysis
The detector signals are processed using the data system to convert the signals into particle properties. In single-detector HDC, particle size is obtained from retention data using calibration standards. In multi-detector HDC, properties such as molar mass and other size parameters are obtained directly from the combined detector responses.
Factors Affecting Hydrodynamic Chromatography
- The particle size is the main factor controlling the separation in HDC. It is an important factor that affects the retention and separation process.
- Sample concentration and injection volume are also important factors to consider. Highly concentrated samples can cause unwanted interactions between sample particles, and injecting too high volumes of sample can cause band broadening.
- The diameter of the capillary and the size of the packing materials in the column affect how particles sample the flow field. Smaller diameter or packing particles usually create narrower flow channels, which can increase resolution.
- The mobile phase composition can affect the sample stability and interactions with the packing materials.
- The flow rate is another important factor affecting HDC. The flow must remain laminar. Very low flow rates can cause weak separation, while very high flow rates can increase disturbances and reduce resolution.
Applications of Hydrodynamic Chromatography
- HDC is commonly used for the analysis of colloids, nanoparticles, latex particles, and emulsions. It is also used for the analysis of ultra-high molecular mass polymers.
- It is used in polymer science to analyze synthetic polymers and biopolymers.
- It can be combined with multiple detectors to obtain particle information, including size distribution, molar mass, particle shape, and structure.
- Microcapillary HDC can be used in bioanalysis for the separation and analysis of DNA fragments and nanoparticle systems.
- It has applications in different fields, including biomedical research, the food industry, pharmaceutical development, and environmental monitoring.
Advantages of Hydrodynamic Chromatography
- HDC is based on a simple physical separation principle in which particles are separated purely based on size in solution. The experimental setup is also simple.
- Sample preparation for HDC is also minimal.
- It does not need porous stationary phases and involves minimal interactions between the sample and the stationary phase. This reduces the shear stress and allows gentle analysis of biomolecules.
- It is more suitable for large particles, macromolecules, and ultra-high molar mass polymers.
- It is compatible with multiple advanced detectors, which can be used to determine the size and structural parameters of the sample.
Limitations of Hydrodynamic Chromatography
- HDC has lower separation resolution compared to other size-based separation methods.
- Smaller molecules are poorly separated by HDC.
- HDC separation is sensitive to flow conditions.
- It depends strongly on calibration when single-detector setups are used.
- Reliable information depends highly on advanced detector combinations.
- Columns for HDC are not readily available.
Hydrodynamic Chromatography Troubleshooting and Safety Considerations
- Weak detector signal or no peaks are observed in HDC due to an unsuitable detector, too low sample concentration, or a blocked injector needle. This can be solved by using a compatible detector, proper sample preparation, and checking the injector for blockage.
- Split peaks are caused by an incompatible solvent or poor equilibration of the column or capillary. This can be reduced by preparing the sample in the same solvent as the mobile phase and increasing equilibration time before injections.
- Peak tailing can occur due to particle-surface interactions and damaged or contaminated column packing. This can be resolved by increasing the ionic strength or adding a suitable surfactant and replacing or cleaning the column.
- Broad peaks can occur due to high flow rates and column bed disturbance. This can be resolved by reducing the flow rate and replacing the column if needed.
- An unstable baseline can be caused by air bubbles in the detector flow cells, temperature instability, and contaminated mobile phase. This can be resolved by degassing all mobile phases, maintaining a stable temperature, replacing the mobile phase, and cleaning detector cells when needed.
- All solvents and additives should be carefully handled according to standard chemical safety practices, including the use of gloves and eye protection, preparation of mobile phases in well-ventilated areas, and proper chemical disposal.
- Columns and capillaries should be stored in protective cases to prevent damage. They should also be flushed with a suitable solvent to remove contaminants that could damage the packing.
- Damaged columns and capillaries should be disposed of according to waste disposal policies.
- All laboratory staff should be trained in chromatography system operation and good laboratory practice.
