Polymerase Chain Reaction is a thermal process used for the amplification of the gene of interest. Invented in 1983 by Kary Mullis, this method works on the principle that when provided with optimal conditions, DNA polymerase synthesizes a copy of DNA from a strand of template DNA. The optimal temperature varies for the three steps in the PCR: Denaturation, annealing, and extension, and such an environment is provided by an instrument referred to as a thermocycler.
Multiplex PCR is a variant of the polymerase chain reaction in which two or more target DNA sequences are simultaneously amplified in a single reaction using multiple primer pairs.

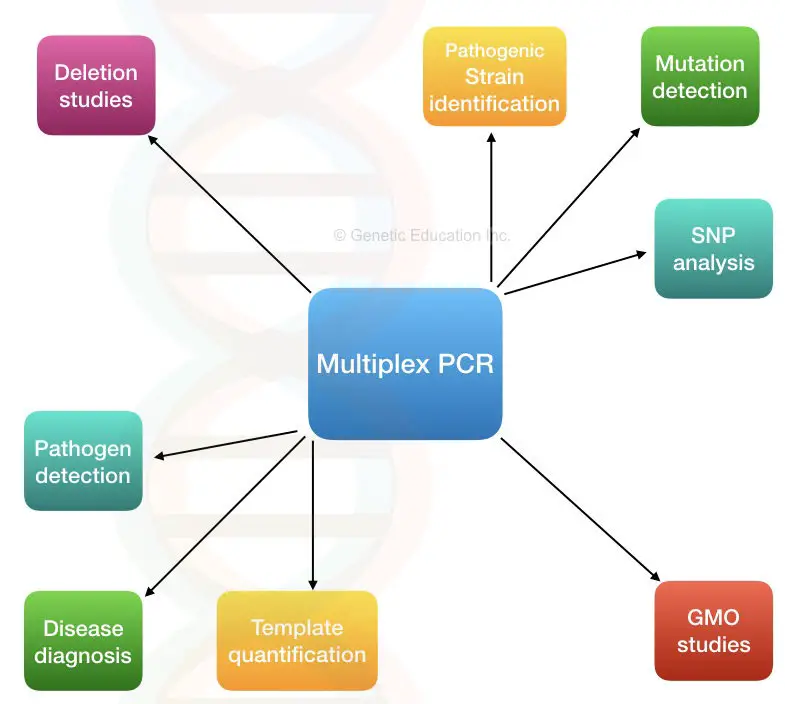
Multiplex PCR improves efficiency by enabling concurrent amplification of multiple targets, thereby reducing time, reagent use, and sample requirements compared to conventional PCR. It is widely applied in gene deletion analysis, mutation and polymorphism detection, quantitative studies, and reverse transcription-based assays. Due to its high throughput capability, it is commonly used in clinical diagnostics and research screening workflows.
Objectives of Multiplex PCR
The objectives of multiplex PCR include:
- To speed up the process of PCR involving the same targets.
- To be cost-effective and time-sensitive.
- To reduce the chances of false negative results that are usually not detected in traditional PCR methods.
- Multiple copies of more than two targets can be produced from a single PCR cycle.
Requirements of Multiplex PCR
The materials used for Multiplex PCR include:
- Primers: short stretches of oligonucleotides that bind to the target DNA to start the amplification process
- Deoxynucleotides
- Buffers
- DMSO
- Taq DNA polymerase
- Thermocycler
- Magnesium Chloride
- Template DNA
Principle of Multiplex PCR
Multiplex PCR works on the same principle as traditional PCR techniques. In traditional PCR, the reaction mixture containing the primers, buffers, DNA polymerase, etc, is prepared and then poured into the thermocycler for the amplification to take place. This allows for only a single template DNA to be used. In the case of the multiplex PCR technique, the reaction mixture is prepared; however, one or more target DNAs are utilised here. The amount to be used is based on the requirement of the result. The primers, which are specific to these target DNAs, are added so that multiple PCR reactions can take place within the same PCR cycle.
