Carbohydrate metabolism disorders are metabolic abnormalities that impact the anabolism and catabolism of carbohydrates. Most of these illnesses are caused by an inability to use carbohydrate metabolites efficiently.
They are classified into inborn errors of carbohydrate metabolism and acquired carbohydrate metabolic disorders according to their etiology.
Inborn errors of carbohydrate metabolism
- Inborn errors of carbohydrate metabolism refer to hereditary metabolic defects caused by mutation of genes encoding enzymes, transporters, or cofactors that are important in carbohydrate catabolism or anabolism.
- These genetic defects are usually inherited according to the Mendelian patterns (in most cases, autosomal recessive) and cause the disruption of biochemical pathways.
Major inborn errors of carbohydrate metabolism are:
- Glycogen Storage Disease (GSD): The disorder is caused by the impairment of glycogenolysis enzymes, including glucose-6-phosphatase, resulting in Hepatomegaly and hypoglycemia.
- Congenital Galactosemia: A deficiency of galactose-1-phosphate uridyltransferase (GALT) blocks the Leloir pathway, leading to cataracts and liver failure following the intake of milk.
- Galactokinase Deficiency: Cataracts and galactitol buildup are caused by impaired galactose phosphorylation.
- Essential Fructosuria: Fructose-related benign fructokinase defect results in the fructose excretion in the urine.
- Hereditary Fructose Intolerance: An aldolase B deficiency results in the development of hypoglycemia and liver and kidney damage in the presence of fructose.
- Essential Pentosuria: A lack of L-xylulose reductase causes innocuous pentose excretion.
Acquired carbohydrate metabolic disorder
- Acquired carbohydrate disorders are disorders that occur following birth, and they are not associated with inborn genetic defects.
- Rather, they are caused by environmental, lifestyle, systemic, or physiological abnormalities in carbohydrate metabolism.
- Prevalent in general populations.
- Hormonal dysregulation and metabolic syndrome are frequently linked.
- Hyperglycemia, hypoglycemia, and metabolic problems resulting from other illnesses are examples of symptoms.
Different acquired Carbohydrate Metabolic Disorders
- Absorptive/Intolerance Types: Lactose intolerance (primary adult-onset or secondary)
- Hyperglycemic/Insulin Resistance Types: Type 2 diabetes mellitus (T2DM), prediabetes
- Hypoglycemic/Deficiency Types: Reactive hypoglycemia, secondary hypolactasia from mucosal damage
Glycogen Storage Diseases (GSDs): An Overview of Types I through VII
- Glycogen storage diseases (GSDs), also called glycogenoses, are hereditary metabolic disorders that are caused by a deficiency of enzymes or transporters that support the synthesis or degradation of glycogen.
- They lead to impaired glucose homeostasis by causing an abnormal accumulation and/or use of glycogen.
- Incidence ranges between 1/20,000 and 43,000 live births.
- The main clinical signs are fasting hypoglycemia, hepatomegaly, hyperlipidemia, exercise intolerance, muscle cramps, rhabdomyolysis, and muscle weakness.
- GSDs are categorized according to the missed enzyme or transporter and tissues affected (liver, muscle, or both) in the first place.
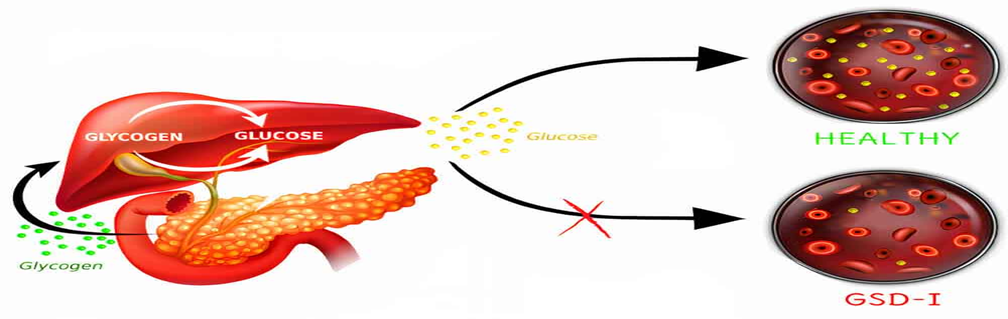
GSD‑I (von Gierke disease):
- The GSD-I is caused by the lack of glucose-6-phosphatase (GSD-Ia) or the glucose-6-phosphate transporter (GSD-Ib).
