- Cholesterol is the most important sterol in the human body.
- Almost half of the cholesterol in the body derives from de novo biosynthesis.
- The liver and intestines are major contributors to endogenous production.
- The liver is the primary site of de novo cholesterol synthesis, accounting for approximately 80% of total cholesterol synthesis in mammals.
- HMG-CoA reductase (3-hydroxy-3-methylglutaryl-coenzyme A reductase) is the key regulatory and rate-limiting enzyme.
- The synthesis starts from acetyl-CoA, which is derived from protein, carbohydrate, and fat metabolism.
- The process is energy-intensive and requires ATP and NADPH.
- To produce one mole of cholesterol, 18 moles of acetyl-CoA, 36 moles of ATP, and 16 moles of NADPH are required.
Site and Significance of Cholesterol Biosynthesis
- Biosynthesis of cholesterol primarily occurs in the liver’s cytoplasm and endoplasmic reticulum.
- This synthesis provides cholesterol, which is essential for cellular homeostasis, contributing to membrane rigidity and fluidity.
- It is the necessary precursor to the steroid hormones such as glucocorticoids, mineralocorticoids, and sex hormones produced in the endocrine tissues.
- Synthesis of bile acids out of cholesterol facilitates the excretion of cholesterol and the digestion of lipids in the diet.
- Vitamin D is synthesized from a cholesterol backbone and regulates calcium homeostasis and bone mineralization.
The Pathway Breakdown: From Acetyl-CoA to Cholesterol
The synthesis of cholesterol may be learnt in 5 stages
- Synthesis of HMG CoA
- Formation of mevalonate (6C)
- Production of isoprenoid units (5C)
- Synthesis of squalene (30C)
- Conversion of squalene to cholesterol (27C).
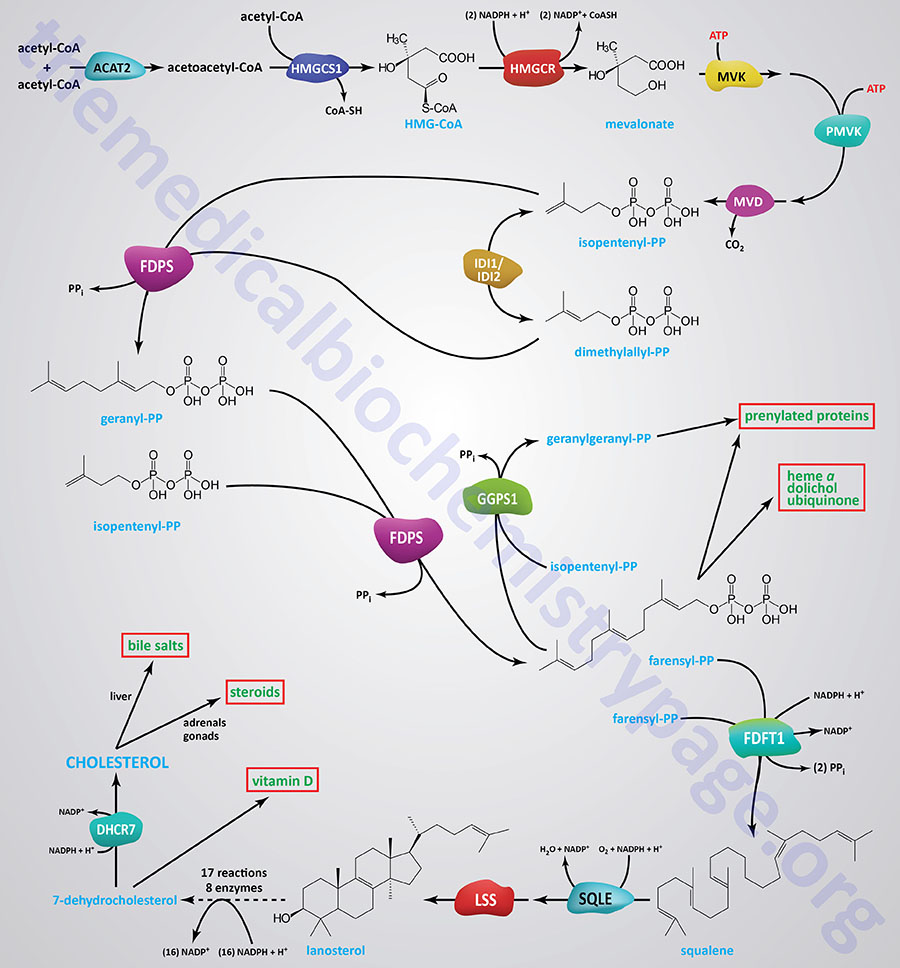
1. Formation of HMG-CoA: The Initial Condensation Steps
- The process starts with the two molecules of acetyl-CoA, which combine to start the biosynthesis of cholesterol.
