Gas chromatography (GC) is a type of chromatography used to separate and analyze volatile compounds. This separation occurs in an instrument called gas chromatograph that uses an inert gas as a mobile phase to move a vaporized sample through a column packed with a stationary phase which causes the components to separate.
GC was developed in the early 1950s by Archer John Porter Martin and Anthony T. James who created the first gas chromatograph system using a gaseous mobile phase and a liquid stationary phase. This method has evolved to also include solid stationary phases. At present, GC systems are widely commercialized and used in different fields.
Principle of Gas Chromatography (GC)
Gas chromatography separates mixtures based on their distribution between the stationary and the mobile phase which is determined by their properties like polarity, molecular size, and boiling point. The stationary phase can be a solid adsorbent or a thin liquid layer coated on the inner wall of the column or on a solid support. The mobile phase is an inert carrier gas that moves the sample through the system.
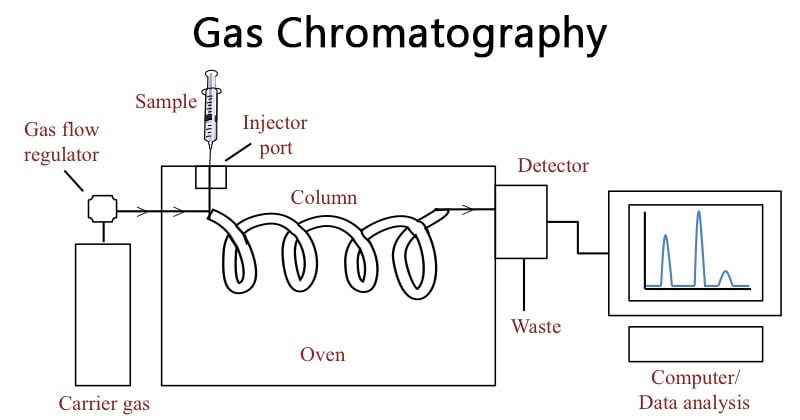
GC works by vaporizing the sample in a heated inlet and carrying it through a column using an inert carrier gas. As the gas moves through the column, the individual components of the sample interact differently with the stationary phase and separate based on their unique physical and chemical properties. The different interactions cause the components to move at different speeds and separate. The separated components are detected by a suitable detector that generates electrical signals. These signals are processed to produce a chromatogram where each peak represents an individual component. This is used to identify compounds by retention time and peak size.
Components of Gas Chromatography (GC)
- GC Inlet: The inlet or injector introduces the sample into the system and transfers it onto the column. Modern GC systems use heated sample ports that rapidly vaporize the sample as it is injected. Split/splitless (SSL) inlet is most commonly used. Other common GC inlets used are direct inlet and on-column inlet.
- Column: It is a long and narrow tube where separation occurs. The column is connected between the sample inlet and the detector. Two common types of columns available for GC are capillary columns and packed columns. Capillary columns are lined internally with a thin layer of polymeric stationary phase while packed columns are filled with solid particles coated with stationary phase. Capillary columns generally give higher resolution than packed columns.
- Column Oven: The column is placed within an oven that maintains the temperature of the column. The column oven can work in two ways: isothermal programming and temperature programming. In isothermal mode, the oven temperature remains constant which is suitable for compounds with similar boiling points. In temperature programming, the temperature is gradually increased which is suitable for samples with different boiling points.
- Stationary Phase: It is the fixed material inside the column that interacts with the sample. It can be solid or liquid coated on an inert support. The stationary phase is selected based on the type of separation. Some of the commonly used stationary phases are polydimethylsiloxane (PDMS), polyethylene glycol (PEG), alumina, and porous polymers.
- Mobile Phase: It is a chemically inert gas that moves the vaporized sample through the column. It is called carrier gas. The gas is stored in a high-pressure cylinder with regulators, flow meters, and pressure gauges. Commonly used carrier gases are helium, hydrogen, nitrogen, and argon. The carrier gas is selected based on the detector used and the properties of the sample. Helium is the most widely used because it is compatible with many detectors and is comparatively safer and efficient.
- Detector: Detectors are used to monitor separated compounds as they elute from the column. Detectors can be categorized into non-selective, selective, and specific based on how they respond to the sample components. Commonly used detectors in GC include mass spectrometer (MS), flame ionization detector (FID), thermal conductivity detector (TCD), and electron capture detector (ECD).