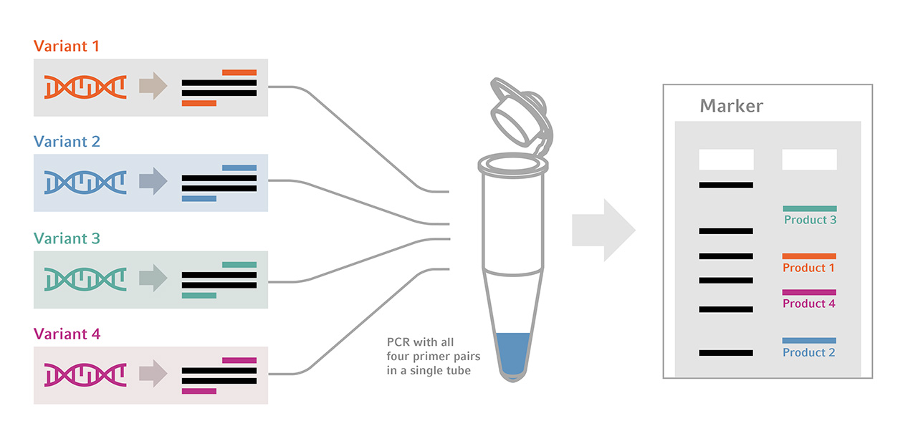
Multiplex PCR utilizes multiple primer pairs to amplify multiple targets simultaneously. Therefore, it is necessary to obtain a primer-template ratio to ensure there are no discrepancies, such as the formation of primer dimers, during the amplification process. The PCR reaction should be optimized in a way that decreases any nonspecific interactions as such. This can be done by taking into consideration the primer designs, such as homology of the primer with the target DNA, the length, the GC content, and concentration. The optimal primer length is 18-24 bp or higher with a GC content of 35%-60% with an annealing temperature of 55°-58°C. Longer primer length accounts for the production of non-specific products.
Steps of Multiplex PCR
The basic protocol of multiplex PCR includes:
Preparation of the mixture required for the PCR reaction (25mL vol)
Water was ultrafiltered and autoclaved, and utilized for the preparation of the buffer. dNTP mixture (200 mM each), Primers (0.04–0.6 mM each), DMSO, Glycerol, Taq DNA polymerase (1–2 U/25 mL), and the genomic template DNA (150 ng/25 mL), all of appropriate quantities, were prepared. These components were added in any order (with water being first). The pipetting of the reaction mixture into the vials was done on ice, and these vials were placed on a pre-heated metal block or a water bath (94°C) of the thermal cycler.
Amplification
Multiplex PCR follows the basic amplification steps: denaturation, annealing, and extension. The initial denaturation is done at 90-95°C for 5 minutes, while the subsequent ones are for 1 minute at the same temperature. Upon denaturation of the template DNA, annealing of the primers to the target DNA takes place. The annealing temperature is 55-60°C for 50 seconds. Next is the extension at 72°C for 1 minute and the final extension for 7 minutes at the same temperature. The entire process takes about 25-28 cycles.
Post-amplification analysis
The analysis of the PCR products was done using gel electrophoresis. The products were loaded onto the gel slot. The electrophoresis was performed, and the results were observed after staining the gel with ethidium bromide.
Types of Multiplex PCR
Depending on the type of template, multiplex PCR is of two types:
- Uni-template Multiplex PCR
- Multi-template Multiplex PCR
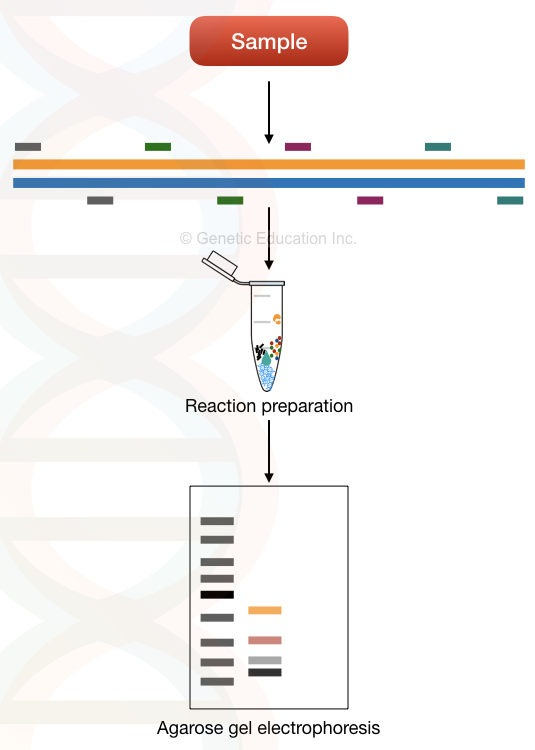
Uni-template Multiplex PCR
Unlike the normal multiplex PCR, which utilizes multiple template DNAs, here various loci of a single DNA target are amplified. It amplifies the multiple regions present in a single gene using a different set of primers. For example, 5 different mutations of beta-thalassemia can be detected from a single beta-globin gene by using 5 different sets of primers. This method is useful in the application of detecting and genotyping loci for screening single-gene inherited diseases.