- This malfunction inhibits the last stage of glycogenolysis and gluconeogenesis and causes severe fasting hypoglycemia.
- GSD-Ia is marked by hypoglycemia, lactic acidosis, hyperlipidemia, and hepatomegaly.
- GSD-Ib: Other than the metabolic symptoms, neutropenia, frequent infections, and inflammatory bowel disease are common in patients.
GSD- II (Pompe Disease)
- GSD-II is a condition that is caused by the lack of acid α-glucosidase, an enzyme of lysosomal localization, which leads to the accumulation of glycogen in the lysosomes, particularly in the heart and skeletal muscles.
- Infantile-onset: presents with severe cardiomyopathy and profound muscle weakness; often fatal within the first year.
- Late-onset: characterized by progressive skeletal muscle weakness, with variable cardiac involvement.
GSD – III (Cori/Forbes Disease)
- It is caused by the insufficiency of glycogen debranching enzyme (AGL) and causes the accumulation of glycogen in the form of limit-dextrin.
- Type IIIa: is known to attack the liver and muscle (including the heart).
- Type IIIb: It mostly affects the liver.
- Clinical characteristics involve hepatomegaly, hypoglycemia, muscle weakness, and cardiomyopathy in some instances.
GSD- IV (Andersen Disease)
- It is a mutation of the glycogen branching enzyme (GBE1). The resultant glycogen is defective, amylopectin-like, and it builds up in tissues.
- Classic forms often present in early childhood with progressive liver cirrhosis and liver failure.
- There is a development of neuromuscular complications or the development of adult-onset polyglucosan body disease in some patients.
GSD-V (McArdle Disease)
- This form is characterized by a lack of muscle glycogen phosphorylase (PYGM) and mostly affects skeletal muscle.
- Exercise intolerance, muscle cramps, and fatigue are the common symptoms exhibited by patients.
- One notable point worth noting is the phenomenon of the second wind, where the symptoms get better as the individual continues to work.
GSD-VI (Hers Disease)
- GSD-VI is caused by the lack of liver glycogen phosphorylase (PYGL).
- Clinical manifestations usually include hepatomegaly and mild hypoglycemia, and the degree may be different.
- It may cause complications like fibrosis or cirrhosis, but they are mild in most situations.
GSD- VII (Tarui Disease)
- The cause of this type is the deficiency of muscle phosphofructokinase (PFKM) that affects the metabolism of muscle energy.
- Patients have exercise intolerance, cramps, and myoglobinuria.
- Serious ones can also manifest him/herself with hemolytic anemia and multisystem involvement.
Von Gierke Disease (Type I): Glucose-6-Phosphatase Deficiency
- A genetic metabolic disease due to a lack of glucose-6-phosphatase or the carrier of its transporter, preventing the final stage of glycogen breakdown and glucose synthesis.
- Due to the inability of glucose-6-phosphate to be metabolized into glucose, the liver is unable to sustain normal blood glucose levels, leading to severe fasting hypoglycemia.
- The glucose-6-phosphate accumulated is diverted to other metabolites with the resultant lactic acidosis, hyperlipidemia, and hyperuricemia.
- The liver and kidneys are considered the main sites of extra glycogen, which causes severe hepatomegaly.
- The disorder is of Type Ia, which is caused by enzyme(glucose-6-phosphatase) deficiency, and Type Ib, which is caused by impaired transport of glucose-6-phosphate into the endoplasmic reticulum.
- It is an autosomal recessive inheritance.
Pompe Disease (Type II): Lysosomal Alpha-Glucosidase Deficiency
- Caused by a deficiency of the enzyme alpha-1,4-glucosidase, which is the enzyme that breaks down glycogen in lysosomes.
- Accumulates glycogen in lysosomes, which affects mostly skeletal and cardiac muscle cells.
- Infantile-onset form: characterized by early-life heart failure, cardiomegaly, and severe muscle weakness.
- Late-onset/juvenile form: In this form, cardiac involvement is less evident, and it manifests in progressive skeletal muscle weakness, especially in the respiratory muscles.
- Impaired glycogen breakdown leads to dysfunction of the cells and enlargement of the lysosomes, with clinical consequences.
- One of these treatment alternatives is enzyme replacement therapy (ERT): diagnosing the condition requires genetic testing or enzyme assays.