- Data system: It converts the electrical signals from the detector into a chromatogram which is a graph showing peaks for each separated compound. The retention time identifies each component while the peak area or height quantifies its concentration.

Types of Gas Chromatography (GC)
Gas chromatography is classified into two main types based on the physical state of the stationary phase:
1. Gas-Liquid Chromatography (GLC)
In this method, the stationary phase is a liquid coated onto a solid support or inside the column. This is the most widely used form of GC and is used to separate a wide range of volatile and thermally stable organic compounds. In this method, separation occurs due to partitioning of compounds between the two phases.
2. Gas-Solid Chromatography (GSC)
This method uses a solid stationary phase. This method is less common than GLC and is suitable for low molecular weight compounds. It is applicable in analyzing trace impurities in gases. Here, separation occurs due to adsorption onto the active surfaces of the solid material.
Procedure or Steps of Gas Chromatography (GC)
1. Sample Preparation
- This is the first step that makes the sample suitable for analysis.
- Common sample preparation techniques include extraction, dilution, filtration, and derivatization.
- Advanced sample pre-treatment methods like headspace sampling, purge-and-trap, thermal desorption, pyrolysis, and solid-phase microextraction (SPME) are also used.
- Headspace sampling involves sealing the sample in a vial and heating it. The gas in the headspace is injected into the GC.
- Purge-and-trap method purges the volatile analytes from the sample into a sorbent trap which is heated to release them into the GC.
- Thermal desorption collects samples on a sorbent tube and the tube is heated to release trapped volatile compounds into the GC stream without the need for solvents.
- Pyrolysis is used for polymer and macromolecule analysis. The sample is heated rapidly and thermally degraded into smaller volatile fragments.
- SPME is a solvent-free extraction method that uses a coated fiber to adsorb analytes from the sample.
2. Sample Injection and Vaporization
- The prepared sample is injected into the heated GC inlet through a septum using a syringe or an autosampler where it is vaporized.
- Gaseous samples are introduced using a gas tight syringe and liquid samples are injected with a micro-syringe.
- Different injection methods are used depending on the properties of the sample. Some commonly used ones are:
- Split injection introduces only a small fraction of the sample into the column. This is suitable for concentrated samples. It the most widely used injection method. Most of the sample is vented out through the split valve so it prevents column overload.
- Splitless injection transfers the entire sample into the column and is useful for trace analysis. It uses the same inlet as split injection but the split valve is closed for a short period of time to transfer the entire sample.
- Direct injection introduces the whole sample into the injector liner where it vaporizes before entering the column.
- On-column injection introduces the whole sample directly into the column at a low temperature and then gradually vaporizes the sample by controlling temperature of the inlet. This is useful for heat-sensitive compounds.
3. Separation
- The carrier gas moves the vaporized sample through the column.
- Inside the column, sample components interact with the stationary phase and separate based on their properties.
- These interactions cause the compounds to move through the column at different speeds thus separates them.
- Compounds with stronger interactions with the stationary phase are retained longer while those with weak interactions move faster and elute first.
4. Detection and Analysis
- The separated compounds leave the column and enter the detector.
- Detectors produce electrical signals that are processed by a data system to generate chromatogram showing peaks that represent each separated compound.
- Peak position helps identify the compounds and the peak area or height determines the concentration.
- Some commonly used detectors are:
- Flame Ionization Detector (FID) detects the ions produced when analytes are burned in a hydrogen-air flame.
- Thermal Conductivity Detector (TCD) measures changes in thermal conductivity between the carrier gas and the sample.
- Mass Spectrometer (MS) fragments molecules and separates the ions by mass-to-charge ratio.
Factors Affecting Gas Chromatography (GC)
- Sample properties like boiling point and polarity can affect the retention time and the separation process.
- The type of carrier gas and flow rate also affect the separation process. A higher flow rate decreases retention time but can reduce resolution.
- The column temperature is another important factor in GC. Increasing the temperature reduces retention time. However, too high temperature can reduce separation efficiency by reducing interactions with the stationary phase.
- The column dimensions including length and inner diameter are also factors affecting GC. Longer and narrower columns generally provide better separation but may result in increased backpressure and longer analysis time.
- Proper stationary phase selection is also important for improving separation.
- The sample volume is also important. Introducing too much sample can overload the column resulting in distorted peaks and poor separation.
