- Water is an indispensable part of our lives, yet it wouldn’t be wrong to state that the water from different sources is contaminated with heavy metals, dissolved salts, and microorganisms.
- As the global population grows and industrial activity expands, the quality of these sources faces unprecedented challenges, with raw pharmaceutical waste and waste from many more industries and human interventions polluting these water bodies.
- These contaminants must be meticulously removed from the water before it can be safely consumed or reused.
- Hence, an emerging necessity for precise, reliable purification has elevated membrane technology from a laboratory novelty to the indispensable cornerstone of the modern water industry.

- A membrane acts as a highly selective, physical barrier that acts as a molecular sieve, operating under pressure to separate pure water from virtually all unwanted constituents
- The ability of specialized membranes, such as Reverse Osmosis (RO), to desalt seawater, or Ultrafiltration (UF) to eradicate pathogens, ensures that humanity can secure its water future by making previously unusable sources safe, sustainable, and reliable for consumption, agriculture, and industry worldwide
https://watertreatmentsuppliers.com/types-of-membrane-filtration/
Membrane working, mechanism, and uses
- As mentioned before, a membrane acts as a sieve that does not allow several particles to pass through it. This is the semi-permeable nature of the membrane.
- A membrane is a thin, semipermeable sheet, typically constructed from polymers (like plastic) or ceramic materials. It functions as a precise barrier containing microscopic pores (tiny openings) that allow water to pass through while physically blocking larger contaminants.
- The water is pushed under pressure to pass through the membrane. Membranes don’t work passively; they require energy to overcome the natural resistance of the water and the contaminants. Hence, pressure is applied to move water through the tiny channels of the membrane (pores).
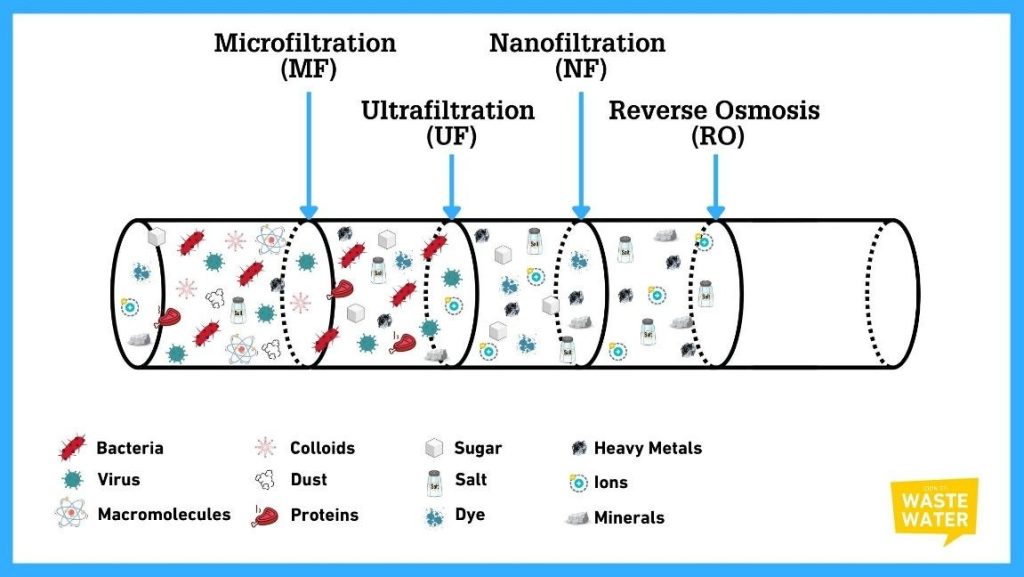
- Different types of membranes have different pore sizes, which are required to separate different contaminants of various sizes.
- This separation separates the feed water into permeate, which is the clean water that successfully passes through the membrane, and the Reject, which is water that contains all the concentrated contaminants that were blocked.
Types of Membrane Technology
Microfiltration
- Microfiltration is an essential first line of defence in water filtration, operating as a highly efficient physical sieve.
- It has the largest pore size, ranging from 0.1 to 10 μm.
- Driven by low pressure, its primary function is to achieve size exclusion, physically blocking contaminants larger than its pores.
- MF is indispensable in water treatment because it reliably removes suspended solids, turbidity, algae, and nearly all types of bacteria and large pathogenic protozoa (like Giardia and Cryptosporidium).
- While it does not remove viruses or dissolved salts, its high-volume throughput and relatively low energy demand make it the ideal, economical pre-treatment step for clarifying raw source water and safeguarding the more delicate, finer membranes (like Ultrafiltration and Reverse Osmosis) that follow downstream.