Hydrodynamic Chromatography Recent Advances and Innovations
- Miniaturized and microfluidic HDC systems are emerging, which reduce sample and solvent consumption and also allow faster separation.
- The use of advanced detector combinations in a multi-detector system has allowed the direct measurement and correlation of particle size, molar mass, shape, and structure.
- Improvements in column and channel technology have also improved separation performance. It has allowed more uniform flow fields and better packing materials.
- Microcapillary and nanocapillary HDC systems are widely used for biomolecules, especially DNA. This has expanded the application of HDC to genomic research.
Conclusion
Hydrodynamic chromatography (HDC) is a size-based separation method in which particles of different sizes are separated based on how they sample the parabolic velocity profile of a laminar flow field in a capillary tube or packed column. Larger particles elute before smaller ones. This elution order is the same as SEC, but the separation principle is different.
HDC does not provide high resolution like other separation methods, but it is still useful for particles like colloids, nanoparticles, and macromolecules. Developments in miniaturized and multi-detector systems have increased the importance of HDC in polymer analysis. Modern HDC systems can be used to determine the molar mass, size, shape, and structure of particles, polymers, and biomolecules.
References
- Brewer, A. K. (2021). Hydrodynamic Chromatography: The Underutilized Size-Based Separation Technique. Chromatographia, 84(9), 807–811. https://doi.org/10.1007/s10337-021-04065-4
- DiMarzio, E. A., & Guttman, C. M. (1969). Separation by flow. Journal of Polymer Science Part B: Polymer Letters, 7(4), 267–272. Portico. https://doi.org/10.1002/pol.1969.110070405
- Henderson, T. J. (2025, March 4). Chromatography Column Safety: Essential tips for safe handling and operation. Lab Manager. https://www.labmanager.com/chromatography-column-safety-6748
- Roman, M. (2021). Hydrodynamic chromatography for the characterization of inorganic nanoparticles. In Comprehensive analytical chemistry (pp. 121–171). https://doi.org/10.1016/bs.coac.2021.02.005
- Sample Preparation Techniques | Thermo Fisher Scientific – NP. (n.d.). https://www.thermofisher.com/np/en/home/industrial/chromatography/chromatography-learning-center/chromatography-consumables/sample-prep.html
- Sharma, K. (2024, March 9). Hydrodynamic Chromatography: Definition, principle, and applications. Science Info. https://scienceinfo.com/hydrodynamic-chromatography-definition-principle-and-applications/
- Striegel, A. M. (2011). Hydrodynamic chromatography: packed columns, multiple detectors, and microcapillaries. Analytical and Bioanalytical Chemistry, 402(1), 77–81. https://doi.org/10.1007/s00216-011-5334-3
- Striegel, A. M. (2015). HISTORY OF CHROMATOGRAPHY Hamish Small: experimenter extraordinaire. https://pmc.ncbi.nlm.nih.gov/articles/PMC6605081/
- Striegel, A. M. (2021). Multi-detector hydrodynamic chromatography of colloids: following in Hamish Small’s footsteps. Heliyon, 7(4), e06691. https://doi.org/10.1016/j.heliyon.2021.e06691
- Striegel, A. M., & Brewer, A. K. (2012). Hydrodynamic Chromatography. Annual Review of Analytical Chemistry, 5(1), 15–34. https://doi.org/10.1146/annurev-anchem-062011-143107
- Thompson, J. W., Lieberman, R. A., & Jorgenson, J. W. (2009). Hydrodynamic chromatography for the size classification of micron and sub-micron sized packing materials. Journal of Chromatography A, 1216(45), 7732–7738. https://doi.org/10.1016/j.chroma.2009.08.088
- Wang, Y., Zhou, Y., Zhang, D., Wang, X., & Liu, S. (2021). Extension of hydrodynamic chromatography to DNA fragment sizing and quantitation. Heliyon, 7(9), e07904. https://doi.org/10.1016/j.heliyon.2021.e07904
