The discovery of streptomycin in 1943 by Selman Waksman and Albert Schatz was a monumental moment in medical history. As the first successful drug against tuberculosis (TB), this aminoglycoside antibiotic immediately became a critical tool, transforming the treatment landscape for what was then a devastating global epidemic.
Streptomycin is biologically obtained from the microbe Streptomyces griseus, which belongs to the class Actinomycota, often appearing as a thin thread-like structure under a microscope, a filamentous and Gram-positive bacterium renowned for its prolific production of bioactive compounds, making it one of the most important microorganisms in the field of natural product discovery.
These are primarily found in soil and decaying vegetation; they are essential for breaking down complex organic matter. Colonies are often tough, leathery, and have a chalky, powdery, or dusty appearance due to the formation of spores on aerial hyphae. They typically produce grey spore masses. Along with other Streptomyces species, they are responsible for the distinct “earthy” smell of healthy soil, which comes from a volatile compound they produce called geosmin.
It is known to produce over 30 different structural types of bioactive compounds beyond just streptomycin, including other antibiotics (like grisein and candicidin) and enzymes (like pronase E). the production of these secondary metabolites (such as antibiotics) gives S. griseus a competitive advantage in the nutrient-scarce and microbe-filled soil environment, allowing it to inhibit the growth of competing bacteria and thrive.
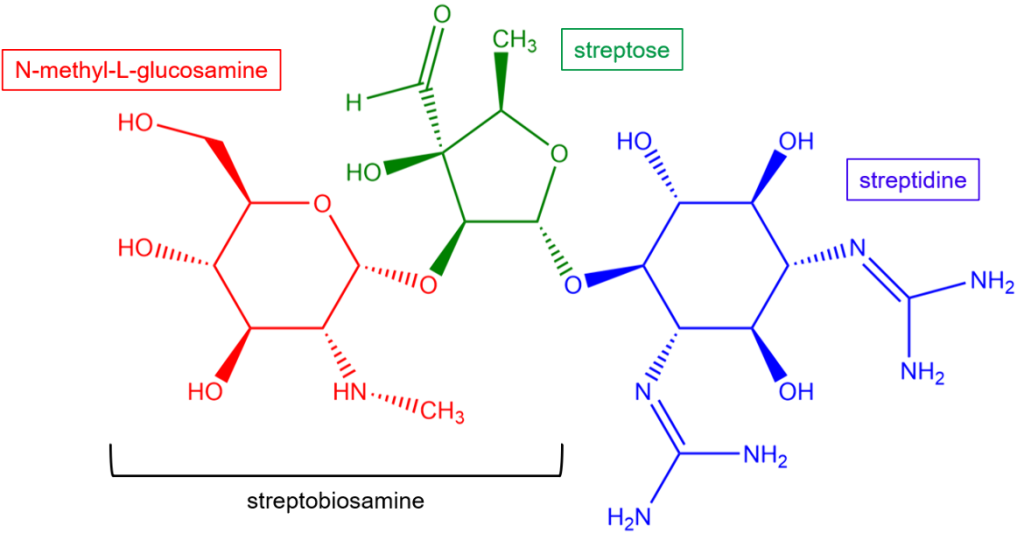
Chemical structure of Streptomycin
Aminoglycosides are named for their structure, which features amino sugars joined by a glycosidic bond. These drugs are highly basic because they possess many amino groups, causing them to be positively charged (polycations) in the body.
Streptidine is one of the three main components, making it the aminocyclitol moiety that connects the two sugar components (streptose and N-methyl-L-glucosamine).
It is a 1,3-dideoxy-1,3-diguanidinoscyllo-inositol. The ring itself is an inositol derivative (a sugar-like cyclic alcohol), but its defining feature is the presence of two guanidino groups. The presence of the basic guanidino groups is crucial for the drug’s positive charge and its ability to bind to the bacterial ribosome.
Streptidine is attached to 2 amino sugars, that is, streptose and N-methyl-glucosamine,e with a glycosidic bond. The amino sugars are collectively termed Streptobiosamine. Streptose is a unique, branched-chain five-carbon sugar that exists in this furanose form, having four carbon atoms and one oxygen atom, resembling the structure of tetrahydrofuran, whereas N-methyl-L-glucosamine, being a derivative of pyranose, exists in this more stable, six-membered pyranose form.
https://pdb101.rcsb.org/global-health/antimicrobial-resistance/drugs/antibiotics/protein-synthesis/ribosome/aminoglycosides/streptomycin/streptomycin
Streptose has an aldehyde group whose reduction to alcohol results in dihydrostreptomycin production. Dihydrostreptomycin was found to cause more frequent and irreversible damage to the auditory part of the inner ear, leading to permanent deafness. Streptomycin, by comparison, primarily affects the vestibular (balance) portion of the inner ear. Due to this severe toxicity profile in humans, dihydrostreptomycin is now predominantly used in veterinary medicine to treat bacterial diseases in livestock like cattle, pigs, and sheep.