- Microfiltration is often paired with disinfection (like chlorination) for safety, or used in tandem with finer membranes for complete purification.
Ultrafiltration
- Ultrafiltration (UF) is the crucial, pressure-driven membrane technology that succeeds microfiltration in terms of fineness and elevates water purity.
- It functions as an extremely tight physical barrier, effectively removing the smaller contaminants that MF misses, such as all viruses and large organic molecules
- UF also works on the size exclusion principle, having tiny and uniform pores, a size range typically from 0.01 to 0.1 μm.
- UF operates at significantly lower pressures (and thus lower energy costs) than RO. This applied pressure is what creates the necessary pressure gradient to push the liquid through the pores.
- The typical operating pressure required for Ultrafiltration (UF) systems is moderate, usually ranging from 10 to 100 psi (pounds per square inch).
Nanofiltration
- Nanofiltration (NF) is the next filtration step after UF.
- It is a pressure-driven membrane technology that leverages a dual mechanism separating contaminants based on both their physical size and their electrical ionic charge.
- Nanofiltration (NF) is often called “loose Reverse Osmosis (RO)” because it functions as an intentionally less restrictive version of RO.
- While structurally and operationally similar to RO, NF is engineered to be less retentive (or “looser”), specifically targeting multivalent ions (like calcium and magnesium) for removal while allowing most harmless monovalent ions (like common salt) to pass through.
- This design choice results in lower operational pressure and significantly greater energy efficiency compared to the “tight” full desalination achieved by RO.
- NF operates at medium-to-high pressures (typically 70 to 150 psi), which is significantly higher than Ultrafiltration (UF), but still substantially lower than RO.
- NF uses a powerful principle called Charge Repulsion (or Donnan Exclusion) in addition to physical size-sieving.
- NF membrane possesses a slight negative charge on its surface, which creates an electrical field that actively repels ions as they approach the membrane pores.
- The strength of the repulsion is determined by the ion’s valency (the magnitude of its charge).
- Multivalent ions like Calcium (Ca2+) and Magnesium (Mg2+) are subject to strong repulsion and are therefore highly rejected. This is the primary mechanism NF uses for water softening, whereas Single-charged monovalent ions like Sodium (Na+) and Chloride (Cl−) experience much weaker repulsion and pass through the membrane easily.
Reverse Osmosis
- Osmosis is the natural movement of water (the solvent) across a semipermeable membrane from an area of low solute concentration to an area of high solute concentration. This process is passive, driven by nature’s attempt to equalize the concentrations on both sides of the membrane.
- Based on osmosis, Reverse Osmosis (RO) is an engineered process that applies high external pressure to a concentrated (salty) solution, forcing water molecules across a non-porous membrane, effectively leaving nearly all dissolved salts and contaminants behind to produce highly purified water. The principle is based on the solution diffusion mechanism.
- To achieve Reverse Osmosis, an external hydraulic pressure is applied to the feed water side that is greater than the natural osmotic pressure. This high pressure forces the flow in reverse.
- Dissolved inorganic salts, heavy metals, and most organic molecules have a very low solubility in the membrane material and are physically blocked or heavily impeded. They are swept away in the concentrated reject stream (brine), whereas Water molecules (H2O) are small enough and have enough chemical affinity to dissolve into the polymer film and diffuse across to the low-pressure side.
- Ro membrane pore size ranges from 0.001(μm), or about 0.0001 microns.
- The pressure required for Reverse Osmosis (RO) is the highest among all membrane filtration types because it must actively counteract the natural force of osmosis.
- There is no single PSI number for RO; the pressure required depends entirely on the salinity (salt concentration) of the source water.
References
- Ezugbe, E. O., & Rathilal, S. (2020). Membrane Technologies in Wastewater Treatment: A Review. Membranes, 10(5), 89. https://doi.org/10.3390/membranes10050089
- (PDF) Membrane Technologies in Wastewater Treatment: A Review
- https://www.snowate.com/parts/membrane/supports/membrane-comparison.html
- Shon, H. K., Phuntsho, S., Chaudhary, D. S., Vigneswaran, S., & Cho, J. (2013). Nanofiltration for water and wastewater treatment – a mini review. Drinking Water Engineering and Science, 6(1), 47–53. https://doi.org/10.5194/dwes-6-47-2013
- Racar, M., Dolar, D., Špehar, A., & Kožutić, K. (2017). Application of UF/NF/RO membranes for treatment and reuse of rendering plant wastewater. Process Safety and Environmental Protection, 105, 386–392. https://doi.org/10.1016/j.psep.2016.11.015
