Countercurrent chromatography (CCC) is a liquid chromatography (LC) method that separates chemical compounds in a mixture based on their differential solubility between two immiscible liquid phases in a solid-support-free system.
Countercurrent chromatography (CCC) is also called liquid-liquid chromatography (LLC) or liquid partition chromatography as both stationary and mobile phases are liquids. Instead of using a solid support to hold the stationary phase like in traditional chromatographic systems, the stationary phase is held in place by gravity or centrifugal force in this method.
Although the name countercurrent suggests the two liquid phases moving in opposite directions, most CCC instruments involve only one moving phase. So, there is no true countercurrent flow which makes the term technically inaccurate. However, the term is still acceptable as it is widely used and well-established in scientific literature.
- The basic concept of CCC comes from the countercurrent distribution method developed by Craig and Post in the 1940s that used gravity to separate compounds in connected tubes.
- The first true CCC system was introduced in 1966 by Dr. Yoichiro Ito and his team. This was the start of the modern CCC system which used centrifugal force to hold the stationary phase.
- In the early 1970s, Tanimura and team introduced Droplet CCC (DCCC) which is a gravity-based method that improved on earlier designs but phase retention was weak and separation took days.
- Later in the 1970s, Dr. Ito developed High-Speed CCC (HSCCC) that could complete separations in a few hours by using helical coils rotating at high speeds.
- In the 1980s, the Japanese company Sanki introduced centrifugal partition chromatography (CPC) which is a hydrostatic system with radial chambers and stronger centrifugal force.
- Newer CCC systems were introduced in the 2000s including high-performance CCC which resulted in faster and more stable separations.
Principle of Countercurrent Chromatography
Countercurrent Chromatography works on the principle of partitioning of compounds between two immiscible liquid phases based on their differential solubility. The liquidstationary phase is retained in the column by centrifugal force generated by rotating the column and the mobile phase is pumped through the column along with the sample. The centrifugal force pushes the stationary phase away from the center helping to hold them in place against the flow of the mobile phase. When the liquid mobile phase flows through the column, the compounds in the sample distribute between the two liquids based on their solubility or partition coefficient (K):