Common Products and Manufacturers of Gas Chromatography
| Common Products | Manufacturers |
| Agilent 8860 GC system, Intuvo 9000 GC system, 990 Micro GC system, Auto sampler systems, Headspace sampler, GC inlets, GC detectors, Packed GC columns | Agilent |
| TRACE 1600 GC, TRACE 1610 GC, AI/AS 1610 Liquid Autosampler, TriPlus 500 GC Headspace Autosampler, TraceGOLD columns, GC accessories like gas filters and injection port liners, iConnect injectors and detectors, Chromeleon software | Thermo Fisher Scientific |
| GC MXT capillary columns, Rtx capillary columns, Packed columns, Inlet supplies, Detector supplies, Gas generators | Restek |
| Clarus GC, GC 2400, TurboMatrix headspace, GC-MS instruments | PerkinElmer |
| Zebron GC columns, Inlet liners, GC syringes, other accessories | Phenomenex |
| Nexis GC-2030, Brevis GC-2050, GC-2010 Pro, GC-2014, SH series GC columns, LabSolutions LC/GC software | Shimadzu |
Applications of Gas Chromatography (GC)
- GC is used in pharmaceutical industry to detect and analyze compounds in drug formulations. This is useful in drug discovery and quality control.
- It is used in food industry for food safety testing and quality control.
- It can be used in environmental monitoring to detect and identify pollutants in environmental samples.
- It is used in forensic science for toxicology screening including blood alcohol testing and drug detection.
- It can also be used to detect performance-enhancing drugs or other prohibitive substances used by athletes in sports. This supports doping control and forensic investigations.
- It can be used to determine the quality and composition of petroleum products like crude oil and natural gas.
Advantages of Gas Chromatography (GC)
- GC can be used to separate complex mixtures with high resolution.
- It can detect compounds at extremely low concentrations using very small sample volumes.
- It allows faster analysis compared to most other chromatographic methods.
- It is applicable to a wide range of volatile compounds which makes it versatile for different applications. Some non-volatile compounds can also be chemically modified through derivatization.
- Different types of stationary phases and detectors are commercially available for GC.
- It can be used with other methods like mass spectrometry which broadens its applications.
Limitations of Gas Chromatography (GC)
- The main limitation of GC is that it is only suitable for thermally stable and volatile compounds that can vaporize without decomposition.
- It is also limited to low to medium molecular weight compounds. While some higher molecular weight compounds can be analyzed, GC is more suitable for compounds less than 1,000 Da. Larger compounds are difficult to vaporize.
- Many samples require derivatization step to modify the compound and make it suitable for analysis. This is a complex and time-consuming sample preparation step.
- Strict safety protocols are required due to the use of high-pressure gas cylinders and highly flammable gases like hydrogen.
- It requires precise control of temperature and carrier gas flow rate. Regular maintenance and monitoring are necessary.
Troubleshooting and Safety Considerations
- Gas leaks can change carrier gas flow, reduce separation efficiency, and waste gases. Regular leak checks, proper installation of parts, and using high-purity gases help avoid these issues.
- Baseline instability occurs due to column bleed, system contamination, or detector instability. This can be resolved by replacing the column, cleaning or replacing the detector components, and using a stable carrier gas.
- Peak tailing can occur due to dirt buildup in inlet liner, active sites in the system, damaged column ends, or overloading. This can be resolved by replacing the inlet liner and column, derivatizing active compounds, or reducing the sample concentration.
- Poor compound resolution or overlapping peaks are caused by using unsuitable column or sample overloading. This can be fixed by using appropriate stationary phase or column dimensions and reducing injection volume.
- Signal spikes or baseline noise can be caused by loose electrical connections or contaminated detectors. All connections must be secured and detector components must be cleaned regularly to fix this.
- When using flammable carrier gases like hydrogen, proper ventilation and leak detection systems are required.
- Laboratory safety protocols should be followed to prevent fire or explosion hazards.
- Samples and solvents can be toxic so they should be carefully handled.
Recent Advances and Innovations of Gas Chromatography (GC)
- Innovations in column technology include improved coatings, advanced packing materials, and development of new stationary phases like porous polymers and monolithic columns.
- Advanced sample preparation methods like headspace analysis and thermal desorption allow the analysis of a wide range of sample types.
- Miniaturized GC instruments offer portability and rapid analysis. This is also useful for reducing sample and solvent consumption.
- Multidimensional GC methods enhance separation of complex mixtures.