McArdle Disease (Type V): Muscle Phosphorylase Deficiency and Exercise Intolerance
- McArdle disease is A metabolic myopathy of a rare hereditary nature due to the inability to break down glycogen in skeletal muscle because of the absence of the enzyme muscle glycogen phosphorylase (myophosphorylase), the essential enzyme in degrading glycogen in skeletal muscle.
- It is a metabolic autosomal recessive disorder that causes mutations in the PYGM gene; affected individuals do not have an active myophosphorylase enzyme in the muscle cells.
- Exercise intolerance is the characteristic clinical feature, as people become tired quickly and are unable to continue exercising, because the stored muscle glycogen cannot be utilized to provide energy.
- Patients usually have painful muscle cramps, early fatigue, weakness, and stiffness within a short period of commencing exercise, which change with rest.
- Second Wind Phenomenon: A second-wind phenomenon is common in many patients who experience an improvement in symptoms upon 10 minutes of moderate exercise or short rest as the muscles start utilizing blood-borne fuels rather than glycogen.
- Long-term or strenuous exercise can lead to rhabdomyolysis, release of muscle protein, e.g., creatine kinase and myoglobin. This may, in the worst-case scenario, lead to acute renal failure.
- Exercise intolerance and the lack of myophosphorylase on muscle biopsies are important diagnostic characteristics; genetic testing verifies PYGM mutations.
Galactosemia: Genetic Defects in Galactose Metabolism
- Galactosemia is a genetic metabolic disease that is very rare and occurs due to genetic defects in galactose metabolic enzymes.
- The Leloir pathway normally converts galactose, which is mostly derived from milk sugar (lactose), into glucose; genetic mutations in the GALT, GALK1, or GALE genes interfere with this process, causing a toxic buildup of galactose and its metabolites.
- The mutation affects the galactose to glucose conversion process, as a result of which there are high levels of Galactose and galactose-1-phosphate.
Types of galactosemia
- Type I – Classic (GALT deficiency): Most severe form with almost absent enzyme activity. The neonatal symptoms comprise poor feeding, lack of vigour, retarded growth, jaundice, liver dysfunction, predisposition to E. coli sepsis, and low blood sugar levels. In the long term, there might be developmental delay, speech problems, cataracts, and ovarian failure in women.
- Type II (GALK deficiency): Primarily affects galactokinase, often milder. Mainly causes cataracts in the eyes because of galactitol crystals. There is no severe brain or hepatic damage; it is prevented by diet.
- Type III (GALE deficiency): There are three forms of epimerase deficiency of varying degrees of severity. The peripheral type is primarily related to blood cells, the intermediate type presents a partial deficit in various tissues, and the severe generalized type is uncommon, systemic, and similar to classic galactosemia.
Hereditary Fructose Intolerance (HFI): Aldolase B Deficiency
- HFI is a rare autosomal recessive disorder caused by mutations in the ALDOB gene, resulting in a lack of aldolase B, which is a fructose metabolizing enzyme.
- Results in a lack of aldolase B, which is a fructose-metabolizing enzyme.
- Deficiency causes fructose-1-phosphate accumulation, cellular toxicity, phosphate trapping, ATP depletion, and impaired glucose release.
- The symptoms tend to manifest during infancy with the introduction of fructose or sucrose into the diet.
- Clinical features include nausea, vomiting, abdominal pain, hypoglycemia, jaundice, hepatomegaly, and failure to thrive.
- Genetic ALDOB mutation testing is the initial method of diagnosis since fructose tolerance tests pose a high risk of the disease.
- Management encompasses lifetime avoidance of fructose, sucrose, and sorbitol. The problem of dieting with a metabolic specialist helps avoid the acute and long-term effects of complications.
Disorders of Gluconeogenesis: Hypoglycemia and Lactic Acidosis
- Inherited defects in the metabolic pathways that produce glucose from non-carbohydrate precursors, such as lactate, glycerol, and amino acids.
- Primary sites of gluconeogenesis are the Liver and kidneys, and enzyme deficiencies in these tissues can lead to systemic metabolic consequences.
Major Enzyme Deficiencies:
- Fructose-1,6-bisphosphatase deficiency impairs the conversion of fructose-1,6-bisphosphate to fructose-6-phosphate, causing severe fasting hypoglycemia.
- Pyruvate carboxylase deficiency – limits oxaloacetate formation from pyruvate, disrupting gluconeogenesis and the TCA cycle.
- Phosphoenolpyruvate carboxykinase deficiency – blocks conversion of oxaloacetate to phosphoenolpyruvate, reducing glucose output.