Partition coefficient (K) = Amount of solute in stationary phase / Amount of solute in mobile phase
Each compound has a unique solubility in each liquid phase and moves at different speeds based on how much they prefer one liquid over the other. This differential solubility results in separation of the compounds. These separated compounds are collected and analyzed.
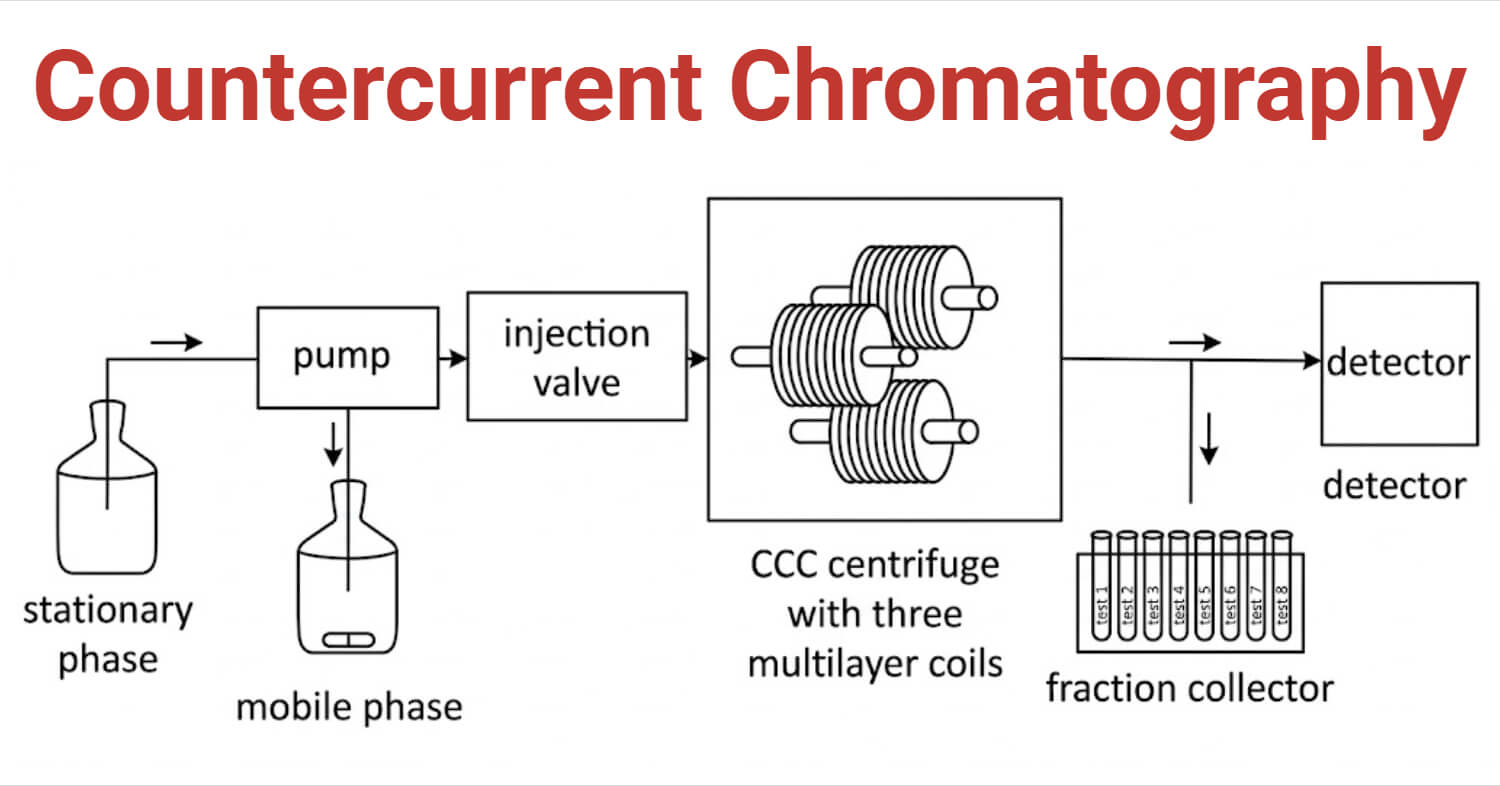
Components of Countercurrent Chromatography
1. Column
The Countercurrent chromatography column contains a coiled tube or a series of connected chambers arranged in different geometries depending on the specific CCC type. All modern CCC columns use rotating parts to create centrifugal field that stabilizes the stationary phase. It includes rotors or spools for holding the coiled tubing, motors and speed controllers to spin the system at controlled speeds, rotating seals and gears to allow continuous flow during rotation, cooling units, and other technical parts. The column is usually made of materials like Teflon or PTFE and has no solid support.
2. Solvent/ Liquid Phases
There are two immiscible liquid phases in Countercurrent chromatography. One is the stationary phase which is retained in the column by centrifugal force and the other is the mobile phase which flows through the column. Common solvent systems used in this method include aqueous-organic mixtures like chloroform/methanol/water (ChMWat) and n-hexane/ethyl acetate/methanol/water (HEMWat).
3. Pumps
They are used to deliver the mobile phase to the column. They introduce and maintain flow of the mobile phase at a controlled flow rate.
4. Sample Injectors
They introduce the sample into the column. They include injection valve and sample loop or port.
5. Detectors
They are used to monitor the results of the separation. Detectors like UV-Visible absorbance detectors, fluorescence detectors, Evaporative light-scattering detectors (ELSD), nuclear magnetic resonance (NMR) spectrometers, and mass spectrometry (MS) are commonly used.
6. Fraction Collectors
They are used to collect fractions of separated components as they elute from the column for further use or analysis.