- These two acetyl-CoA molecules are condensed to acetoacetyl-CoA, which is catalyzed by the enzyme thiolase in the cytosol, also known as acetyl-CoA acetyl transferase 2 (ACAT2).
- Acetoacetyl-CoA then reacts with a 3rd molecule of acetyl-CoA to produce HMG-CoA.
- This reaction is catalyzed by the cytosolic HMG-CoA synthase (encoded by HMGCS1).
- The HMG-CoA thus formed acts as a precursor of mevalonate synthesis, which is the subsequent stage of cholesterol biosynthesis.
2. The Rate-Limiting Step: Synthesis of Mevalonate by HMG-CoA Reductase
- HMG-CoA is transformed into mevalonate by HMG-CoA reductase (HMGR), which is an endoplasmic reticulum-bound enzyme.
- Two molecules of NADPH are required for this reduction, making it NADPH-dependent.
- This is the rate-limiting and most important regulatory step in cholesterol biosynthesis.
3. From Mevalonate to Squalene: Formation of Isoprenoid Units
- Mevalonate is phosphorylated by mevalonate kinase and phosphomevalonate kinase to form mevalonate-5-pyrophosphate, requiring ATP.
- Mevalonate-5-pyrophosphate is decarboxylated by the ATP-dependent decarboxylase enzyme, mevalonate-5-pyrophosphate decarboxylase, to give isopentenyl pyrophosphate (IPP) and CO 2.
- IPP is isomerized to dimethylallyl pyrophosphate (DMAPP) by isopentenyl pyrophosphate isomerase.
- IPP and DMAPP condense to form geranyl pyrophosphate (GPP, 10-carbon molecule), catalyzed by geranyl pyrophosphate synthase.
- GPP combines with another IPP to produce farnesyl pyrophosphate (FPP, 15-carbon molecule) with the help of the farnesyl pyrophosphate synthase (FDPS).
- Two molecules of FPP are condensed by squalene synthase (FDFT1) to give squalene (30-carbon molecules).
4. Cyclization of Squalene: Creating the Steroid Nucleus (Lanosterol)
- Squalene is cyclized in two steps to produce lanosterol.
- In the first step, squalene epoxidase (SQLE) introduces an epoxide at the 2,3 position, forming 2,3-oxidosqualene; this reaction requires NADPH and O₂.
- The second step involves the lanosterol synthase (LSS), which is a catalyst that helps in catalyzing the cyclization of 2, 3-oxidosqualene to create lanosterol, the initial steroid nucleus.
- This step establishes the four-ring steroid structure, which is essential for all downstream cholesterol and steroid derivatives.
5. Final Processing: Conversion of Lanosterol to Cholesterol
- The conversion of Lanosterol to cholesterol occurs through 19 enzymatic reactions that include demethylation, desaturation, isomerization and reduction.
- The primary human pathway is the Kandutsch-Russell pathway, producing 7-dehydrocholesterol as the immediate precursor.
- Key reactions include reduction of carbon atoms from 30 to 27, removal of two methyl groups at C4 and one at C14, shift of a double bond from C8 to C5, and reduction of the double bond between C24 and C25.
- Important intermediates include desmethyl lanosterol, zymosterol, cholestadienol, and desmosterol.
- The last step is the conversion of 7-dehydrocholesterol into cholesterol with the help of DHCR7.
Regulation of Biosynthesis: Hormonal Control and the SREBP Pathway
Hormonal control
- HMG-CoA reductase is the rate-limiting enzyme in cholesterol biosynthesis, and it regulates the biosynthesis of cholesterol.
- There are two forms of HMG-CoA reductase, the dephosphorylated (active) and the phosphorylated (inactive) forms.
- Insulin and thyroxine stimulate cholesterol production by increasing the production of the active dephosphorylated enzyme.
- The synthesis of cholesterol is inhibited by glucagon and glucocorticoids, which stabilize the phosphorylated form.
SREBP Pathway (Transcriptional Regulation)
- SREBPs (Sterol Regulatory Element-Binding Proteins) are transcription factors that are produced in the ER membrane as inactivated precursors.
- At low cellular cholesterol, SREBPs are bound by SCAP (SREBP cleavage-activating protein) and delivered to the Golgi.
- Site-1 and site-2 proteases cleave SREBP in the Golgi, releasing the active domain to the nucleus.
- Nuclear SREBP stimulates genes of HMG-CoA reductase, HMG-CoA synthase, and other enzymes of the mevalonate pathway that produce cholesterol.
- ER cholesterol elevation inhibits the cleavage of SREBP through INSIG proteins, reducing the transcription of cholesterol biosynthetic genes.