Pathophysiology
- Impaired gluconeogenesis leads to lactate building up because it cannot be efficiently converted into glucose, causing lactic acidosis. At the same time, low blood sugar (hypoglycemia) forces the body to activate compensatory pathways, stressing overall energy balance.
Clinical features
- Disorders of gluconeogenesis impair glucose production during fasting, causing hypoglycemia, lactate accumulation with metabolic acidosis, and sometimes ketosis, leading to energy deficits and clinical symptoms like weakness and fasting intolerance.
Pyruvate Dehydrogenase Complex Deficiency (PDCD): Energy Crisis in the Brain
- A genetic mitochondrial disorder caused by mutations in genes encoding the pyruvate dehydrogenase complex (PDC).
- Impaired PDC activity prevents the conversion of pyruvate to acetyl‑CoA, reducing ATP production and forcing pyruvate into lactate, causing lactic acidosis.
- Since the brain is highly energy dependent, diminished ATP production due to PDC deficiency leads to neuronal energy failure, contributing to neurological dysfunction.
- Often presents in infancy with developmental delay, hypotonia, seizures, and structural brain abnormalities such as dysgenesis of the corpus callosum or Leigh syndrome.
- Energy deficits in neural tissue explain the severity of symptoms. Early diagnosis is essential for management and genetic counseling.
Mucopolysaccharidoses (MPS): Defects in Glycosaminoglycan Degradation
- Rare inherited lysosomal storage disorders are caused by genetic defects in enzymes that degrade glycosaminoglycans (GAGs). GAGs are long sugar chains crucial for connective tissue and organ structure.
- Deficiency of multiple lysosomal hydrolases halts the stepwise breakdown of GAGs such as heparan sulfate, dermatan sulfate, keratan sulfate, and chondroitin sulfate, leading to GAG accumulation in lysosomes.
- Excessive GAG storage impairs cellular function, triggers inflammation, and disturbs organ systems progressively.
- Clinical manifestations include coarse facial features, skeletal deformities (dysostosis multiplex), organomegaly, joint stiffness, and variable neurological impairment depending on MPS types.
Conclusion
- Carbohydrate metabolism disorders consist of genetic abnormalities that interfere with sugar processing.
- These can be inherited, a result of enzyme or transporter defects, or acquired subsequently through hormonal or metabolic imbalances.
- Such disorder disrupts glucose balance, leading to hypoglycemia, lactic acidosis, organ enlargement, muscle weakness, or neurological damage.
- Conditions including glycogen storage disorders, galactosemia, fructose intolerance, and gluconeogenic defects illustrate the systemic impact of impaired carbohydrate metabolism on organ function and energy homeostasis.
References
- Davis, M. (2021, December 7). Glycogen storage disease type I (GSD-I) | Von Gierke disease. A2Z Healthy. https://a2zhealthy.com/glycogen-storage-disease-type-i/
- Demczko, M. (2025). Overview of carbohydrate metabolism disorders. MSD Manual Professional Edition.
- Frye, R. E., & Slonim, A. E. (2020). Pyruvate dehydrogenase complex deficiency. In M. P. Adam et al. (Eds.), GeneReviews®. University of Washington, Seattle. https://www.ncbi.nlm.nih.gov/books/NBK571223/
- Guffon, N., & Beck, M. (2021). Mucopolysaccharidoses. In A. Burlina (Ed.), Inborn Errors of Metabolism: Diagnosis and Treatment (pp. 755–770). Springer. https://link.springer.com/chapter/10.1007/978-3-030-67727-5_64
- Gümüş, E., & Özen, H. (2023). Glycogen storage diseases: An update. World journal of gastroenterology, 29(25), 3932–3963. https://doi.org/10.3748/wjg.v29.i25.3932
- Lakhan, S. E. (2025, January 30). Disorders of carbohydrate metabolism. Medscape. https://reference.medscape.com/article/1183033-overview
- Metabolic Support UK. (n.d.). Hereditary fructose intolerance. https://metabolicsupportuk.org/condition/hereditary-fructose-intolerance/
- National Library of Medicine. (n.d.). Galactosemia. MedlinePlus Genetics. https://medlineplus.gov/genetics/condition/galactosemia/
- Wassel, J. (2025, October 30). Galactose metabolism disorders. The Medical Biochemistry Page. https://themedicalbiochemistrypage.org/galactose-metabolism-disorders/