Types of Countercurrent Chromatography
Countercurrent chromatography systems can be classified into the following types based on how the stationary phase is retained in the column:
A. Gravity-based CCC
This system uses gravity to hold the stationary phase. It is slower than modern centrifugal systems. Example of this type is droplet CCC (DCCC) which uses gravity to move droplets of mobile phase through a column filled with the stationary phase
B. Centrifugal-based CCC
It uses centrifugal force to hold the stationary liquid in place. Most CCC systems in use at present are centrifugal-based. There are two types of this system:
1. Hydrodynamic System
- This system rotates the column in a planetary motion to generate varying centrifugal force which helps retain the stationary phase in the column.
- The column is made of coiled tubing and rotates both around its own axis and the central axis of the centrifuge at the same speed and in the same direction. This dual rotation helps stabilize the stationary phase within the coils.
- Examples include High-Speed Countercurrent Chromatography (HSCCC), slow rotatory CCC, and Nonsynchronous CCC.
2. Hydrostatic System
- This system uses a single-axis rotation to create a constant centrifugal field. There is no planetary motion and it has simpler mechanics than hydrodynamic systems.
- The column contains a series of interconnected chambers arranged in a spinning rotor. Each chamber has an outlet and inlet to allow the mobile phase to enter and exit.
- This system also includes rotary seals on both ends and are designed to maintain stable phase distribution without coiled tubing.
- It is also called centrifugal partition chromatography (CPC).
Procedure or Steps of Countercurrent Chromatography
- Solvent/Phase Selection: A biphasic solvent system containing two immiscible liquids that acts as the mobile and stationary phases are carefully selected. Solvent systems are usually grouped into set of mixtures containing 3-5 solvents. Most widely used solvent systems include HEMWat, ChMWat, and ARIZONA system (heptane/ethyl acetate/methanol/water). The most commonly used solvents in CCC are n-hexane, ethyl acetate, methanol, and water.
- Sample Preparation: The next step is sample preparation. Proper sample preparation is important for improving resolution and getting accurate results. The sample is prepared by dissolving in a small volume of the mobile phase or the biphasic solvent system. It is then filtered to remove any particulates that could block the tubing or valves. For complex mixtures, additional pre-treatment may be necessary.
- Column Setup: The CCC column is usually a coiled tube or interconnected chambers. It is filled with the stationary phase and rotated to generate centrifugal force which retains the stationary phase in the column and prevents it from being washed away by the mobile phase.
- Sample Injection: The prepared sample is introduced into the rotating column containing the stationary phase through an inlet or sample loop. The sample volume injected can vary depending on the column size and the purpose of separation. Overloading the column with sample should be avoided as it can reduce resolution.
- Separation: After sample injection, the separation process begins. The mobile phase is introduced into the rotating column at a controlled flow rate using pumps. As the mobile phase flows through the stationary phase, it carries the sample components which partition between the two liquid phases based on their chemical properties. Compounds with higher solubility in the mobile phase move more quickly and are eluted faster than compounds with higher affinity for the stationary phase. This differential partitioning separates the compounds.
- Detection and Collection: After separation, the eluted compounds are detected and collected in fractions for further analysis. Detectors like UV, MS, ELSD, NMR or fluorescence are used to monitor the elution. This generates chromatograms showing peaks corresponding to different compounds as they elute. The separated fractions can be further analyzed using methods like TLC or spectroscopy to identify the collected compounds.
Factors Affecting Countercurrent Chromatography
- The solvent system is an important factor affecting the separation process. Both stationary phase and mobile phase should be carefully selected. Properties of the solvents like solvent polarity, viscosity, and solubility can affect separation.
- The column type and design like internal geometry and volume can also affect the separation process.
- The rotation speed and type of motion can affect how well the stationary phase is retained. Too low speed can cause poor retention and too high speed can cause high pressure or noise. This must be optimized for better separation.