- SREBP is also inhibited by oxysterols (e.g., 25-hydroxycholesterol) as another feedback inhibitor.
Clinical Pharmacology: How Statins Inhibit Cholesterol Synthesis
- The statins are FDA-approved medications used to treat hyperlipidemia and hypercholesterolemia.
- They are reversible competitive inhibitors of the HMG-CoA reductase, the rate-limiting enzyme in cholesterol synthesis.
- Structurally, statins are analogs of HMG-CoA, which means that they can compete with the natural substrate to bind the enzyme.
- HMG-CoA reductase inhibition prevents the conversion of HMG-CoA to mevalonate, thereby inhibiting the de novo synthesis of cholesterol in the liver.
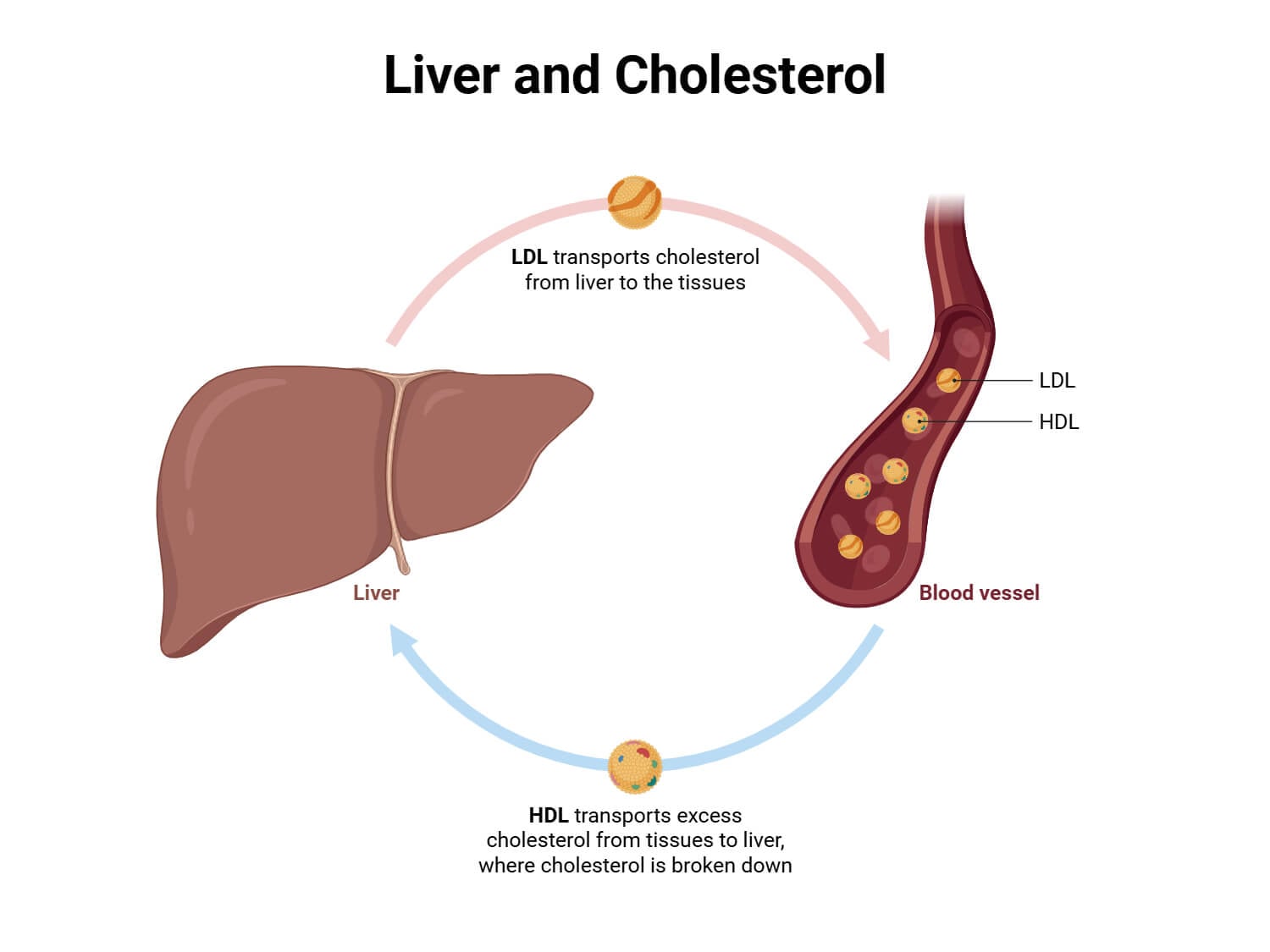
- The low intracellular cholesterol triggers the transcriptional regulation via SREBP and increases the expression of LDL receptors.