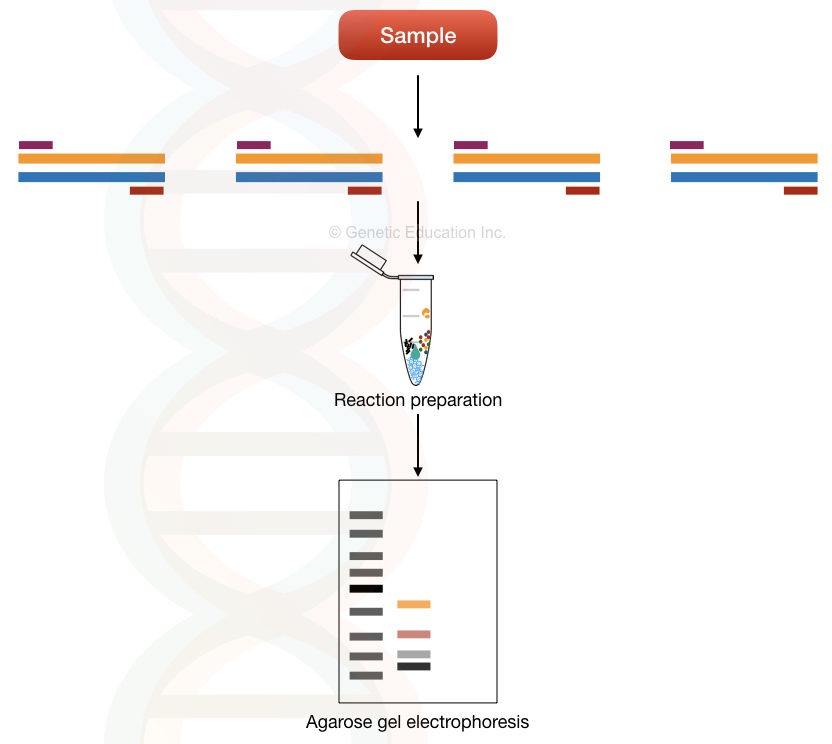
Multi-template Multiplex PCR
Here, the amplification of different templates using different sets of primers is performed in a single reaction. The primers used are specific to each template and therefore can only amplify the specific target DNA. This reduces the chance of the formation of primer dimers.
This method is used in applications such as microbial genetics, pathogen identification, and microbial detection. Unlike Uni-template PCR, this is not useful for single-gene disorder detection.
Examples of Multiplex PCR Kits
CAR/TCR Gene Copy Number Detection Kit (Multiplex qPCR) (Hillgene)
- It is used for the detection of the CAR gene, utilising multiplex PCR technology
- Accurately detects CAR copy number. Dual detection due to incorporated sequences as well as references.
- This kit utilises the fluorescent probe method coupled with multiplex PCR for detection.
Multiplex PCR Kit (HIMEDIA)
- Designed for parallel amplification of multiple fragments in a single PCR assay.
- The kit can be utilised for routine PCR reactions and is suitable for multiple target gene amplification in a single tube.
- The kit is a ready-to-use 2x solution containing standardised polymerase composition, dNTPs, MgCl2, and reaction buffers.
GeneProof Flu Multiplex PCR Kit (GeneProof)
- Used for the detection of Influenza A, Influenza B, and RSV in a single reaction tube.
- Contains all components required for PCR amplification, does not require additional pipetting
- Utilises the multiplex PCR method for the detection of the viruses.
Multiplex PCR 5X Master Mix (New England Biolabs)
- Allows the simultaneous detection of two or more products in a single reaction.
- Provides optimal performance with the use of enhanced buffer formulation.
- Has an easy reaction setup and is only required to add the template DNA and the primers
QIAGEN Multiplex PCR Kit
- A highly specific and sensitive kit with no requirement for optimisation before use.
- It is a 2x Master mix kit that provides a final concentration of 3mM MgCl2, 3 x 0.85ml, additionally a 5 x Q-solution (1 x 2.0 ml), and RNase-free water (2 x 1.7mIt iishighly specific since there is no requirement for optimization. Additionally, it is equipped with hot start technology.
- It is easy to use and cost-effective.
- The PCR buffer used contains a novel synthetic Factor MP. This factor, along with optimized salt concentrations, helps in stabilizing specifically bound primers and enables the proper extension of any primer used in the reaction.
- Another additive, referred to as the Q solution, enables the efficient amplification of templates that are difficult to amplify, such as GC-rich templates.
- This kit is used on applications such as the typing of transgenic organisms and microsatellite analysis.
Applications of Multiplex PCR
Since multiplex PCR works on the principle of amplification of multiple targets at the same time, it finds itself in various applications, such as:
- Microbial Genetics: It is widely used in pathogen detection, identification, and characterization. The traditional PCR method can be time-consuming and prone to false results from contamination from repeating the steps multiple times. Multiplex PCR can combat these issues when it comes to pathogen studies. Various strains of pathogens present in a sample can be identified and studied in an accurate and efficient manner through multiplex PCR. Computational tools can further help increase efficiency. This method is also applied in gene deletion and mutation detection studies.