- The flow rate of the mobile phase can also affect the separation resolution and stationary phase retention.
- Sample properties like solubility, chemical structure, and interactions with phases also influences the process.
- Temperature can affect properties like solubility, density, viscosity, and partition coefficients.
Common Products and Manufacturers of Countercurrent Chromatography
| Common Products | Manufacturers |
| Quattro CPC and CCC systems | AECS-QuikPrep Ltd. |
| FCPC (Fast Centrifugal Partition Chromatography) series, Peripheral Equipment (pumps, detectors, fraction collectors, and autosamplers) | Kromaton |
| CPC 250, CPC 1000 Pro, Gilson Glider software | Gilson (Armen) |
| TBE series HSCCC (20A, 300B/C, 1000A, 5000A) | Tauto Biotech |
| Pharma-Tech CCC-1000 | Pharma-Tech |
| Spectrum CCC System | Dynamic Extractions |
| CPC Modeler, iCPC, rCPC, pCPC, Continuous CPC | RotaChrom Technologies |
Applications of Countercurrent Chromatography
- Countercurrent Chromatography is commonly used for isolating natural products like phytochemicals from herbs and medicinal plants. Different plant-derived compounds like alkaloids, flavonoids, and essential oils can be separated using this method.
- One notable application of this method is the extraction and purification of cannabinoids from cannabis.
- It has pharmaceutical applications to purify active drug ingredients and isolate bioactive molecules. This is important in drug development and biochemical research.
- CCC is suitable for separating large biomolecules like proteins, DNA, and other macromolecules while preserving their biological activity.
- It can also be used to separate and analyze contaminants, flavors, additives, and natural pigments in food. This helps in quality control and safety testing in the food industry.
- This method is also used in the agriculture industry to purify herbicides and pesticides which ensures chemical purity.
Advantages of Countercurrent Chromatography
- Countercurrent Chromatography does not use a solid support which avoids problems like irreversible adsorption, sample loss, low sample recovery, and denaturation of sensitive compounds. This is useful for purifying sensitive or complex compounds.
- This method is cost-effective as it removes the need for expensive solid columns. The initial cost of instrument can be high but it is a one-time investment so the only main running cost is the solvent. This reduces long-term operational expenses.
- CCC columns are usually made from materials like Teflon which is much more chemically resistant and can tolerate a wide pH range better than silica-based HPLC columns.
- Since there are no harsh interactions with solid surfaces, it is a gentle method that preserves the original properties and biological activity of compounds.
- It has a high sample loading capacity which is useful for preparative separations.
Limitations of Countercurrent Chromatography
- Countercurrent Chromatography is less suitable for very small sample volumes.
- It can be slower than other chromatography methods as it takes longer run times.
- It requires careful solvent selection and specialized equipment like rotating coil devices.
- Solvent system optimization is complex and time-consuming. It is difficult to choose the right two-phase system in CCC and optimization is needed for each new sample or compound.
- CCC instruments need precise sealing systems because the spinning columns under pressure can leak.
Troubleshooting and Safety Considerations
- Using an inappropriate solvent system or rotor speed can result in poor retention of the stationary phase. This can be resolved by increasing the rotor speed and optimizing the solvent composition.
- When the selected biphasic solvent system contains incompatible components, emulsification or phase mixing can occur.
- Any blockages in tubing or filters must be cleaned and the pump system must be regularly maintained to avoid irregular flow.
- Personal protective equipment (PPE) must be worn to avoid exposure to harmful chemicals and potential hazards.
- Pressurized systems must be carefully monitored to prevent leaks.
- Electrical components like power supplies must be checked regularly to maintain electricals safety due to the use of high-speed motors and detectors.
- The rotor chamber must remain closed during operation and should not be opened or touched while the instrument is running as the high-speed rotation can result in mechanical hazards.
Recent Advances and Innovations
- Modern Countercurrent Chromatography systems have enhanced software, faster rotors, chamber designs, can handle higher pressures, and better automation for faster and easier operation.
