Fast Protein Liquid Chromatography (FPLC) is a liquid chromatography technique used for the separation and purification of proteins and other biomolecules.
It was developed in the 1980s by Pharmacia and was initially known as fast performance liquid chromatography.
FPLC works under medium pressure which allows fast flow rates and high sample loading capacities while maintaining gentle conditions. This helps preserve the structure and function of sensitive biological molecules. Unlike High-Performance Liquid Chromatography (HPLC) which is mainly used for small molecules and runs at high pressures, FPLC is ideal for purifying large and fragile biomolecules like proteins. So, FPLC is suitable for applications that require both high resolution and gentle purification conditions.
Principle of Fast Protein Liquid Chromatography
FPLC works on the principle of using medium pressure to separate biomolecules based on their properties such as size, charge, hydrophobicity, and binding affinity to specific ligands. FPLC is usually done in multiple stages so different separation methods are used in this process.
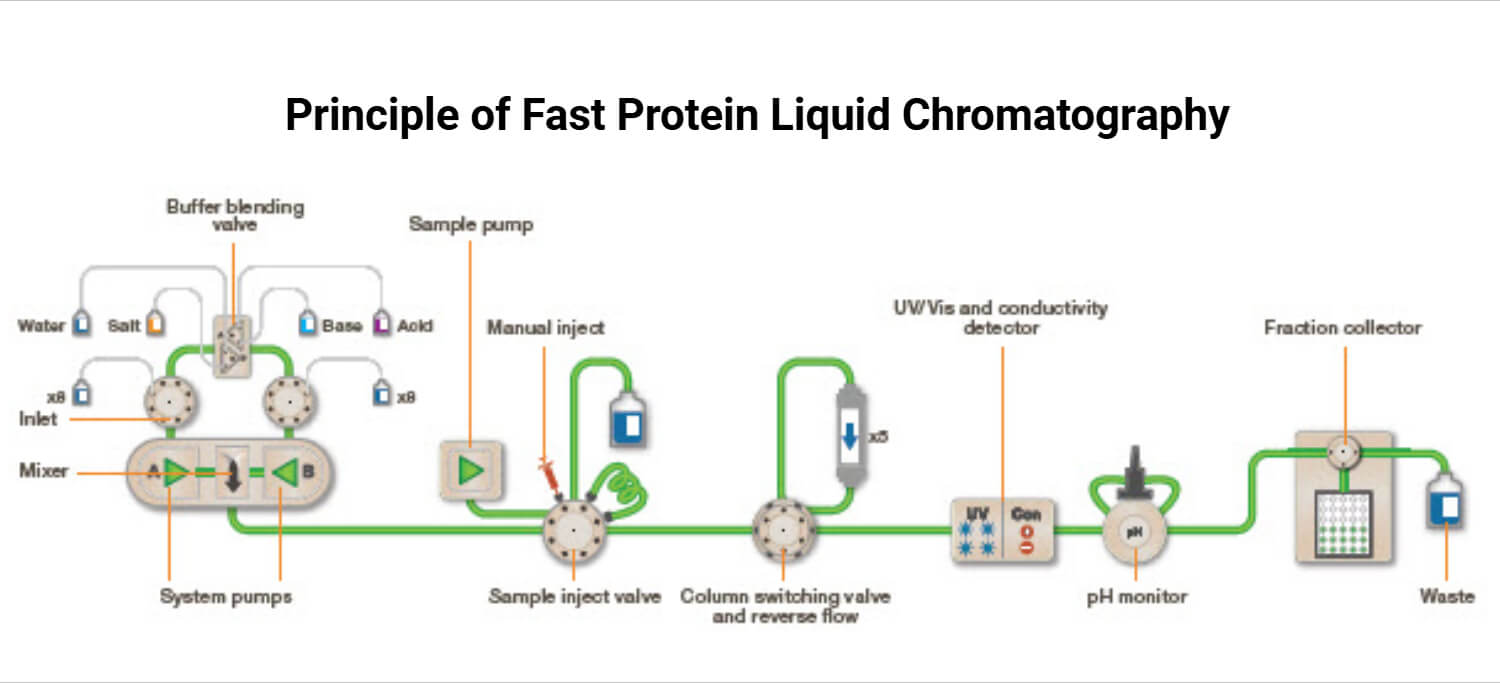
The process begins by loading a sample mixture into a column packed with porous resin beads that act as stationary phase. An aqueous buffer solution is used as mobile phase that flows through the column at a controlled flow rate. As the sample flows through the column, different components interact with the stationary phase based on their properties. Unbound molecules are washed away, while the target molecules bound to the resins are released by changing the buffer conditions. Detectors like UV absorbance, conductivity, and pH sensors are used to monitor the elution in real time. The eluted fractions are collected in individual tubes for further use or analysis.