- DNA typing: Multiplex PCR is an ideal technique for DNA typing, for applications such as paternity testing, forensic identification, and population genetics. This is because the probability of identical alleles occurring in individuals decreases with the number of polymorphic loci.
- Metagenomic Studies: Multiplex PCR can be used to study DNA obtained from environmental samples such as soil, water, dead leaves, or any sort of biomass. Computational studies can be done to make the results more accurate.
- Infection Detection: Multiplex PCR is widely used in the detection of SARS-CoV. There are various detection kits for infections such as VZV, CMV, T. gondii, influenza, and adenovirus.
- Single Nucleotide Polymorphism genotyping: Traditional PCR can only identify a couple of genotypes from a single encounter with a single nucleotide polymorphism. With multiplex PCR, multiple SNPs can be identified in a single reaction, allowing for the study of multiple genotypes. However, with an increased set of primers, the chances of reaction failure and false-positive reactions can occur.
- Genetically Modified Organism studies: Since GMOs are about the study of genes and their alterations, the use of Multiplex PCR allows for studying multiple genes in a single reaction.
- Qualitative and quantitative analysis of templates: The Quality of the template DNA determines the results of a PCR reaction. This is crucial in cases of studies such as sequencing and microarray, where a good quality template provides reliable results. Since multiple allows theofvariouss geneare allowed at a single time, many temperate ‘regions’ (DNA loci) can be studied easily.
Some other applications of multiplex PCR include: linkage analysis, forensic analysis, and gene transfer study.
Advantages of Multiplex PCR
Some of the advantages of Multiplex PCR include:
- Amplification of multiple DNA templates in a single reaction or amplification of multiple loci in a single gene.
- Rapid and cost-effective, since the use of reagents and other components multiple times is reduced.
- Highly reliable and efficient method providing accurate results.
- The amplicon in the reaction serves as an ‘internal control’ for another reaction, therebyhelping tos encounter false-positive results.
- The quality of the DNA can be analyzed by comparing different amplicons of the same gene or DNA.
- Reduces the chances of pipetting errors.
Limitations of Multiplex PCR
The limitations of Multiplex PCR include:
- The chances of non-specific interactions occurring are high, for example, the formation of primer dimers. If the reaction is not done properly, there is a chance of high reaction failure.
- Longer templates cannot be amplified properly. Accurate amplification occurs only up to 100bp.
- Diversity in the use of templates cannot be achieved; shorter templates provide proper results.
- Every primer set is not compatible with every other one all the time.
- Reaction failure may occur with the use of a large number of templates.
Conclusion
The traditional PCR method, despite being reliable, can be time-consuming and expensive owing to the fact that multiple reactions need to be performed for various target DNAs. This issue can be combatted with a multiplex PCR, where multiple target DNAs can be amplified with different sets of primers in a single reaction. This can also be done for multiple loci in the required target DNA. Such a method is useful for various applications such as microarray, pathogen detection, gene mutation and alteration studies, forensic analysis, etc. Despite the limitations with multiplex DNA, if performed properly, Multiplex PCR is a very useful method to obtain reliable and efficient results.
References
- https://geneticeducation.co.in/multiplex-pcr-principle-process-protocol-advantages-limitations-and-applications/#Applications_of_multiplex_PCR
- Edwards, M. C., & Gibbs, R. A. (1994). Multiplex PCR: advantages, development, and applications. Genome Research, 3(4), S65-S75.
- https://www.qiagen.com/us/products/discovery-and-translational-research/pcr-qpcr-dpcr/pcr-enzymes-and-kits/end-point-pcr/qiagen-multiplex-pcr-kit
- https://en.hillgene.com/products/tcr-gene-copy-number-detection-kit-multiplex-qpcr/
- https://www.himedialabs.com/in/mbt118-multiplex-pcr-kit.html
- https://www.neb.com/en/products/m0284-multiplex-pcr-5x-master-mix
- https://www.geneproof.com/geneproof-flu-multiplex-pcr-kit/p6684
- Markoulatos, P., Siafakas, N., & Moncany, M. (2002). Multiplex polymerase chain reaction: a practical approach. Journal of clinical laboratory analysis, 16(1), 47-51.
- Henegariu, O., Heerema, N. A., Dlouhy, S. R., Vance, G. H., & Vogt, P. H. (1997). Multiplex PCR: critical parameters and step-by-step protocol. Biotechniques, 23(3), 504-511.
- https://frontlinegenomics.com/what-is-multiplex-pcr/