- Sustainable practices are being adopted that reduce environmental impact and solvent use. For example, helium is often replaced with greener and more cost-effective alternatives such as hydrogen.
- GC systems are integrated with advanced detectors like mass spectrometer, infrared spectrometer, and electron capture detectors.
- Advanced software tools for data analysis improve peak detection and compound identification.
Conclusion
Gas chromatography is an important separation method for the analysis of volatile compounds. It has wide applications in pharmaceuticals, environmental monitoring, forensics, and quality control. From its development in the 1950s to its widespread use at present, GC has advanced in instrumentation, column technology, and detection systems.
References
- Acevedo, A. (2024, December 26). A Year in Review: Key Trends in Gas Chromatography for 2024. Chromatography Online. Retrieved from https://www.chromatographyonline.com
- Choosing the right GC Injection Technique. (n.d.). Retrieved from https://community.agilent.com/technical/consumables/w/wiki/4972/choosing-the-right-gc-injection-technique
- Drawell. (2023, September 20). 7 Common Problems of Gas chromatography (GC) and How to Troubleshoot – Drawell. Retrieved from https://www.drawellanalytical.com/what-are-the-7-common-problems-of-gas-chromatography-gc-and-how-to-troubleshoot/
- Drawell. (2025, July 7). GC sample preparation – Techniques and challenges – Drawell. Retrieved from https://www.drawellanalytical.com/gc-sample-preparation-techniques-and-challenges/
- Gas Chromatography Fundamentals | Agilent. (n.d.). Retrieved from https://www.agilent.com/en/product/gas-chromatography/what-is-gas-chromatography
- Gas Chromatography: Definition, Purpose, Detection, & Uses. (2024, February 27). Retrieved from https://www.chemistrylearner.com/chromatography/gas-chromatography
- Helmenstine, A. M. (2025, June 9). Gas Chromatography – what it is and how it works. Retrieved from https://www.thoughtco.com/gas-chromatography-4138098
- Isothermal vs. Temperature Programming – What’s the Difference? | This vs. That. (n.d.). Retrieved from https://thisvsthat.io/isothermal-vs-temperature-programming
- Jones, K. (2022, August 4). Trends and Developments in Gas Chromatography. Chromatography Online. Retrieved from https://www.chromatographyonline.com
- Jwaili, M. (2019). Pharmaceutical applications of gas chromatography. Open Journal of Applied Sciences, 09(09), 683–690. https://doi.org/10.4236/ojapps.2019.99055
- Patrick, G. (2023, November 2). Top 7 gas chromatography manufacturers easily separating highly complex chemicals. Retrieved from https://www.verifiedmarketresearch.com/blog/top-gas-chromatography-manufacturers/
- PerkinElmer Blog. (2023, November 20). How does gas chromatography work? | PerkinElmer blog. Retrieved from https://blog.perkinelmer.com/posts/gas-chromatography-explained-what-it-is-and-how-it-works/
- Turner, D. (2024, March 12). Gas Chromatography – How a gas chromatography machine works, how to read a chromatograph and GCXGC. Retrieved from https://www.technologynetworks.com/analysis/articles/gas-chromatography-how-a-gas-chromatography-machine-works-how-to-read-a-chromatograph-and-gcxgc-335168

Wow this has really helped me for the assignment. i really appreciate, looking forward to more and more updates. God bless you
actually was stuck but when I landed here my my course work was solved in minutes
THANKS PROFESSOR
Your notes are so helpful dear.
Thanx.
THANK U SIR , Students who learn gas chromtography have very clearly remembered that very usefull
Wow wonderful it’s the information am looking for very helpful.
Thanks alot.
This has helped my Work tremendously. And will be looking forward for your assistance, Please.
*My Institution- University of Mkar Benue State Nigeria, procured a New Copy of a Gas Chromatography System and Infrared (IR) System, about 10 years ago nd is not been installed.
Please, can you help me with any of your Technician Specialist that can install the two of them for us?
Thank You
My email is as appeared in my address.
Really helpful
Thanks for sir very most important information
The note was very helpful for my assignment , looking forward for more information on separation analysis techniques.
Thank you.
Thanks for being organised Dr.
Very nicely presented in brief…very easy for a student to understand
Awesome description about GC analysis method…
Really helpfull
Thank you sir, valuable information about GC…
“Higher working temperatures up to 5000C”
this is indeed a high working temperature, but should this be rather 500 °C (still high, though)?
Awesome very organised answer