Components of Fast Protein Liquid Chromatography
- Chromatography Column: It is usually a vertical cylinder made of glass or plastic where the separation process occurs. The column is packed with stationary phase and allows the flow of buffer and sample through it.
- Stationary Phase: It consists of small, porous beads usually made from cross-linked agarose or other polymeric resins that is packed inside the column. Different types of resins are used depending on the separation method.
- Mobile Phase: FPLC systems use aqueous buffer solution as the mobile phase which carries the sample through the column. It allows the interaction between the sample molecules and the stationary phase.
- Pumps: The pump delivers the buffer system through the column at a controlled flow rate. Modern FPLC pumps allow mixing different buffer solutions for gradient elution. FPLC systems usually operate at medium pressures which is gentle but powerful enough for efficient separation.
- Sample Injection System: This introduces the sample into the column. FPLC systems involve two methods for sample loading: manual injection and automated injection. The sample can be injected manually using a sample loop. Alternatively, a sample pump or autosampler can automatically load them onto the column.
- Detectors: In FPLC, detectors are used to monitor the elution of target molecules as they pass through the column. Different types of detectors can be used based on the properties of the target compounds. FPLC systems can use detectors including UV detectors, diode array detectors (DAD), conductivity and pH monitors, fluorescence detectors, and refractive index detectors (RID).
- Fraction Collector: It is used to collect eluted fractions automatically into separate tubes for further analysis. It can work based on time, volume, or detector signal.
- Column Switching Valves: This is one of the additional features of modern FPLC systems that allows the user to switch between different columns automatically. This is useful for running different purification steps without disconnecting equipment manually. This saves time and manual work.
- Buffer Blending Valves: These valves mix two or more buffer solutions in real time to create gradients that is useful for changing conditions like salt concentration or pH. This is used for controlling binding and elution conditions.
- Software Interface: The software controls and monitors the entire FPLC system. Modern FPLC systems include user-friendly software interfaces that provide control over every step of the process including setting up purification methods, designing gradients, controlling hardware, and analyzing data. It also supports multiple system configurations that allows control of more than one chromatography system within the same software platform.
Types of Fast Protein Liquid Chromatography
FPLC supports different chromatographic methods. Some of the different chromatography modes of FPLC are:
- Ion Exchange Chromatography (IEC) separates biomolecules based on their charge. Molecules bind to oppositely charged resins and the bound molecules are eluted by increasing salt concentration or adjusting the pH. There are two types of resins used in IEC: cation exchange and anion exchange. Cation exchange resins bind positively charged proteins while anion exchange binds negatively charged proteins.
- Size Exclusion Chromatography (SEC) or Gel Filtration separates molecules by size. The resins used in this method have pores of defined sizes. Larger molecules cannot enter the pores so they elute first. Smaller ones can enter the pores and they elute later. SEC uses porous resins like agarose, dextran, or polyacrylamide.
- Affinity Chromatography uses specific binding interactions between the target molecule and ligand immobilized on the column matrix to selectively bind and purify the target. Example: antibody-antigen or enzyme-substrate interactions. It uses resins like Protein A, Ni-NTA or resins with specific ligands.
- Hydrophobic Interaction Chromatography (HIC) separates molecules based on their hydrophobic regions. It uses resins coated with hydrophobic ligands. Proteins bind in high salt conditions and elute as salt concentration decreases. It is a good method for preserving native structure compared to reverse-phase methods.
- Reverse Phase Chromatography (RPC) separates molecules using strong hydrophobic interactions with a non-polar stationary phase. It uses hydrophobic silica or polymer-based resins. It provides higher resolution than HIC but denatures proteins so it is typically used for peptides, small proteins, and organic molecules.