- New operating modes like dual-flow, pH-zone refining, and ion-exchange CCC allow separation of ionizable or complex compounds and improves the sample recovery.
- Green solvent systems are being developed to reduce toxicity and make separations more environmentally friendly.
- CCC can be integrated with analytical techniques like MS, NMR, and HPLC for faster and precise identification of separated compounds.
- Multidimensional CCC setups are also being developed to improve separation of complex mixtures.
- Miniaturized CCC systems are emerging that reduce solvent usage and handle small sample volumes.
- High-speed and high-performance CCC instruments generate much stronger centrifugal force which allows faster separations with improved resolution.
Conclusion
Countercurrent Chromatography is a liquid-liquid separation method that uses centrifugal force to retain the liquid stationary phase without the need for any solid support. This allows for a gentle and effective separation of a wide range of compounds based on how they partition between two immiscible liquid phases. CCC does not actually involve two liquids flowing in opposite directions as the name suggests. Instead, centrifugal force is used to keep one liquid phase in place while another liquid moves through it.
However, advancements in operational modes have led to development of methods like dual-flow CCC in which both liquid phases are actively pumped in opposite directions. CCC is widely used for applications like natural product isolation, pharmaceutical purification, and biochemical research.
References
- Berthod, A. (2017). Separation and Purification with a Liquid Stationary Phase. Separations, 4(4), 30. https://doi.org/10.3390/separations4040030
- Berthod, A., & Faure, K. (2015). Separations with a Liquid Stationary Phase: Countercurrent Chromatography or Centrifugal Partition Chromatography. Analytical Separation Science, 1177–1206. https://doi.org/10.1002/9783527678129.assep046
- Berthod, A., Maryutina, T., Spivakov, B., Shpigun, O., & Sutherland, I. A. (2009). Countercurrent chromatography in analytical chemistry (IUPAC Technical Report). Pure and Applied Chemistry, 81(2), 355–387. https://doi.org/10.1351/pac-rep-08-06-05
- Chaudhary, A. (2024, November 13). Counter current chromatography. Retrieved from https://veeprho.com/counter-current-chromatography/
- Commercially available countercurrent separation (CCS) instruments. (2016, December 5). [Slide show]. Retrieved from https://www.slideshare.net/slideshow/commercially-available-countercurrent-separation-ccs-instruments/69845662
- Countercurrent Chromatography – Unacademy. (2022, February 26). Retrieved from https://unacademy.com/content/kerala-psc/study-material/bioinstrumentation/countercurrent-chromatography/
- Factors affecting chromatographic separation | Solubility of things. (n.d.). Retrieved from https://www.solubilityofthings.com/factors-affecting-chromatographic-separation
- Hopmann, E., Arlt, W., & Minceva, M. (2010). Solvent system selection in counter-current chromatography using conductor-like screening model for real solvents. Journal of Chromatography A, 1218(2), 242–250. https://doi.org/10.1016/j.chroma.2010.11.018
- Kostanyan, A. A., Voshkin, A. A., & Belova, V. V. (2020). Analytical, Preparative, and Industrial-Scale separation of substances by methods of countercurrent Liquid-Liquid chromatography. Molecules, 25(24), 6020. https://doi.org/10.3390/molecules25246020
- Marston, A., & Hostettmann, K. (2022, April 15). Modern countercurrent chromatography. Chromatography Online. Retrieved from https://www.chromatographyonline.com
- Michel, T., Destandau, E., & Elfakir, C. (2013). New advances in countercurrent chromatography and centrifugal partition chromatography: focus on coupling strategy. Analytical and Bioanalytical Chemistry, 406(4), 957–969. https://doi.org/10.1007/s00216-013-7017-8
- News-Medical. (2023, July 19). Countercurrent chromatography. Retrieved from https://www.news-medical.net/life-sciences/Countercurrent-chromatography.aspx