Mechanism of Streptomycin Production
The part of streptomycin that is most crucial for its antibiotic activity is the entire molecule’s ability to bind irreversibly to the bacterial ribosome, which is facilitated by its multiple basic (amino/guanidino) groups. Streptomycin is effective because its many amino and guanidino groups become positively charged (polycations) at the body’s natural pH. This charge is the key to its function.
The positive charge helps the drug get attracted to and pulled into the bacterial cell, which has a negative charge.
Once inside, the multiple positive charges allow it to tightly bind to the negatively charged backbone of the 16S ribosomal RNA within the bacterial 30S ribosomal subunit.
The entire three-part structure of streptomycin, which is composed of streptidine, streptose, and N-methyl-L-glucosamine, locks into the ribosomal pocket. This binding causes two lethal effects:
It blocks the initiation of new protein chains (protein synthesis).
It causes the ribosome to misread the mRNA code, resulting in the production of faulty, non-functional proteins.
Ultimately, while the streptidine and its guanidino groups provide the crucial positive charge for initial binding, the entire molecular structure is required to secure the perfect fit on the 30S subunit and achieve its bactericidal (cell-killing) effect.
Industrial production of Streptomycin by fermentation
The production takes place in large, sterile bioreactors (fermenters) under carefully controlled conditions. This process is called submerged fermentation.
The culture medium is an aqueous broth containing necessary nutrients for the growth of the Streptomyces and the production of streptomycin.
The fermentation must be continuously supplied with sterile air (since S. griseus is aerobic) and maintained at an optimal temperature and pH for several days.
The media for the growth of the Streptomyces and the production of streptomycin include these basic components, which have been used for ages but were modified accordingly depending upon the strain and the nature of the industry.
| Made by | Components | Year |
| Woodruff & McDaniel | 1% Soybean meal (Nitrogen Source) 1% Glucose (Carbon source) 0.5% NaCL | 1954 |
| Hockenhull | 2.5% Glucose Carbon source) 4% Extracted Soybean meal (Nitrogen Source) 0.5% Distilleries dried soluble 0.25% NaCL pH- 7.3 to 7.5 before sterilization | 1963 |
The commercial fermentation of streptomycin can be divided into 3 phases.
The very 1st phase lasts for 24 hours, and rapid growth occurs in this phase with maximum mycelium production for fermentation. Ammonia is released into the medium due to the proteolytic activity of the microbe, and carbon is utilized slowly for growth. There is a slight production of streptomycin with a pH ranging from 6.8 to 7.5.
Later, entering phase 2 of the fermentation, there is rapid and increased production of streptomycin; this phase lasts for about 24 hours to 6 or 7 days of incubation. There is no mycelial growth, which indicates a constant weight of mycelium. Ammonia is utilized, and glucose is withdrawn from the medium so that only a little is left by the end of this phase. pH is fairly constant from 7.8 to 8.
As the cycle reaches phase 3, glucose is depleted from the medium, streptomycin production terminates, pH increases, cells lyse, and ammonia is released.
Harvest of Streptomycin
After the fermentation period is complete, the streptomycin must be separated from the culture broth and purified.
Streptomycin, being a basic, positively charged compound (a polycationic aminoglycoside), is effectively captured by techniques like ion-exchange chromatography (using a resin that binds positive ions).
The captured streptomycin is then eluted (washed off) from the resin using an acid or base solution. It is finally precipitated out of the solution, often as the stable streptomycin sulphate salt.
The final product is dried and prepared for clinical use.
The overall efficiency and cost-effectiveness of the process depend critically on optimizing the fermentation conditions and the purification steps.
References
- PDB101: Global Health: Antimicrobial Resistance: undefined: Streptomycin. (2016). RCSB: PDB-101. https://pdb101.rcsb.org/global-health/antimicrobial-resistance/drugs/antibiotics/protein-synthesis/ribosome/aminoglycosides/streptomycin/streptomycin
- Petrides, D., Mustafa, A., Da Gama Ferreira, R., & Misailidis, N. (2022). Streptomycin Production via Fermentation – Process Modeling and Techno-Economic Assessment (TEA) using SuperPro Designer. ResearchGate. tsr067.pdf
- Author, A. A., Author, B. B., & Author, C. C. (Year). Optimization of a culture medium for streptomycin production using response-surface methodology. Journal Title, Volume(Issue)
- A Method for the Large-scale Production of Streptomycin by Surface Culture | Microbiology Society
- https://www.youtube.com/watch?v=rVcMt4wLFQA
- https://www.youtube.com/watch?v=OQlC65DD3Ig
- Production of Streptomycin – Definition, Production & Uses – Biology Reader