Procedure or Steps of Fast Protein Liquid Chromatography
- Sample Preparation: The sample solution is first clarified using methods like filtration or centrifugation to remove particulates. The sample buffer should be compatible with the column equilibration buffer.
- Column Preparation: A suitable column is selected based on the method of separation. The column is washed with starting buffer to stabilize pH and ionic strength. Suitable running and elution buffers are also selected depending on the method.
- Sample Loading: The sample is introduced onto the column either manually using syringes and sample loop or automatically using a sample pump or autosampler. Overloading the column should be avoided to prevent low resolution.
- Elution: After the sample is applied, unbound sample molecules are washed away with buffer while bound molecules are eluted by using buffer gradients. Elution occurs based on charge, size, or affinity of the target molecule based on the type of column used.
- Detection: The elution is monitored in real time using detectors that generate chromatograms with peaks representing different eluted compounds. UV absorbance and conductivity detectors are the most common in FPLC. UV detectors measure absorbance at 280 nm to detect proteins while conductivity meters monitor changes in salt concentration during elution. For compounds that do not absorb UV light, other methods like fluorescence and refractive index detectors are used.
- Fraction Collection: The eluted fractions corresponding to detected peaks are collected for further purification and analysis. Fraction collection can be done using three ways: manual, volume-based, and peak-based. Manual collection is done by the operator directly and is suitable for small-scale experiments. Volume-based collection is done based on fixed pre-set volumes. Peak-based collection is automatically done using the detector signal.
- Data Analysis: The chromatogram peaks are analyzed to evaluate the purity and concentration of the separated molecules. The collected fractions can be analyzed using additional methods like SDS-PAGE, Western blot, and spectrophotometry to confirm protein identity and purity. Different software tools can help enhance data analysis by improving peak detection and data interpretation.
Stages of Fast Protein Liquid Chromatography
FPLC purification is done in multiple stages to obtain high purity of target molecules.
- Capture step: This is the initial purification step used to isolate and purify the target molecule from the crude mixture. Affinity chromatography is commonly used in this step as it provides high selectivity and can quickly separate the molecule of interest.
- Intermediate purification step: Once the target protein is captured, further purification is done to remove other similar or partially bound contaminants. Ion-exchange and hydrophobic interaction chromatography are often used at this stage.
- Polishing step: This is the final step that removes trace or remaining impurities. Size exclusion chromatography is used in this step.
Factors Affecting Fast Protein Liquid Chromatography
- The pH of the buffer can affect the solubility of target molecules and their interactions with the stationary phase.
- Ionic strength or salt concentration affects the binding and elution especially in ion exchange and hydrophobic interaction chromatography.
- The pore size and type of stationary phase used can determine how molecules are separated.
- Column dimensions including length, diameter, and packing can affect the resolution and flow rate.
- The sample volume is also a factor affecting the process. Overloading the column can cause peak broadening and reduce separation efficiency.
- Temperature can affect protein stability and interaction with the stationary phase. High temperatures can denature proteins and affect separation.
Common Products and Manufacturers of Fast Protein Liquid Chromatography
| Common Products | Manufacturers |
| AKTA series, HiTrap, Superdex, Sepharose columns, UNICORN software | Cytiva |
| NGC chromatography system, UNOsphere, Bio-scale, Econo-Column | Bio-Rad Laboratories |
| Azura FPLC system, Sepapure FPLC columns, PurityChrom software | Knauer |
| Pierce chromatography cartridges, Ni-NTA, Protein A, Protein G | Thermo Fisher Scientific |
Applications of Fast Protein Liquid Chromatography
- FPLC is commonly used to isolate and purify specific proteins and other biomolecules from complex mixtures. This is useful for both research and therapeutic applications.
- It helps to study the properties and interactions of proteins, which is useful in understanding their functions and roles in different biological processes.
- It has applications in industrial production of proteins, enzymes, and other valuable biomolecules.
- It can also be used in the purification of recombinant proteins, monoclonal antibodies, and therapeutic enzymes in biotechnology and pharmaceutical industries.
- It has applications in quality control to monitor the purity of protein-based pharmaceuticals.
Advantages of Fast Protein Liquid Chromatography
- FPLC works on gentle conditions, which help to preserve the structure and function of sensitive target molecules.