- Higher LDL receptors promote the cellular absorption and clearance of blood LDL cholesterol.
- The net pharmacologic action has been lowering plasma LDL cholesterol and cardiovascular risk levels.
- Atorvastatin, simvastatin, and lovastatin are common statins, and they are fungal-derived HMG-CoA reductase inhibitors.
The Fate of Cholesterol: Bile Acid Synthesis and Esterification
Bile Acid Synthesis
- The only place where cholesterol is converted to primary bile acid (cholic acid, CA; chenodeoxycholic acid, CDCA) is in the liver.
- There are two pathways, the classic (neutral) pathway and the alternative (acidic) pathway.
- Cholesterol 7α-hydroxylase (CYP7A1) catalyzes the rate-limiting first step in the classic pathway, converting cholesterol to 7α-hydroxycholesterol.
- Sterol 12α-hydroxylase (CYP8B1) oxidizes C-12 to cholic acid; the lack of which gives CDCA.
- CYP27A1 catalyzes side-chain oxidation, which results in C24 bile acids.
- The alternative pathway begins with CYP27A1 converting cholesterol to 27-hydroxycholesterol, then hydroxylated by CYP7B1 to CDCA.
- Primary bile acids are conjugated with glycine or taurine in the liver and secreted into bile.
- Bacterial enzymes present in the intestine change the primary bile acids into secondary bile acids (deoxycholic acid, lithocholic acid).
- Bile acids play a crucial role in the process of lipid digestion, the elimination of cholesterol, and reverse cholesterol transport between the peripheral tissues and the liver.
Esterification
- Cholesterol is esterified to promote hydrophobicity in order to be safely stored or transported.
- The addition of the fatty acid to the hydroxyl group of cholesterol in the liver is catalyzed by ACAT (acyl-CoA cholesterol acyltransferase). Forming cholesteryl esters (CEs).
- LCAT (lecithin cholesterol acyltransferase) is the one that carries out the same esterification in plasma with HDL particles.
- Cholesteryl esters are more hydrophobic than free cholesterol, allowing storage in lipid droplets or incorporation into VLDL for transport.
- VLDL transports cholesteryl esters, triglycerides, and phospholipids to tissues to synthesize membranes, produce steroid hormones, and vitamin D.
- Esterification retards the toxic accumulation of free cholesterol in cells and facilitates the regulated cholesterol dispersion.
- This is necessary to maintain cholesterol homeostasis and lipoprotein-mediated transportation.
Conclusion
- Cholesterol is a key sterol, and almost half of cholesterol is synthesized de novo, primarily in the liver and the intestine.
- The production of cholesterol takes place in the cytoplasm and endoplasmic reticulum and involves the use of acetyl-CoA, ATP, and NADPH.
- The pathway goes through five steps: formation of HMG-CoA, mevalonate synthesis, isoprenoid units’ formation, squalene formation, and cholesterol formation.
- The rate-limiting and most important regulatory enzyme of the pathway is HMG-CoA reductase.
- Cholesterol plays a critical role in maintaining membrane integrity, synthesizing steroid hormones, making bile acids and synthesizing vitamin D.
- It is regulated through the hormonal mechanisms in which insulin stimulates and glucagon suppresses the production of cholesterol.
- The SREBP pathway gives transcriptional control in response to intracellular cholesterol.
- Statins reduce cholesterol by blocking HMG-CoA reductase competitively and stimulating the expression of the LDL receptor.
- The liver converts cholesterol to bile acids that help in the digestion of lipids and the excretion of cholesterol.
- ACAT and LCAT facilitation of esterification of cholesterol allows safe storage and transportation of cholesteryl esters.
Reference
- Chiang, J. Y. L., Ferrell, J. M., Wu, Y., & Boehme, S. (2020). Bile Acid and Cholesterol Metabolism in Atherosclerotic Cardiovascular Disease and Therapy. Cardiology plus, 5(4), 159–170.
- Craig, M., Yarrarapu, S. N. S., & Dimri, M. (2023, August 8). Biochemistry, cholesterol. In StatPearls.
- Jakubowski, H., & Flatt, R. (n.d.). Lipid biosynthesis. In Fundamentals of Biochemistry. LibreTexts Biology.
- Satyanarayana, U., & Chakrapani, U. (2017). Biochemistry (5th ed.). Elsevier India.
- Scitable. (n.d.). Cholesterol metabolism. Biology Ease.
- Vasudevan, D. M. (n.d.). Cholesterol synthesis, metabolism, and regulation. The Medical Biochemistry Page.
https://themedicalbiochemistrypage.org/cholesterol-synthesis-metabolism-and-regulation/