- FPLC is scalable for both small-scale research applications and large-scale protein purification.
- It is compatible with different chromatographic methods.
- Although FPLC runs at lower pressures than HPLC, it maintains high flow rates suitable for fast and efficient separation.
- Modern FPLC systems are highly automated and include features such as in-line detectors, fraction collectors, and software-controlled gradients.
- FPLC system allows the use of multiple columns, detectors, and fraction collectors.
Limitations of Fast Protein Liquid Chromatography
- FPLC works at lower pressure than HPLC which can limit the separation speed and resolution.
- Many proteins require multiple stages for complete purification which increases complexity and time.
- The setup and maintenance of FPLC equipment can be expensive.
- Columns used in this method are usually made of glass or plastic and cannot tolerate high pressures compared to HPLC.
- Standard FPLC systems are less suitable for separating small organic molecules or compounds that require organic solvent.
Troubleshooting and Safety Considerations
- A clogged column or filter, narrow tubing, or highly viscous samples and buffers can cause high system pressure. This can be fixed by cleaning the system, replacing the filters, using appropriate tubing, and diluting or filtering the sample before use.
- Air bubbles in the buffer can cause pressure fluctuation which can be fixed by degassing the buffer.
- If the flow cell is dirty or has air trapped inside, it can cause fluctuating or no UV signal. So, the flow cell should be cleaned according to the manual and a back pressure regulator can be used to keep the flow stable.
- Long or wide tubing can cause sample diffusion which leads to peak broadening. Shorter and narrower tubing can be used to minimize this but very narrow tubing can again increase the system pressure so proper tubing diameter should be used.
- Routine maintenance including regular cleaning, performance tests, buffer and sample filtration should be done to ensure consistent performance.
- Buffers may contain hazardous chemicals so it is important to wear protective equipment.
- FPLC often involves biological samples so proper biosafety practices must be followed to avoid exposure and contamination.
- The system setup should be stable before running. Loose fittings, leaks, pressure, and UV signal should be properly checked to avoid spills or equipment damage.
- Safety tools including air sensors, back pressure regulators, and filters should be used to prevent accidents.
Recent Advances and Innovations
- Development of advanced UV detectors using nanomaterials and optical fibers enhances speed and accuracy.
- Modern FPLC systems support multi-wavelength UV detectors that can simultaneously monitor sample elution at different wavelengths. It also supports real-time monitoring of elution.
- Miniaturized FPLC systems are being developed that help reduce sample and buffer volume requirements.
- Newer prepacked columns provide higher binding capacities and faster flow rates.
- Recent developments in automated gradient control and autosampling improves the precision of FPLC.
- Multi-column systems have allowed the integration of two or more purification steps which reduces overall processing time and improves the efficiency of the process.
Conclusion
Fast protein liquid chromatography is widely used for the purification of large biomolecules especially proteins. It works under gentle conditions which helps preserve the structure and function of the target molecule. It is also a versatile method that is compatible with different chromatography methods which makes it suitable for different applications including research, diagnostics, and industrial applications.
References
- ConductScience. (2024, July 25). Fast protein liquid chromatography (FPLC) Protocol. Retrieved from https://conductscience.com/fast-protein-liquid-chromatography-fplc-protocol/
- Fast protein liquid chromatography (FPLC) for protein purification. (2025, May 5). Retrieved from https://www.abcam.com/en-us/knowledge-center/proteins-and-protein-analysis/fplc-protein-purification
- Fast Protein Liquid Chromatography. (n.d.). Retrieved from https://www.bio-rad.com/en-np/applications-technologies/fast-protein-liquid-chromatography?ID=MWHBF4CZF
- FPLC Systems – Bio Purification Solutions. (n.d.). Retrieved from https://www.knauer.net/fplc
- Madadlou, A., O’Sullivan, S., & Sheehan, D. (2011). Fast Protein Liquid Chromatography. Methods in Molecular Biology, 439–447. https://doi.org/10.1007/978-1-60761-913-0_25
- Runde, S. (2016, August 22). FPLC versus Analytical HPLC: Two Methods, One Origin, Many Differences. Chromatography Online. Retrieved from https://www.chromatographyonline.com
